Advanced Copper-Catalyzed Oxidative Thiocyanation for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Copper-Catalyzed Oxidative Thiocyanation for Scalable Pharmaceutical Intermediate Manufacturing
The synthesis of alpha-thiocyanated ketones represents a critical transformation in the production of high-value fine chemicals, particularly within the realms of pharmaceutical and agrochemical intermediate manufacturing. As detailed in the groundbreaking patent CN102320909B, a novel synthetic methodology has been established that utilizes molecular oxygen or air as a sustainable oxidant in conjunction with a copper salt catalyst. This technical insight report provides a comprehensive analysis of this oxidative thiocyanation protocol, highlighting its potential to revolutionize the supply chain for reliable pharmaceutical intermediate suppliers. By replacing hazardous stoichiometric oxidants with benign atmospheric oxygen, this process addresses long-standing challenges regarding waste management, cost efficiency, and operational safety in the production of sulfur-containing heterocyclic precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of alpha-thiocyanated ketones has relied heavily on multi-step sequences involving the initial alpha-halogenation of ketones followed by nucleophilic substitution with thiocyanate salts. This traditional pathway is fraught with significant inefficiencies, including the requirement for harsh reaction conditions and the generation of substantial amounts of halogenated waste streams. Furthermore, alternative direct oxidative methods reported in prior art often necessitate the use of expensive and potentially hazardous stoichiometric oxidants such as hypervalent iodine reagents or peroxides. These conventional systems not only inflate the raw material costs drastically but also complicate the downstream purification processes due to the presence of difficult-to-remove oxidant byproducts, thereby limiting their viability for large-scale commercial application.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in patent CN102320909B introduces a streamlined catalytic cycle driven by earth-abundant copper salts and molecular oxygen. This innovative approach enables the direct construction of the carbon-sulfur bond under remarkably mild conditions, typically ranging from 0°C to 100°C, without the need for pre-functionalization of the ketone substrate. The utilization of air or oxygen as the terminal oxidant ensures that the atom economy is maximized, with water being the sole reduction byproduct. This paradigm shift not only simplifies the reaction workflow but also aligns perfectly with modern green chemistry mandates, offering a robust solution for cost reduction in pharmaceutical intermediates manufacturing while maintaining high product purity standards.
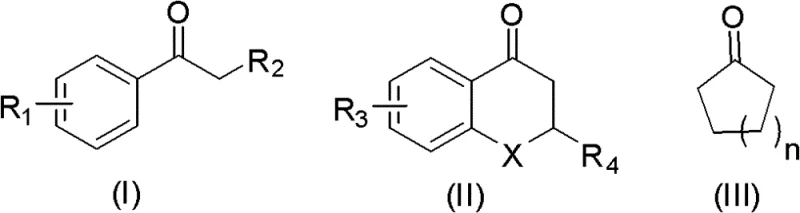
Mechanistic Insights into Copper-Catalyzed Oxidative Thiocyanation
The core of this technological advancement lies in the synergistic interaction between the copper catalyst, the bromide promoter, and the molecular oxygen oxidant. Mechanistically, the copper species facilitates the activation of the alpha-carbon of the ketone, likely through an enol or enolate intermediate, while the bromide source assists in the generation of reactive radical species or electrophilic bromine in situ. This activation allows for the efficient coupling with the thiocyanate anion, overcoming the inherent weak nucleophilicity of the SCN group that often plagues traditional substitution reactions. The catalytic nature of the copper salt means that only substoichiometric amounts are required, typically in the range of 0.1 to 0.6 molar equivalents relative to the substrate, which significantly reduces the burden of heavy metal removal during the workup phase.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring the quality of the final API intermediate. Traditional strong oxidants often lead to over-oxidation of the ketone or degradation of sensitive functional groups on the aromatic ring, resulting in complex impurity profiles that are difficult to separate. By operating at moderate temperatures of 40°C to 80°C and utilizing the selective oxidation power of the copper-oxygen system, side reactions are minimized. This selectivity is crucial for producing high-purity OLED material or pharmaceutical precursors where trace impurities can have detrimental effects on downstream biological activity or material performance, thus validating the method's suitability for stringent quality specifications.
How to Synthesize Alpha-Ketone Thiocyanate Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins with the simple mixing of the ketone substrate, such as p-bromoacetophenone or cyclopentanone, with a thiocyanate salt like potassium thiocyanate in a polar solvent. The addition of a copper catalyst, such as cuprous iodide or copper acetate, along with a catalytic amount of hydrobromic acid or bromine, initiates the system. The reaction is then conducted under a pressurized atmosphere of oxygen or air, allowing for the continuous regeneration of the active catalytic species. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the ketone substrate, thiocyanate salt (e.g., KSCN), copper catalyst, and hydrobromic acid promoter in a suitable solvent such as acetic acid or methanol.
- Seal the reaction vessel and establish an oxygen or air atmosphere at a pressure of 0.1 to 0.5 MPa to serve as the green oxidant.
- Heat the mixture to a temperature between 40°C and 80°C for 5 to 24 hours, followed by aqueous workup and chromatographic purification to isolate the pure alpha-thiocyanated ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this oxidative thiocyanation technology offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The elimination of expensive stoichiometric oxidants and the use of commodity chemicals like air and copper salts fundamentally alter the cost structure of production. This shift translates into significant margin improvements and reduced exposure to volatile pricing of specialized reagents. Furthermore, the simplified workup procedure, which avoids complex quenching of hazardous oxidants, reduces the overall cycle time and labor costs associated with manufacturing, thereby enhancing the overall competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The replacement of costly oxidants like Oxone or hypervalent iodine with free or low-cost molecular oxygen results in a drastic reduction in raw material expenses. Additionally, the catalytic loading of copper is low, and the solvents used are common industrial grades like acetic acid or ethanol, which are readily available at bulk prices. This combination of factors leads to substantial cost savings per kilogram of product, allowing for more aggressive pricing strategies in the competitive fine chemical market without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Relying on air or oxygen as the oxidant removes the dependency on supply chains for specialized oxidizing agents, which can sometimes be subject to regulatory restrictions or availability fluctuations. The raw materials, including various ketones and thiocyanate salts, are commodity chemicals produced globally in massive quantities. This abundance ensures a stable and continuous supply of inputs, minimizing the risk of production stoppages due to raw material shortages and guaranteeing consistent delivery schedules to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The green nature of this process, characterized by water as the only byproduct, significantly simplifies waste treatment and disposal protocols. This reduces the environmental compliance burden and associated costs for wastewater treatment facilities. Moreover, the mild reaction conditions and the use of standard pressure vessels make the process inherently safer and easier to scale from pilot plant to multi-ton commercial production, facilitating rapid capacity expansion to meet growing market demand for these valuable intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed oxidative thiocyanation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on substrate scope, reaction parameters, and environmental impact. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs.
Q: What are the primary advantages of this oxidative thiocyanation method over traditional halogenation routes?
A: Unlike traditional methods requiring pre-halogenation and harsh nucleophilic substitution, this patent describes a direct oxidative coupling using air or oxygen. This eliminates stoichiometric oxidants, reduces waste generation to merely water, and significantly lowers raw material costs while improving operational safety.
Q: Which types of ketone substrates are compatible with this copper-catalyzed system?
A: The methodology demonstrates broad substrate scope, successfully converting both aromatic ketones (such as acetophenones and tetralones) and aliphatic cyclic ketones (including cyclopentanone and cyclohexanone) into their corresponding alpha-thiocyanated derivatives with moderate to high efficiency.
Q: How does the use of molecular oxygen impact the environmental profile of the synthesis?
A: Utilizing molecular oxygen or air as the terminal oxidant ensures that the only reduction byproduct is water. This fundamentally shifts the process towards green chemistry principles, avoiding the toxic heavy metal waste or iodine residues associated with stoichiometric oxidants like Oxone or hypervalent iodine reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketone Thiocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed oxidative thiocyanation technology described in CN102320909B for the production of high-value sulfur-containing intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of alpha-ketone thiocyanate meets the exacting standards required by global pharmaceutical and agrochemical innovators.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can optimize your supply chain and reduce your overall cost of goods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your project timelines and enhance your product portfolio.
