Advanced Manufacturing of Manidipine Hydrochloride via Optimized Hantzsch Cyclization and Crystallization
Advanced Manufacturing of Manidipine Hydrochloride via Optimized Hantzsch Cyclization and Crystallization
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with industrial scalability, particularly for complex calcium channel blockers like Manidipine Hydrochloride. Patent CN102875451A introduces a transformative approach to the synthesis of CV-4093, addressing critical bottlenecks in traditional manufacturing workflows. This innovation focuses on the strategic optimization of post-treatment procedures, effectively eliminating the need for high-vacuum rectification and silica gel column chromatography, which have historically plagued the production of this antihypertensive agent. By re-engineering the purification logic through precise solvent systems and pH-controlled extractions, the new method ensures that the maximum single impurity is rigorously controlled within 0.1%, meeting the stringent standards of the Japanese Pharmacopoeia (JP15). This technical breakthrough not only enhances the chemical integrity of the final active pharmaceutical ingredient but also establishes a foundation for more reliable supply chains and cost-efficient manufacturing processes.
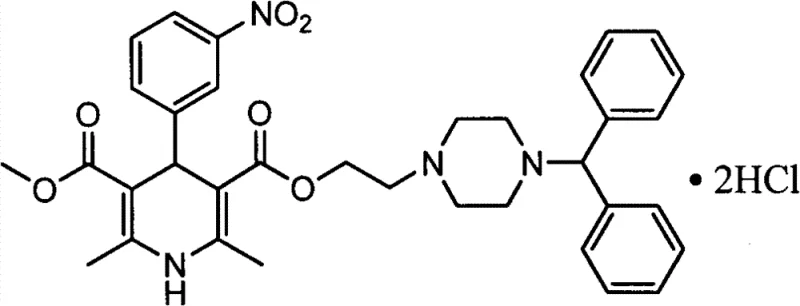
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Manidipine Hydrochloride has been hindered by purification techniques that are inherently difficult to scale and economically inefficient. Traditional routes described in earlier patents often rely heavily on high-vacuum rectification and silica gel column chromatography to isolate the desired dihydropyridine derivative from complex reaction mixtures. These methods present significant operational challenges; column chromatography, while effective for analytical purity, is notoriously labor-intensive, consumes vast quantities of solvents, and results in substantial product loss due to irreversible adsorption on the stationary phase. Furthermore, high-vacuum rectification requires specialized equipment that increases capital expenditure and maintenance costs, making the process less attractive for large-volume commercial production. The reliance on chlorinated solvents like trichloromethane in some legacy methods further complicates environmental compliance and waste management, creating a barrier to sustainable manufacturing.
The Novel Approach
The improved methodology disclosed in the patent data fundamentally shifts the paradigm from separation-based purification to crystallization-driven isolation. Instead of relying on chromatographic columns, this novel approach leverages the differential solubility of the target compound and its impurities in specific mixed solvent systems. The process utilizes a carefully calibrated mixture of ethyl acetate, isopropyl ether, and n-hexane to induce selective crystallization of the Manidipine free base. This is complemented by a sophisticated acid-base extraction protocol where pH values are meticulously adjusted between 1-2 and 9-10 to partition impurities into the aqueous phase while retaining the product in the organic layer. By removing the chromatographic step entirely, the process drastically simplifies the operational workflow, reduces solvent consumption, and significantly shortens the production cycle time, thereby enhancing the overall throughput and economic viability of the synthesis.
Mechanistic Insights into Hantzsch Pyridine Synthesis and Purification
The core of this synthesis lies in the classic Hantzsch dihydropyridine condensation, where the key intermediate, 2-(4-diphenyl-methyl-1-piperazinyl) ethyl acetoacetate, reacts with methyl 3-aminocrotonate and m-nitrobenzaldehyde. In this optimized route, the reaction is conducted in isopropanol under reflux conditions, which provides an ideal thermal environment for the multicomponent condensation to proceed efficiently. The mechanistic advantage here is the stability of the intermediate ester under these conditions, preventing premature hydrolysis or degradation that could lead to complex impurity profiles. The subsequent workup involves distilling off the isopropanol and redissolving the residue in ethyl acetate, setting the stage for the critical purification sequence. The choice of isopropanol as the reaction medium is strategic, as it facilitates the dissolution of all three reactants while allowing for easy removal prior to the extraction phase, ensuring a clean transition to the purification steps.
Impurity control is achieved through a multi-stage physicochemical separation rather than chemical derivatization. After the initial cyclization, the crude mixture is treated with hydrochloric acid to adjust the pH to between 1 and 2, causing the basic Manidipine species to form a water-soluble salt while neutral organic impurities remain in the organic phase or precipitate out. Following filtration, the filter cake is redissolved in ethyl acetate, and the pH is raised to 9-10 using sodium hydroxide, regenerating the free base which partitions back into the organic layer. This acid-base swing effectively scrubs the product of acidic and neutral byproducts. The final crystallization from the ternary solvent system (ethyl acetate:isopropyl ether:n-hexane in a 1:3:6 ratio) exploits the subtle differences in lattice energy and solubility to exclude trace impurities, resulting in a free base with HPLC purity greater than 99% before the final salt formation step even begins.
How to Synthesize Manidipine Hydrochloride Efficiently
The synthesis of Manidipine Hydrochloride via this optimized route involves three distinct operational stages that prioritize yield and purity without the need for complex chromatographic equipment. The process begins with the acylation of the piperazine starting material, followed by the critical Hantzsch cyclization, and concludes with salt formation and recrystallization. Each step is designed to maximize material throughput while minimizing the generation of hazardous waste. The detailed standardized synthesis steps, including specific reagent ratios, temperature controls, and stirring times required to replicate the high-purity results described in the patent, are outlined in the technical guide below.
- Acylation of 1-benzhydryl-4-(2-hydroxyethyl)piperazine with diketene at 70-80°C to form the acetoacetic ester intermediate.
- Hantzsch condensation of the ester intermediate with methyl 3-aminocrotonate and m-nitrobenzaldehyde in isopropanol, followed by pH-controlled extraction.
- Salt formation using hydrochloric acid in methanol, followed by recrystallization to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the elimination of column chromatography and high-vacuum rectification represents a substantial opportunity for cost reduction and risk mitigation. Traditional purification methods are not only expensive due to the cost of silica gel and specialized equipment but also introduce variability in batch-to-batch consistency. By adopting this crystallization-based purification strategy, manufacturers can achieve a more predictable production schedule with significantly reduced downtime between batches. The simplified workflow means fewer unit operations, which directly translates to lower labor costs and reduced energy consumption. Furthermore, the avoidance of chlorinated solvents like trichloromethane aligns with increasingly strict environmental regulations, reducing the liability and cost associated with hazardous waste disposal and solvent recovery.
- Cost Reduction in Manufacturing: The removal of silica gel column chromatography eliminates one of the most expensive consumables in fine chemical synthesis, leading to direct savings in raw material costs. Additionally, the process avoids the significant product loss typically associated with chromatographic separation, thereby improving the overall mass balance and effective yield of the final API. The use of common industrial solvents such as ethyl acetate, isopropanol, and n-hexane further ensures that solvent procurement remains cost-effective and readily available in the global market, avoiding the price volatility of specialized reagents.
- Enhanced Supply Chain Reliability: Simplifying the synthesis route by removing complex purification steps inherently increases the robustness of the supply chain. Fewer processing steps mean fewer points of failure and a reduced likelihood of batch rejection due to out-of-specification impurities. The ability to consistently produce material with single impurities below 0.1% ensures that downstream formulation partners receive high-quality intermediates, reducing the need for costly re-testing or re-processing. This reliability is crucial for maintaining continuous supply agreements with major pharmaceutical clients who demand strict adherence to quality standards.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial suitability, utilizing standard reactor configurations that can be easily scaled from pilot plant to commercial production volumes. The absence of high-vacuum rectification reduces the engineering complexity required for scale-up, allowing for faster technology transfer. From an environmental perspective, the reduction in solvent usage and the elimination of silica gel waste contribute to a greener manufacturing footprint, supporting corporate sustainability goals and facilitating smoother regulatory approvals in environmentally conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Manidipine Hydrochloride using this optimized synthetic route. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on purity standards, yield expectations, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply networks.
Q: How does this process control impurities without column chromatography?
A: The process utilizes a specific mixed solvent crystallization system (ethyl acetate/isopropyl ether/n-hexane) and precise pH adjustments during extraction to selectively precipitate the product while leaving impurities in the mother liquor, achieving single impurity levels below 0.1%.
Q: What are the yield improvements compared to traditional methods?
A: By eliminating material losses associated with silica gel column chromatography and high-vacuum rectification, the overall yield is significantly stabilized, with the final salt formation step achieving yields between 75% and 80%.
Q: Is this method suitable for large-scale GMP production?
A: Yes, the removal of complex purification steps like column chromatography and the use of standard reflux and crystallization techniques make this route highly scalable and compliant with industrial GMP requirements for API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Manidipine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of effective antihypertensive therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Manidipine Hydrochloride meets the highest international standards, including the impurity limits defined in the Japanese Pharmacopoeia.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of this chromatography-free method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a secure and high-quality supply of this vital cardiovascular intermediate.
