Revolutionizing Vinyl Dithiocarbamate Production: A Catalyst-Free Safe Synthesis Strategy for Global Supply Chains
The chemical landscape for synthesizing vinyl dithiocarbamate compounds has long been dominated by hazardous reagents and complex catalytic systems that pose significant risks to both operational safety and environmental compliance. Patent CN113831269A introduces a transformative approach that fundamentally alters the production paradigm by replacing volatile carbon disulfide with stable alkyl xanthates in a catalyst-free environment. This innovation addresses the critical pain points faced by R&D Directors and Supply Chain Heads who struggle with the volatility and toxicity of traditional sulfur sources. By utilizing electron-withdrawing group-substituted alkynes and alkyl xanthates in a mild solvent system, this method achieves exceptional yields and purity without the need for expensive transition metals or inert gas protection. The strategic shift towards safer, solid-state reagents not only mitigates the risk of explosion during storage and handling but also streamlines the downstream purification process, making it an ideal candidate for large-scale commercial adoption in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
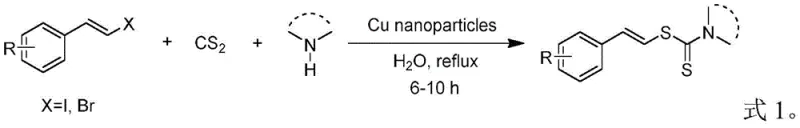
Historically, the synthesis of vinyl dithiocarbamates has relied heavily on methods that introduce substantial operational bottlenecks and safety hazards into the manufacturing workflow. One prevalent conventional technique involves the condensation of amines, carbon disulfide, and styryl halides catalyzed by nano-copper particles in water, as illustrated in the reaction scheme below. This legacy process suffers from severe drawbacks, including the easy oxidation of nano-copper particles which necessitates storage under argon gas, thereby increasing infrastructure costs and complexity. Furthermore, the reaction times are excessively long, often extending beyond six hours, and the yields are frequently compromised by the instability of the catalytic system. The reliance on carbon disulfide, a highly volatile and flammable liquid with a boiling point of merely 46.5°C, creates an inherent safety risk that requires specialized containment and ventilation systems, driving up the capital expenditure for production facilities.
The Novel Approach
In stark contrast to the hazardous and inefficient legacy methods, the novel approach disclosed in the patent utilizes a one-pot reaction strategy that eliminates the need for external catalysts and dangerous gaseous reagents. By employing alkyl xanthates as the sulfur source, the process leverages the stability of solid reagents to ensure consistent reaction kinetics and safer handling protocols. The reaction proceeds smoothly at moderate temperatures ranging from 25°C to 100°C, significantly reducing energy consumption compared to high-temperature reflux conditions required by older methods. This catalyst-free methodology not only simplifies the reaction setup but also ensures that the final product is free from heavy metal contamination, a critical requirement for pharmaceutical intermediates. The operational simplicity allows for a drastic reduction in processing time and waste generation, positioning this technology as a superior alternative for cost reduction in fine chemical manufacturing where safety and purity are paramount.
Mechanistic Insights into Catalyst-Free Nucleophilic Addition
The core of this technological breakthrough lies in the unique mechanistic pathway where alkyl xanthates undergo ester ammonolysis with amines to form substituted dithio ammonium formates in situ. This intermediate then participates in a nucleophilic addition reaction with the electron-deficient alkyne without the need for isolation, creating a seamless telescoped process. The absence of a catalyst is particularly advantageous as it removes the necessity for complex metal scavenging steps, which are often costly and can lead to product loss. The reaction mechanism favors the formation of the vinyl dithiocarbamate structure through a highly selective pathway that minimizes side reactions, ensuring a clean impurity profile. This selectivity is crucial for R&D Directors who need to validate the structural integrity of intermediates for downstream drug synthesis, as it reduces the burden on analytical quality control teams.

Impurity control in this system is inherently robust due to the mild reaction conditions and the specific reactivity of the xanthate anion. Unlike traditional methods that may generate complex by-products from radical pathways or metal-catalyzed side reactions, this process yields a predictable product distribution. The by-products, primarily alcohols like methanol or ethanol, are benign and can be easily recovered or disposed of, contributing to a greener manufacturing footprint. The high purity achieved, often exceeding 97% as demonstrated in specific examples, indicates that the reaction equilibrium strongly favors the product formation. For procurement managers, this high selectivity translates to reduced raw material waste and lower costs associated with purification solvents, making the overall cost of goods sold more competitive in the global market.
How to Synthesize Vinyl Dithiocarbamate Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature to maximize the benefits of the catalyst-free system. The process begins with the preparation of a solvent mixture, which can include water, alcohols, or chlorinated solvents, providing flexibility based on available infrastructure. The reactants are combined at room temperature before heating, which allows for a controlled initiation of the exothermic ammonolysis step. Detailed standard operating procedures regarding the specific molar ratios of alkynes to xanthates and the precise pH adjustment during workup are critical for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent data to ensure consistent high-yield production.
- Mix electron-withdrawing group-substituted alkynes and alkyl xanthates in a solvent containing primary or secondary amines at room temperature.
- Heat the reaction mixture to 25-100°C until completion, then neutralize with dilute hydrochloric acid.
- Extract with ethyl acetate, dry the organic phase, and recrystallize the residue from toluene to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound advantages for supply chain stability and cost management. The replacement of hazardous carbon disulfide with solid alkyl xanthates eliminates the need for specialized hazardous material storage and transport, significantly reducing logistics costs and regulatory compliance burdens. This shift enhances supply chain reliability by sourcing stable, commercially available solids rather than volatile liquids that are subject to strict transportation restrictions. For procurement managers, this means a more resilient supply base with fewer disruptions due to safety incidents or regulatory changes regarding hazardous chemicals. The simplified workup procedure also reduces the demand for extensive purification equipment, allowing for faster turnaround times and increased production capacity without significant capital investment.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a significant cost driver from the bill of materials, as noble or specialized metals often command high market prices. Additionally, the catalyst-free nature of the reaction negates the need for expensive metal scavenging resins or complex filtration systems, leading to substantial cost savings in downstream processing. The ability to recover and reuse solvent systems further contributes to the economic efficiency of the process, lowering the overall operational expenditure. By avoiding the use of inert gas protection required for sensitive catalysts, utilities costs are also minimized, creating a leaner manufacturing cost structure that improves margin potential.
- Enhanced Supply Chain Reliability: The use of stable solid reagents like alkyl xanthates ensures that raw material inventory can be maintained safely for extended periods without degradation, unlike volatile liquids that require constant monitoring. This stability reduces the risk of supply interruptions caused by reagent spoilage or safety shutdowns, ensuring continuous production flow. The simplified reaction conditions allow for manufacturing in a wider range of facilities, diversifying the potential supplier base and reducing dependency on specialized high-tech plants. This flexibility is crucial for maintaining supply continuity in the face of global logistical challenges, ensuring that critical intermediates are available when needed.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal profile and lack of exothermic runaway risks associated with hazardous reagents. This makes the transition from laboratory scale to commercial production smoother and less capital intensive, facilitating rapid market entry. Environmentally, the reduction in hazardous waste and the use of less toxic reagents align with increasingly stringent global environmental regulations, reducing the risk of fines and shutdowns. The generation of benign alcohol by-products simplifies waste treatment protocols, lowering the cost of environmental compliance and enhancing the company's sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The insights provided here aim to clarify the operational benefits and technical capabilities of the new method.
Q: Why is alkyl xanthate preferred over carbon disulfide in this synthesis?
A: Alkyl xanthates are solid, stable, and non-volatile, eliminating the explosion and toxicity risks associated with liquid carbon disulfide while maintaining high reaction activity.
Q: Does this method require expensive transition metal catalysts?
A: No, the patented process operates without any catalyst, which significantly reduces raw material costs and simplifies the removal of metal impurities from the final product.
Q: What is the typical purity achievable with this new route?
A: The method consistently delivers high-purity products, often exceeding 97% HPLC purity, suitable for sensitive pharmaceutical and agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinyl Dithiocarbamate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative synthesis technologies to deliver high-value intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patent are realized at an industrial level. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch meets the exacting standards required by top-tier pharmaceutical and agrochemical companies. We understand the critical nature of supply chain continuity and are equipped to handle the complexities of scaling up catalyst-free processes efficiently.
We invite you to collaborate with us to optimize your supply chain for vinyl dithiocarbamate derivatives. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
