Advanced Manufacturing of Esomeprazole Intermediate Mufelazole via Optimized Condensation
Advanced Manufacturing of Esomeprazole Intermediate Mufelazole via Optimized Condensation
The global demand for high-purity proton pump inhibitors continues to drive innovation in the synthesis of their key precursors. Patent CN107400118B discloses a significantly improved preparation method for mufelazole, a critical intermediate in the production of esomeprazole sodium. This technical breakthrough addresses long-standing challenges in yield optimization and impurity control that have plagued traditional manufacturing routes. By refining the condensation reaction between 5-methoxy-2-mercapto-1H-benzimidazole and 2-chloromethyl-3,5-dimethyl-4-methoxypyridine hydrochloride, the inventors have established a robust protocol suitable for industrial scale-up. The structural complexity of esomeprazole, featuring a chiral sulfinyl benzimidazole core, necessitates precise intermediate synthesis to ensure downstream stereochemical integrity.
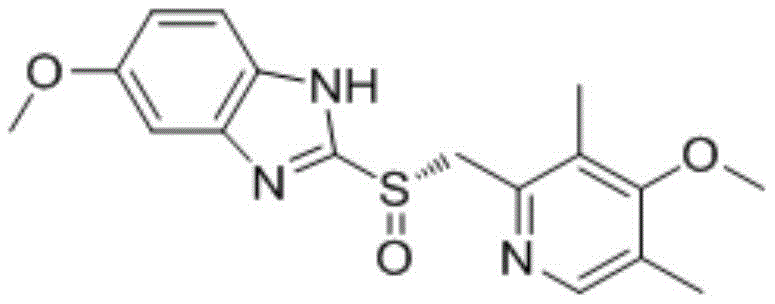
For procurement managers and supply chain directors, understanding the nuances of this intermediate's synthesis is vital for securing a reliable pharma intermediates supplier. The patent details specific reaction conditions that eliminate the need for toxic reagents found in older methodologies, thereby reducing environmental compliance costs and enhancing worker safety. Furthermore, the optimized workup procedure simplifies the removal of inorganic byproducts, directly translating to reduced processing time and lower energy consumption during solvent recovery. This report analyzes the technical merits of this novel approach and its implications for cost reduction in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of wufelazole (mufelazole) has been approached through several distinct pathways, each carrying significant drawbacks for large-scale production. One common prior art route involves the use of p-methoxypyridine mesylate, which requires the handling of highly toxic and corrosive methane sulfonyl chloride or anhydride. This not only poses severe safety risks but also generates hazardous waste streams that are costly to treat. Another prevalent method utilizes a nitro-substituted pyridine intermediate, where the nitro group is subsequently displaced by a methoxy group. However, the structural similarity between the nitro-intermediate and the final product makes it exceptionally difficult to remove trace amounts of unreacted starting material, leading to potential genotoxic impurity concerns in the final API.
Additionally, earlier attempts at direct condensation often suffered from incomplete reactions or excessive impurity generation when standard stoichiometric ratios were employed. For instance, using a simple 1:1 molar ratio frequently resulted in residual starting materials that complicated purification. Conversely, adding excess alkylating agent to drive the reaction to completion often triggered side reactions, generating unknown impurities that required multiple recrystallization steps to remove. These inefficiencies highlight the need for a more precise and controlled synthetic strategy that balances reactivity with selectivity.
The Novel Approach
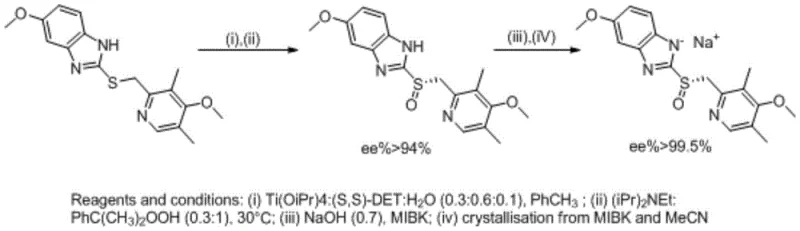
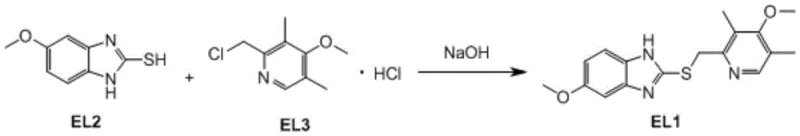
The methodology described in CN107400118B introduces a refined condensation protocol that effectively overcomes these historical hurdles. The core innovation lies in the precise control of reactant stoichiometry and the implementation of a binary solvent system. By utilizing a specific molar ratio of 1:1.05:2.05 for the mercaptan, the chloromethyl pyridine salt, and sodium hydroxide respectively, the process ensures complete consumption of the limiting reagent without triggering significant side reactions. This delicate balance prevents the accumulation of difficult-to-remove impurities that typically arise from reagent excess.
Furthermore, the adoption of an ethanol and acetone mixed solvent system (3:1 mass ratio) represents a significant process improvement. Unlike pure ethanol, which forms azeotropes complicating solvent recovery, or ethyl acetate mixtures, which cause inorganic salts to agglomerate and halt the reaction, this specific blend facilitates smooth reaction kinetics and efficient solid-liquid separation. The result is a streamlined workflow where sodium chloride byproducts can be simply filtered off at room temperature, drastically simplifying the isolation of the crude product and setting the stage for high-yield crystallization.
Mechanistic Insights into Nucleophilic Substitution and Solvent Effects
The reaction mechanism fundamentally relies on the nucleophilic attack of the deprotonated mercaptobenzimidazole anion on the electrophilic chloromethyl carbon of the pyridine derivative. The presence of sodium hydroxide is critical not only for generating the reactive thiolate species but also for neutralizing the hydrochloride salt of the pyridine substrate. However, the concentration of base and the solvation environment profoundly influence the reaction pathway. In the optimized process, the 2.05 equivalents of NaOH ensure rapid deprotonation while maintaining a basic environment that suppresses acid-catalyzed degradation of the sensitive benzimidazole ring. The slight excess of the pyridine electrophile (1.05 eq) drives the equilibrium forward according to Le Chatelier's principle, ensuring that the valuable mercaptan is fully converted.
Impurity control is achieved through the careful modulation of reaction temperature and solvent polarity. At temperatures exceeding 45°C, the kinetic energy of the molecules increases the likelihood of non-selective alkylation or decomposition, leading to a darker reaction mass and lower purity. Conversely, temperatures below 40°C result in sluggish kinetics, leaving unreacted starting materials that are difficult to separate. The ethanol/acetone mixture provides an ideal dielectric environment that solubilizes both the organic reactants and the ionic intermediates sufficiently for the reaction to proceed, yet allows the generated sodium chloride to precipitate or remain insoluble enough for filtration. This physical property difference is exploited to remove the bulk of inorganic contaminants before the final crystallization step, significantly reducing the load on downstream purification units.
How to Synthesize Mufelazole Efficiently
The synthesis of mufelazole described in this patent offers a practical and scalable route for manufacturing facilities aiming to produce high-quality esomeprazole intermediates. The process is designed to be operationally simple, avoiding the need for cryogenic conditions or exotic catalysts. By adhering to the specified parameters regarding temperature, stoichiometry, and solvent composition, manufacturers can consistently achieve yields around 94% with purity levels exceeding 95%. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for process engineers looking to implement this technology.
- Prepare the reaction mixture by dissolving sodium hydroxide in absolute ethanol, then add the mercaptobenzimidazole derivative and the chloromethyl pyridine hydrochloride salt.
- Maintain the reaction temperature strictly between 40-45°C using a mixed solvent system of ethanol and acetone (3: 1 mass ratio) to ensure complete conversion while minimizing impurities.
- Upon completion, filter off the inorganic salts at room temperature, concentrate the filtrate, and crystallize the crude product from ethyl acetate to obtain high-purity Mufelazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this optimized synthesis route offers tangible strategic benefits beyond mere technical performance. The elimination of toxic reagents like methane sulfonyl chloride removes a significant regulatory burden and reduces the cost associated with hazardous waste disposal. Moreover, the simplified workup procedure, which allows for the direct filtration of inorganic salts, minimizes the number of unit operations required. This reduction in processing steps directly correlates to lower utility consumption and shorter batch cycle times, enhancing the overall throughput of the manufacturing facility.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through yield optimization and solvent management. By preventing the formation of hard-to-remove impurities, the need for multiple recrystallizations or chromatographic purifications is eliminated, saving both time and materials. Additionally, the use of commodity solvents like ethanol and acetone, which are readily available and inexpensive compared to specialized organic solvents, further drives down the variable cost of goods sold. The high conversion rate ensures that expensive starting materials are not wasted, maximizing the return on raw material investment.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as the key precursors are established commodities in the fine chemical sector. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in raw material quality or environmental conditions, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining uninterrupted supply lines to downstream API manufacturers, reducing the risk of stockouts or production delays that can ripple through the pharmaceutical supply chain.
- Scalability and Environmental Compliance: The method is inherently scalable, having been demonstrated effectively at multi-kilogram scales in the patent examples. The absence of heavy metal catalysts or persistent organic pollutants aligns well with modern green chemistry principles and stringent environmental regulations. This compliance facilitates easier permitting for new production lines and reduces the long-term liability associated with environmental remediation, making it a sustainable choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis method. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on critical process parameters and quality control measures. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the ethanol/acetone solvent system preferred over pure ethanol or ethyl acetate mixtures?
A: Pure ethanol allows complete reaction but complicates post-treatment due to azeotropes. Ethyl acetate mixtures cause inorganic salts to agglomerate, halting the reaction. The 3:1 ethanol/acetone mix balances solubility for reaction progress with the ability to filter out sodium chloride efficiently.
Q: What is the critical stoichiometric ratio for maximizing yield in this process?
A: The optimal molar ratio of the mercaptobenzimidazole (EL2), the chloromethyl pyridine derivative (EL3), and sodium hydroxide is determined to be 1:1.05:2.05. Deviating from this, such as increasing EL3 to 1.07 equivalents, leads to significant impurity formation.
Q: How does this method improve upon prior art routes involving nitro-intermediates?
A: Prior routes involving nitro-group substitution often leave difficult-to-remove nitro-impurities due to structural similarity. This direct condensation method using the pre-methoxylated pyridine derivative avoids these specific purification challenges, resulting in higher purity (>95%).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mufelazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the efficacy and safety of final pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering mufelazole and other complex intermediates with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can enhance your project's success.
