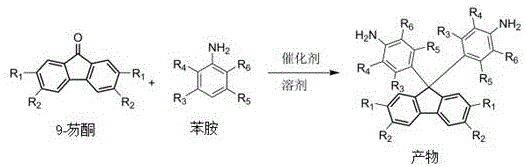
Advanced Synthesis of 9,9-Bis(4-aminophenyl)fluorene Derivatives for Commercial Scale-up
The chemical industry is constantly evolving towards greener, more efficient synthetic pathways, particularly for high-value intermediates used in advanced materials. Patent CN111675621A introduces a transformative synthesis method for 9,9-bis(4-aminophenyl)fluorene derivatives, a critical building block for organic dyes, nonlinear optical materials, and liquid crystal displays. This innovation addresses long-standing challenges in traditional condensation reactions by replacing corrosive liquid acids with a reusable solid acid catalyst, Montmorillonite K10. For R&D directors and procurement specialists seeking a reliable electronic material supplier, this technology represents a significant leap forward in process sustainability and cost-efficiency. By optimizing the molar ratio of reactants and simplifying the post-reaction workup, this method not only enhances product quality but also aligns with stringent environmental regulations governing modern chemical manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9,9-bis(4-aminophenyl)fluorene derivatives has relied heavily on harsh reaction conditions and stoichiometrically excessive reagents that complicate downstream processing. Traditional protocols often utilize strong organic acids like methanesulfonic acid or inorganic acids such as aniline hydrochloride, which necessitate a massive excess of aniline, sometimes up to 10 equivalents, to drive the equilibrium forward. This approach creates severe operational bottlenecks, including the generation of substantial volumes of wastewater contaminated with neutralization salts and residual amines. Furthermore, the use of corrosive hydrogen chloride gas or complex anhydrous salt preparations imposes high maintenance costs on reaction equipment due to corrosion, shortening the lifespan of reactors and increasing capital expenditure. The tedious post-treatment processes, involving multiple washing and neutralization steps, not only increase labor costs but also result in significant product loss, thereby reducing the overall economic viability of producing these high-purity intermediates for the electronics sector.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes Montmorillonite K10, a green solid acid catalyst, to facilitate the condensation of 9-fluorenone with substituted anilines under much milder conditions. This innovative route drastically reduces the consumption of aniline to a molar ratio of merely 1:2 to 1:4 relative to the fluorenone substrate, minimizing the burden on solvent recovery systems. The heterogeneous nature of the catalyst allows for simple filtration to separate the catalyst from the reaction mixture, enabling its direct reuse for multiple batches without significant loss of activity. This elimination of liquid acid catalysts removes the need for aqueous neutralization steps entirely, thereby preventing the formation of salt-laden wastewater and simplifying the isolation of the crude product. The process operates effectively in solvents like chlorobenzene or ethylene glycol monomethyl ether, offering a robust and scalable solution that significantly lowers the environmental footprint while maintaining high conversion rates essential for commercial production.
Mechanistic Insights into Montmorillonite K10-Catalyzed Condensation
The core of this technological advancement lies in the unique catalytic properties of Montmorillonite K10, which acts as a solid Brønsted and Lewis acid to activate the carbonyl group of the 9-fluorenone. The clay surface provides acidic sites that protonate the oxygen atom of the ketone, increasing its electrophilicity and facilitating the nucleophilic attack by the amino group of the aniline derivative. This mechanism proceeds through a hemiaminal intermediate, which subsequently undergoes dehydration to form the stable C-N bonds at the 9-position of the fluorene ring. The presence of a water separator in the reaction setup is critical, as it continuously removes the water byproduct formed during condensation, shifting the chemical equilibrium towards the product side according to Le Chatelier's principle. Unlike homogeneous acid catalysts that remain dissolved and require quenching, the solid acid remains distinct, ensuring that the reaction medium remains free of dissolved ionic species that could complicate purification or degrade sensitive functional groups on the aromatic rings.
Controlling impurity profiles is paramount for applications in optoelectronics, where trace contaminants can adversely affect the performance of final devices. The solid acid catalysis mechanism inherently suppresses side reactions such as polymerization or over-alkylation that are common with stronger liquid acids. The mild acidity of Montmorillonite K10 ensures high chemoselectivity, preserving sensitive substituents like halogens or alkoxy groups on the aniline ring, which is crucial for synthesizing diverse derivatives for specific material properties. Furthermore, the ability to recycle the catalyst reduces the introduction of fresh impurities in each batch, leading to a more consistent impurity spectrum that is easier to manage during the final recrystallization step. This consistency is vital for supply chain heads who require batch-to-batch reproducibility to maintain the quality standards required by downstream manufacturers of liquid crystals and OLED materials.
How to Synthesize 9,9-Bis(4-aminophenyl)fluorene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and safety. The process begins with the sequential addition of 9-fluorenone, the appropriate aniline derivative, the Montmorillonite K10 catalyst, and the chosen solvent into a standard three-neck flask equipped with a stirrer and a water separator. This setup is designed to maximize contact between the solid catalyst and the liquid reactants while efficiently removing water. The reaction is conducted under a nitrogen atmosphere to prevent oxidation of the amine reactants, which is a common issue in high-temperature condensations. Detailed standardized operating procedures regarding temperature control, stirring speeds, and filtration techniques are essential to replicate the high yields reported in the examples, ensuring that the transition from bench scale to commercial manufacturing is seamless and risk-free.
- Charge 9-fluorenone, aniline, Montmorillonite K10 catalyst, and solvent into a reactor equipped with a water separator.
- Protect with Nitrogen, stir, and heat to reflux for 5 to 22 hours to drive the condensation reaction to completion.
- Filter to recover the solid catalyst, distill filtrate to recover solvent and excess aniline, and recrystallize the residue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis method translates into tangible strategic advantages beyond mere technical feasibility. The shift from corrosive liquid acids to a reusable solid catalyst fundamentally alters the cost structure of manufacturing these specialized intermediates. By eliminating the need for expensive corrosion-resistant equipment and reducing the volume of hazardous waste requiring treatment, the overall operational expenditure is significantly lowered. The simplified workup procedure, which avoids complex neutralization and extraction steps, reduces the cycle time per batch, allowing for higher throughput in existing production facilities without the need for major capital investment in new infrastructure. These factors combined create a more resilient supply chain capable of meeting fluctuating market demands for high-performance electronic materials.
- Cost Reduction in Manufacturing: The implementation of Montmorillonite K10 as a catalyst offers profound economic benefits by drastically cutting down on raw material consumption and waste disposal costs. Since the catalyst can be recovered via simple filtration and reused for multiple cycles, the recurring cost of catalytic reagents is minimized compared to single-use liquid acids. Additionally, the reduction in aniline usage from a ten-fold excess to a near-stoichiometric amount significantly decreases the load on solvent recovery distillation columns, saving energy and reducing losses of valuable amine starting materials. The absence of neutralization steps means there is no need to purchase base reagents for quenching, nor is there a cost associated with treating large volumes of saline wastewater, leading to substantial overall cost savings in the production of high-purity electronic chemical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis routes that rely on hazardous or hard-to-handle reagents. This new method enhances reliability by utilizing commercially available, stable solid acids and common organic solvents that are easily sourced from multiple suppliers. The mild reaction conditions reduce the risk of runaway reactions or equipment failure, ensuring consistent production schedules. Furthermore, the robustness of the catalyst allows for flexibility in sourcing; if one batch of catalyst underperforms, it can be easily replaced without halting the entire production line. This stability is crucial for maintaining long-term contracts with downstream clients in the display and semiconductor industries who demand uninterrupted delivery of critical monomer intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this solid-acid catalyzed route is inherently scalable due to its heterogeneous nature. The ease of separating the solid catalyst from the liquid product mixture simplifies the engineering requirements for large-scale reactors, avoiding the complexities of liquid-liquid extraction on a metric ton scale. From an environmental compliance perspective, the process generates significantly less hazardous waste, aligning with increasingly strict global regulations on industrial emissions and effluent discharge. The reduction in three wastes (wastewater, waste gas, and solid waste) not only mitigates regulatory risk but also enhances the corporate sustainability profile, making the final product more attractive to environmentally conscious buyers in the global market.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its potential for integration into their supply chains. The following questions address common concerns regarding catalyst performance, product quality, and process adaptability. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making. Whether you are assessing the feasibility of scaling this process or evaluating the purity specifications for your specific application, these insights clarify the operational realities of implementing this green chemistry solution in a commercial setting.
Q: What are the advantages of using Montmorillonite K10 over traditional acid catalysts?
A: Montmorillonite K10 is a reusable solid acid that eliminates the need for neutralization steps, significantly reducing wastewater containing salts and amines compared to liquid acids like methanesulfonic acid.
Q: Can this process be scaled for industrial production of optoelectronic intermediates?
A: Yes, the process features mild reaction conditions, simple filtration for catalyst recovery, and easy solvent recycling, making it highly suitable for large-scale commercial manufacturing.
Q: What is the typical yield and purity achievable with this method?
A: Experimental data indicates yields ranging from 68% to over 90%, with high purity achievable after recrystallization, depending on the specific aniline substituents and reaction time optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,9-Bis(4-aminophenyl)fluorene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the advancement of next-generation electronic materials. Our team of expert chemists has thoroughly analyzed the patented synthesis route for 9,9-bis(4-aminophenyl)fluorene derivatives and is fully prepared to translate this laboratory innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green catalytic process are realized at the volume your business requires. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of material delivered meets the exacting standards necessary for high-performance optical and electronic applications.
We invite you to collaborate with us to optimize your supply chain for these valuable compounds. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can secure a sustainable and cost-effective supply of 9,9-bis(4-aminophenyl)fluorene derivatives, empowering your R&D efforts and ensuring your position at the forefront of the electronic materials industry.
