Advanced One-Step Esterification for High-Purity Cyclohexane Dicarboxylic Acid Monoesters
The rapid evolution of the display and optoelectronic industries demands increasingly sophisticated intermediates capable of delivering precise optical anisotropy and thermal stability. Patent CN115850070A introduces a groundbreaking synthesis method for cyclohexane dicarboxylic acid monoester compounds, which serve as critical side-chain segments in the formation of liquid crystal materials. This technology addresses the longstanding challenges of selectivity and safety in esterification reactions by utilizing a modified carbodiimide coupling strategy. By leveraging 1-ethyl-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDCI) with precisely controlled hydration levels, the process achieves exceptional mono-selectivity without resorting to hazardous activating agents. For procurement teams and R&D directors alike, this represents a significant shift towards safer, more scalable electronic chemical manufacturing protocols that align with modern green chemistry principles.
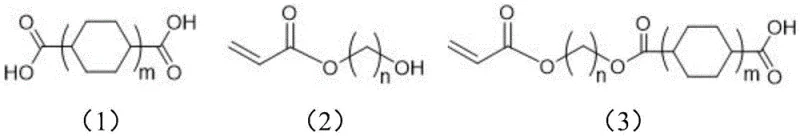
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these vital monoester intermediates has been plagued by inefficient multi-step pathways and the reliance on dangerous reagents that complicate supply chain management. Traditional approaches, such as those cited in prior art, often employ methanesulfonyl chloride as an activating agent, which is classified as a highly toxic substance subject to strict purchasing restrictions and handling regulations. Furthermore, alternative methods involving acid chloride formation require thionyl chloride and multiple reaction steps, leading to cumulative yield losses and significant waste generation. High-temperature esterification routes frequently suffer from poor selectivity, resulting in substantial quantities of diester byproducts that are difficult to separate from the desired monoester target. These purification bottlenecks often necessitate resource-intensive column chromatography, a technique that is notoriously difficult to translate from laboratory benchtop to industrial commercial scale-up. Consequently, manufacturers face inflated production costs and extended lead times due to the complexity of waste disposal and the low throughput of batch purification processes.
The Novel Approach
The methodology disclosed in CN115850070A revolutionizes this landscape by introducing a direct, one-step esterification protocol that operates under remarkably mild conditions. By employing EDCI as a dehydrating agent in conjunction with 4-dimethylaminopyridine (DMAP) as a nucleophilic catalyst, the reaction proceeds efficiently at temperatures ranging from -10°C to 25°C. This approach completely eliminates the need for toxic chlorinating agents like methanesulfonyl chloride or thionyl chloride, thereby removing significant regulatory hurdles and safety risks from the production facility. The core innovation lies in the strategic modulation of the EDCI reagent's water content, which serves as a kinetic control mechanism to favor mono-esterification over di-esterification. This results in a reaction mixture where the target monoester constitutes over 80% of the crude content, drastically simplifying the downstream purification requirements. As a reliable electronic chemical supplier, adopting this route allows for the use of common solvents like dichloromethane or toluene and enables purification via simple recrystallization rather than chromatography.
Mechanistic Insights into EDCI-Mediated Selective Esterification
The success of this synthesis hinges on the nuanced understanding of carbodiimide chemistry and the specific role of trace water in modulating the reactivity of the O-acylisourea intermediate. In a standard EDCI coupling, the carboxylic acid reacts with the carbodiimide to form a highly reactive O-acylisourea species, which is then attacked by the alcohol nucleophile to form the ester bond. However, in the case of dicarboxylic acids, the presence of a second carboxyl group creates a risk of double substitution, leading to the unwanted diester byproduct. The patent reveals that the water content within the EDCI hydrochloride salt acts as a subtle 'brake' on the reaction kinetics. When the water content is too low (below 1.0wt%), the dehydrating power of EDCI is too aggressive, promoting the rapid activation of both carboxyl groups and leading to high levels of diester impurities. Conversely, excessive water (above 3.0wt%) quenches the activated intermediate prematurely, resulting in poor conversion and high recovery of starting materials.
By maintaining the EDCI water content strictly between 1.0wt% and 3.0wt%, the process achieves an optimal balance where the first esterification occurs readily, but the steric and electronic environment of the mono-ester intermediate discourages the second activation event. This selectivity is further enhanced by the molar ratio of reactants, specifically using a slight deficit of the hydroxyalkyl acrylate relative to the dicarboxylic acid. The DMAP catalyst accelerates the acylation step by forming a more reactive acylpyridinium intermediate, ensuring the reaction completes within 1 to 8 hours without requiring thermal stress. This mechanistic control ensures that the crude reaction profile is clean, with gas chromatography (GC) analysis showing monoester content exceeding 80% before any purification takes place. Such high crude purity is essential for minimizing solvent usage and energy consumption during the final isolation stages, directly contributing to cost reduction in electronic chemical manufacturing.

How to Synthesize Cyclohexane Dicarboxylic Acid Monoester Efficiently
Implementing this synthesis requires strict adherence to the moisture control parameters and stoichiometric ratios defined in the patent to ensure reproducible high yields. The process begins with the dissolution of the dicarboxylic acid and catalyst in an anhydrous solvent, followed by the careful addition of the conditioned EDCI reagent. Temperature control is maintained throughout the addition of the acrylate component to prevent exothermic spikes that could degrade the sensitive acrylate functionality. While the general procedure is robust, the specific stoichiometry and workup details are critical for achieving the reported 99%+ purity specifications required for high-end display applications. The following guide outlines the standardized operational framework derived from the patent's preferred embodiments.
- Dissolve trans-1,4-cyclohexanedicarboxylic acid and DMAP catalyst in dichloromethane or toluene under nitrogen protection and reflux briefly.
- Cool the mixture to 20-25°C and add EDCI hydrochloride with a controlled water content of 1.0wt% to 3.0wt% to modulate reactivity.
- Dropwise add the hydroxyalkyl acrylate solution, maintain temperature for 1-8 hours, and purify the crude product via aqueous washing and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this EDCI-mediated pathway offers profound logistical and economic benefits compared to legacy synthetic routes. The elimination of highly regulated toxic precursors simplifies the procurement process, removing the need for special permits and reducing the administrative burden associated with hazardous material management. Furthermore, the ability to purify the product through crystallization rather than column chromatography represents a massive leap in process scalability, allowing for the efficient production of metric-ton quantities without proportional increases in silica gel waste or solvent consumption. This streamlined workflow not only shortens the overall cycle time but also enhances the reliability of supply by reducing the number of potential failure points in the manufacturing sequence. Companies seeking a reliable supplier for these intermediates will find that this method supports consistent batch-to-batch quality essential for long-term contracts.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous activating agents like methanesulfonyl chloride with commodity-grade EDCI and DMAP significantly lowers the raw material cost base. Additionally, the simplified workup procedure, which relies on aqueous washing and recrystallization instead of chromatography, drastically reduces the consumption of high-purity solvents and stationary phases. This reduction in consumable usage translates directly into lower variable costs per kilogram of finished product, improving overall margin structures. The mild reaction conditions also reduce energy expenditures associated with heating and cooling, further contributing to operational efficiency.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents and avoiding supply-constrained toxic chemicals, the risk of production stoppages due to raw material shortages is substantially mitigated. The robustness of the reaction against minor fluctuations in conditions ensures high reproducibility, which is critical for maintaining steady inventory levels for downstream clients. Moreover, the reduced generation of hazardous waste simplifies disposal logistics and lowers compliance costs, ensuring uninterrupted operation even under tightening environmental regulations. This stability makes the supply chain more resilient to external shocks and regulatory changes.
- Scalability and Environmental Compliance: The one-pot nature of the reaction and the use of standard organic solvents facilitate easy scale-up from pilot plants to full-scale commercial reactors without the need for specialized equipment. The absence of heavy metal catalysts or corrosive acid chlorides minimizes the corrosion risk to reactor vessels and reduces the load on wastewater treatment facilities. This alignment with green chemistry principles not only improves the environmental footprint of the manufacturing process but also enhances the brand value for end-users seeking sustainable sourcing options. The process is inherently designed for commercial scale-up of complex electronic chemicals with minimal environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method based on the patent data. Understanding these nuances is vital for process engineers aiming to replicate the high purity and yield metrics reported in the literature. The answers provided are derived directly from the experimental examples and comparative data found in CN115850070A.
Q: Why is the water content of EDCI critical in this synthesis?
A: The water content of EDCI (1.0wt%-3.0wt%) acts as a selectivity regulator; too little water promotes diester formation, while too much reduces conversion efficiency.
Q: How does this method improve upon traditional methanesulfonyl chloride routes?
A: This method eliminates the use of highly toxic methanesulfonyl chloride, operates at mild temperatures (20-25°C), and avoids complex multi-step sequences.
Q: What is the typical purity achievable with this recrystallization process?
A: Following the specified aqueous workup and toluene/methylcyclohexane recrystallization, GC purity levels exceeding 99.0% are consistently achieved.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexane Dicarboxylic Acid Monoester Supplier
The technological advancements detailed in CN115850070A underscore the potential for producing high-performance liquid crystal intermediates with unprecedented efficiency and safety. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the critical moisture content of reagents and the isomeric purity of the final products, guaranteeing consistency across every batch. We are committed to translating these innovative academic and patent findings into robust industrial processes that drive value for our global partners.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to secure your supply chain with high-quality, sustainably produced electronic chemical intermediates.
