Breakthrough Synthesis of High-Purity 2,4-Bis(Phenylazo)Resorcinol B-Type Isomer for Advanced Stealth Coatings
The global demand for advanced wave-absorbing materials in the aerospace and defense sectors has intensified the search for high-performance organic intermediates capable of precise electromagnetic modulation. Patent CN110305037B, published in early 2022, introduces a transformative synthesis method for the selective production of the B-type isochromic isomer of 2,4-bis(phenylazo)resorcinol. This specific isomer is critical for next-generation stealth coatings due to its exceptional ultraviolet absorption and radar wave attenuation capabilities. Unlike previous methodologies that struggled with isomer separation and low yields, this invention leverages a sophisticated aqueous-phase coupling strategy utilizing resorcinol monopotassium salt and specialized phase transfer catalysts. The technical breakthrough lies not only in the chemical transformation but in the precise control of molecular geometry through hydrogen bonding manipulation, ensuring the final product meets the rigorous purity standards required for optical and electronic applications.
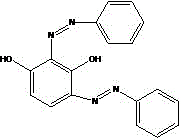
For R&D directors evaluating process feasibility, understanding the limitations of conventional synthesis routes is paramount to appreciating the value of this new technology. Historically, the synthesis of 2,4-bis(phenylazo)resorcinol has been plagued by poor regioselectivity and complex impurity profiles. Direct coupling of resorcinol with aniline diazonium salt typically results in a chaotic mixture containing mono-azo derivatives, the undesired 4,6-bis(phenylazo) isomer, and over-reacted 2,4,6-tris(phenylazo) byproducts. Furthermore, the seminal work by Professor Herbert H. Hodgson proposed a route involving m-halophenols (m-chloro, m-bromo, or m-iodo phenols) followed by hydrolysis. While chemically sound, this approach is commercially prohibitive due to the exorbitant cost and scarcity of m-halophenols, particularly m-bromophenol and m-iodophenol, which are often unavailable domestically and command prices exceeding significant thresholds per ton. Additionally, the hydrolysis of the chloro-intermediate is notoriously difficult, often leaving residual halogenated structures that compromise the purity of the final dye.
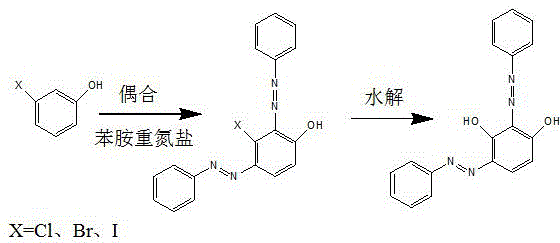
In stark contrast, the novel approach detailed in the patent circumvents these economic and technical bottlenecks by utilizing inexpensive, commodity-grade resorcinol as the starting substrate. The innovation centers on the in-situ formation of resorcinol monopotassium salt, which is then subjected to a coupling reaction with aniline diazonium salt in a water phase. This method eliminates the need for hazardous organic solvents during the reaction phase and avoids the use of expensive halogenated precursors entirely. By optimizing the molar ratio of reactants and employing a specific pH buffering system, the process achieves a molar conversion rate of the target B-type structural isomer exceeding 90 percent based on resorcinol. This represents a substantial leap in efficiency compared to prior art, which often yielded mixtures requiring extensive and yield-eroding purification steps. The ability to directly access the target isomer from cheap raw materials fundamentally alters the cost structure of manufacturing this high-value functional material.
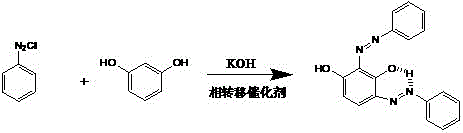
Mechanistically, the success of this synthesis relies on a delicate interplay between phase transfer catalysis and pH-controlled regioselectivity. The reaction employs crown ether compounds, such as 18-crown-6 or dibenzo-24-crown-8, or multi-nitrogen macrocyclic heterocycles like cyclen. These catalysts function by forming stable supramolecular complexes with the potassium ions of the resorcinol monopotassium salt. This complexation serves a dual purpose: it significantly enhances the solubility of the organic salt in the aqueous medium and activates the aromatic ring towards electrophilic attack by the diazonium ion. Without this catalyst, the solubility of the salt would be insufficient at the mild operating temperatures of 15 to 20°C, leading to precipitation and low conversion. Furthermore, the patent highlights the critical role of pH in directing the formation of the specific B-type isomer. Research indicates that the N and H atoms in the molecule form weak hydrogen bonds, and the configuration of these bonds determines the isomeric form (A, B, C, or D).
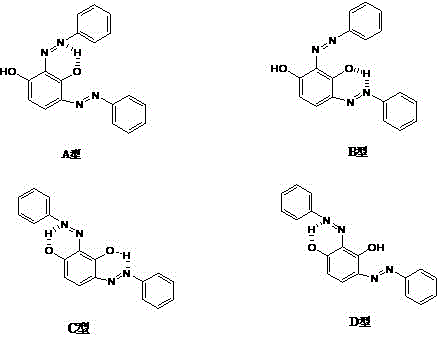
Experimental data within the patent demonstrates that maintaining the reaction pH strictly between 5.5 and 5.8 is essential for maximizing the yield of the B-type isomer. At pH values above 10, the reaction favors the 4,6-bis isomer, while pH levels between 7 and 10 promote the formation of the tris-azo impurity. The use of a disodium hydrogen phosphate buffer allows for precise tracking and maintenance of this narrow acidic window throughout the dropwise addition of the diazonium salt. This level of control ensures that the hydrogen bonding network stabilizes the desired B-type configuration, which is characterized by its wine-red color and superior solubility in ethyl lactate. For quality control teams, this mechanistic insight provides clear critical process parameters (CPPs) to monitor, ensuring batch-to-batch consistency and minimizing the formation of hard-to-remove impurities like the mono-azo derivatives which have moderate solubility in most organic solvents.
How to Synthesize 2,4-Bis(Phenylazo)Resorcinol Efficiently
The synthesis protocol outlined in the patent offers a robust framework for scaling this reaction from laboratory to commercial production. The process begins with the preparation of the resorcinol monopotassium salt solution, followed by the careful addition of the phase transfer catalyst to ensure complete complexation. The subsequent coupling step requires precise temperature control and pH monitoring to drive the reaction towards the desired bis-azo product while suppressing tris-azo formation. Following the reaction, a unique purification strategy involving DMF reflux and ethyl lactate recrystallization is employed to isolate the high-purity B-type isomer.
- Preparation of Resorcinol Monopotassium Salt: React resorcinol with equimolar potassium hydroxide in water, then add a crown ether phase transfer catalyst.
- Controlled Coupling Reaction: Add aniline diazonium salt solution dropwise at 15-20°C while maintaining pH between 5.5 and 5.8 using a phosphate buffer.
- Purification and Isolation: Remove tris-azo impurities via DMF reflux, then recrystallize the filtrate in ethyl lactate to isolate the pure B-type isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from halogenated substrates to commodity resorcinol represents a significant strategic advantage in terms of cost stability and sourcing security. The traditional reliance on m-bromophenol or m-iodophenol exposes manufacturers to volatile pricing and supply chain disruptions, as these materials are often imported specialty chemicals with limited supplier bases. By switching to resorcinol, a widely available bulk chemical, companies can drastically simplify their supply chain logistics and reduce raw material expenditure. Furthermore, the elimination of halogenated intermediates removes the need for complex hydrolysis steps and the associated waste treatment costs for halogen-containing effluents. This simplification of the process flow not only lowers the operational expenditure but also aligns with increasingly stringent environmental regulations regarding halogenated waste disposal.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this manufacturing process is the substitution of expensive, scarce m-halophenols with low-cost resorcinol. The patent explicitly notes that imported m-bromophenol and m-iodophenol can cost hundreds of thousands of RMB per ton, whereas resorcinol is a commodity chemical produced at a fraction of that price. Additionally, the high molar conversion rate of over 90 percent minimizes raw material waste, ensuring that the majority of the input mass is converted into valuable product rather than discarded impurities. The use of an aqueous phase reaction system further reduces costs by eliminating the need for large volumes of expensive organic solvents during the synthesis stage, thereby lowering both solvent purchase and recovery costs.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly enhanced because the key raw materials, resorcinol and aniline, are produced globally in massive quantities for various industrial applications. This abundance ensures that production schedules are not held hostage by the availability of niche intermediates. The simplified reaction conditions, operating at mild temperatures of 15 to 20°C and atmospheric pressure, also reduce the risk of equipment failure or safety incidents that could disrupt supply. The robustness of the phase transfer catalytic system means that the process is less sensitive to minor fluctuations in operating conditions, leading to more consistent output and reliable delivery timelines for downstream customers in the electronics and defense sectors.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation in a homogeneous aqueous phase, which facilitates efficient heat transfer and mixing in large reactors. The purification steps, involving filtration and recrystallization, are standard unit operations that can be easily scaled without requiring specialized equipment. From an environmental perspective, the avoidance of halogenated reagents significantly reduces the toxicity of the waste stream. The primary waste products are inorganic salts and aqueous effluents that are easier to treat compared to halogenated organic waste. This ease of waste management lowers the barrier for regulatory compliance and reduces the long-term liability associated with hazardous waste disposal, making the facility more sustainable and resilient against future environmental policy changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized azo compound. These answers are derived directly from the experimental data and technical disclosures found in the patent documentation, providing a reliable basis for decision-making.
Q: Why is the B-type isomer of 2,4-bis(phenylazo)resorcinol preferred for wave-absorbing applications?
A: The B-type isomer exhibits superior ultraviolet absorption and radar wave absorption properties compared to other isomers. Crucially, it demonstrates high solubility in ethyl lactate, forming clear, transparent solutions without flocculent insoluble substances, which is essential for uniform coating application in aerospace and defense industries.
Q: How does this new synthesis method reduce raw material costs compared to traditional routes?
A: Traditional methods taught by Professor Hodgson rely on expensive and scarce m-halophenols (such as m-bromophenol or m-iodophenol), which can cost hundreds of thousands of RMB per ton. This patented method utilizes commodity-grade resorcinol and aniline, significantly lowering the Bill of Materials (BOM) cost while avoiding the difficult hydrolysis steps associated with halogenated intermediates.
Q: What is the role of the phase transfer catalyst in this aqueous coupling reaction?
A: The phase transfer catalyst, specifically crown ethers like 18-crown-6 or dibenzo-24-crown-8, forms a supramolecular complex with the potassium ions of the resorcinol monopotassium salt. This complexation dramatically increases the solubility of the organic salt in the aqueous phase and enhances its nucleophilicity, allowing the coupling reaction to proceed efficiently under mild conditions (15-20°C) with high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Bis(Phenylazo)Resorcinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of advanced functional materials. Our team of expert chemists has thoroughly analyzed the synthetic route described in CN110305037B and possesses the technical capability to implement this efficient, cost-effective process on a commercial scale. We understand that transitioning from laboratory discovery to industrial production requires more than just a recipe; it demands rigorous process optimization and quality control. Our facilities are equipped to handle the specific requirements of this synthesis, including the precise pH control and phase transfer catalysis needed to achieve the high selectivity for the B-type isomer. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability.
We invite you to collaborate with us to leverage this innovative technology for your wave-absorbing coating projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this new route can optimize your bill of materials. Please contact us to request specific COA data for our pilot batches and to discuss route feasibility assessments for your downstream applications. Let us partner with you to secure a stable, high-quality supply of this strategic material.
