Advanced One-Pot Synthesis of N-Acylamino Acid Esters for Commercial Scale Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN1167671C, which discloses a robust process for preparing N-acylamino-acid esters and N-acylamino-acetals. This technology represents a significant departure from classical methodologies, offering a streamlined one-pot reaction that utilizes readily available starting materials such as carboxamides and glyoxal monoacetal derivatives. For R&D directors and procurement specialists alike, the implications of this patent are profound, as it addresses critical pain points regarding raw material toxicity, process complexity, and overall yield optimization. By leveraging a carboxylic acid-catalyzed condensation, this method bypasses the need for hazardous reagents often associated with amino acid precursor synthesis, thereby establishing a new benchmark for sustainable chemical production.
Furthermore, the versatility of this synthetic pathway allows for the precise tuning of product profiles simply by adjusting the molar ratios of the reactants. This flexibility is crucial for manufacturers who need to pivot between producing N-acylamino-acid esters, which are vital for peptide synthesis, and N-acylamino-acetals, which serve as key intermediates for oxazole derivatives. The ability to achieve high purity and excellent yields without the burden of extensive purification steps makes this technology highly attractive for commercial scale-up. As we delve deeper into the technical specifics, it becomes evident that adopting this methodology can provide a reliable pharmaceutical intermediate supplier with a distinct competitive edge in the global market, particularly in the context of cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of alpha-amino acids and their derivatives has been dominated by the Strecker synthesis, a method that, while effective, carries substantial inherent risks and logistical burdens. The traditional Strecker route necessitates the use of formaldehyde and hydrogen cyanide to generate amino-nitriles, which are then hydrolyzed or alcoholized to form the final products. This reliance on hydrogen cyanide introduces severe safety hazards, requiring specialized containment facilities and rigorous emergency response protocols that drive up capital expenditure and operational costs. Moreover, the handling of toxic cyanide waste streams poses significant environmental compliance challenges, often necessitating complex wastewater treatment systems that can bottleneck production throughput. Additionally, the multi-step nature of converting nitriles to esters or acids often results in cumulative yield losses and increased energy consumption due to the isolation and purification of intermediate species.
The Novel Approach
In stark contrast, the novel approach described in the patent data utilizes a direct condensation reaction between a carboxamide and a glyoxal monoacetal derivative in the presence of a carboxylic acid. This methodology elegantly circumvents the need for toxic cyanide sources, replacing them with stable and commercially accessible acetals and amides. The reaction proceeds efficiently under relatively mild thermal conditions, typically ranging from 40-200 °C, and can be conducted under normal pressure or slight vacuum, eliminating the need for high-pressure reactors. A standout feature of this innovation is its adaptability; by simply modulating the stoichiometric ratio of the carboxamide and carboxylic acid relative to the acetal, manufacturers can selectively direct the reaction towards either the ester or the acetal product. This level of control not only simplifies process development but also enhances the economic viability of the operation by minimizing side reactions and maximizing atom economy.
Mechanistic Insights into Carboxylic Acid-Catalyzed Condensation
The core of this technological breakthrough lies in the mechanistic interplay between the glyoxal monoacetal and the carboxamide, facilitated by the carboxylic acid catalyst. The reaction initiates with the activation of the acetal functionality, likely through protonation by the carboxylic acid, which increases the electrophilicity of the carbonyl carbon. This activated species then undergoes nucleophilic attack by the nitrogen atom of the carboxamide, leading to the formation of a transient hemiaminal intermediate. Subsequent elimination of alcohol and rearrangement steps, driven by the thermodynamic stability of the resulting amide bond, yield the final N-acyl derivative. The presence of the carboxylic acid serves a dual purpose: it acts as a catalyst to lower the activation energy of the rate-determining steps and, when used in excess, participates directly in the acylation of the amino group to form the N-acyl moiety. This dual role is critical for achieving the high conversion rates observed in the experimental examples, where yields exceeding 90% are routinely reported.
Crucially, the mechanism also provides a built-in impurity control strategy. By avoiding the formation of reactive nitrile intermediates, the process inherently suppresses the generation of polymeric byproducts and tars that often plague Strecker-type reactions. The selectivity for the desired N-acyl structure is further enhanced by the specific choice of the glyoxal monoacetal derivative, which dictates the stereochemistry and substitution pattern of the final molecule. For instance, when targeting N-formyl-alpha-alanine esters, the use of methyl-glyoxal acetals ensures that the methyl group is correctly positioned at the alpha-carbon, minimizing the risk of regioisomeric impurities. This precision is paramount for pharmaceutical applications where impurity profiles are strictly regulated, ensuring that the resulting high-purity N-acylamino acid esters meet the stringent quality standards required for downstream drug synthesis.
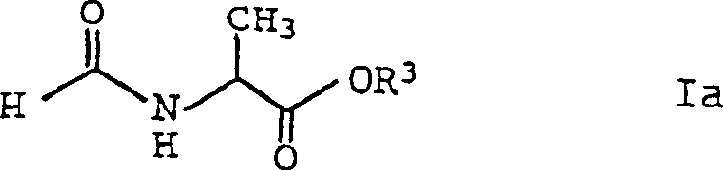
The structural integrity of the N-acylamino-acid ester, as depicted in the formula above, is maintained throughout the reaction due to the mild conditions employed. The ester linkage (COOR3) remains stable under the reaction pH and temperature, preventing hydrolysis which could otherwise lead to the free acid and alcohol byproducts. Similarly, when the reaction conditions are tuned to favor acetal formation, the mechanism shifts to preserve the acetal functionality (CH(OR3)2), resulting in the distinct structure shown below. This ability to toggle between two chemically distinct yet structurally related product classes using the same fundamental reaction platform underscores the versatility of this catalytic system.
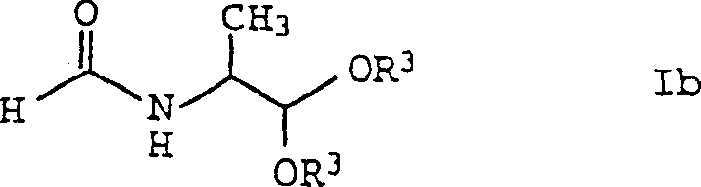
How to Synthesize N-Acylamino Acid Esters Efficiently
Implementing this synthesis route on a production scale requires careful attention to reaction parameters, particularly temperature control and stoichiometric precision. The process is designed to be operationally simple, often proceeding without the need for additional solvents, which aligns with modern green chemistry principles. Operators must ensure that the mixing of the glyoxal monoacetal, carboxamide, and carboxylic acid is thorough to prevent localized hot spots that could degrade the sensitive acetal groups. Detailed standard operating procedures regarding the addition rates and heating ramps are essential to replicate the high yields demonstrated in the patent examples. For a comprehensive guide on the exact step-by-step execution of this protocol, please refer to the standardized synthesis instructions provided below.
- Mix glyoxal monoacetal derivative with carboxamide and carboxylic acid in a reactor, ensuring precise stoichiometric control based on the desired product (ester vs acetal).
- Heat the mixture to a temperature range of 40-200 °C, preferably 60-150 °C, under normal pressure or slight vacuum (200-1000 mbar) to initiate the condensation reaction.
- Upon completion, cool the reaction mixture, wash with dilute sodium carbonate solution to remove acidic byproducts, and purify the final liquid product via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this N-acylamino-acid ester preparation process offers transformative benefits for procurement managers and supply chain leaders. The primary advantage stems from the drastic simplification of the raw material portfolio; by eliminating the need for hazardous cyanide salts and formaldehyde, companies can reduce their regulatory burden and insurance costs significantly. Furthermore, the ability to run the reaction solvent-free or with minimal solvent usage translates directly into reduced waste disposal costs and lower energy consumption for solvent recovery. This efficiency gain is not merely theoretical; the patent data demonstrates that high yields are achievable with straightforward workup procedures involving simple washing and distillation, which shortens the overall cycle time per batch. Consequently, this leads to a substantial increase in plant throughput and asset utilization, allowing manufacturers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the replacement of expensive and dangerous reagents with commodity chemicals like formamide and simple carboxylic acids. By removing the requirement for specialized cyanide handling infrastructure, capital investment for new production lines is significantly lowered, while existing facilities can be retrofitted with minimal disruption. The high atom economy of the reaction ensures that a greater proportion of the input mass is converted into valuable product, thereby reducing the cost of goods sold (COGS). Additionally, the elimination of complex purification steps reduces the consumption of auxiliary materials such as filtration media and chromatography resins, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for any supply chain head, and this process excels by relying on widely available bulk chemicals. Glyoxal acetals and carboxamides are produced on a massive scale for various industries, ensuring a robust and resilient supply base that is less susceptible to geopolitical disruptions or single-source bottlenecks. The simplified logistics of handling non-hazardous materials also streamline transportation and storage, reducing lead times and minimizing the risk of shipment delays due to regulatory compliance issues. This reliability ensures continuous production schedules, safeguarding against stockouts that could jeopardize downstream pharmaceutical manufacturing commitments.
- Scalability and Environmental Compliance: The inherent safety and simplicity of this one-pot reaction make it ideally suited for commercial scale-up of complex pharmaceutical intermediates. The absence of exothermic runaway risks associated with cyanide chemistry allows for larger batch sizes and safer reactor operations. From an environmental standpoint, the process generates significantly less hazardous waste, facilitating easier compliance with increasingly stringent global environmental regulations. The potential for solvent-free operation further enhances the green credentials of the manufacturing process, appealing to eco-conscious partners and helping companies meet their sustainability goals without compromising on productivity or product quality.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology, we have compiled a set of frequently asked questions based on the specific technical disclosures within the patent documentation. These queries address common concerns regarding reaction selectivity, safety protocols, and product applicability. Understanding these nuances is essential for making informed decisions about integrating this process into your existing manufacturing portfolio. The answers provided below are derived directly from the experimental data and claims of the patent, ensuring accuracy and relevance to real-world production scenarios.
Q: What are the primary advantages of this synthesis method over traditional Strecker synthesis?
A: Unlike Strecker synthesis which relies on hazardous hydrogen cyanide and formaldehyde, this process utilizes stable glyoxal monoacetals and carboxamides, significantly improving operational safety and reducing the need for complex waste treatment infrastructure.
Q: How does the stoichiometry affect the product distribution between esters and acetals?
A: The process offers precise control: using a large excess of carboxamide and carboxylic acid (250-800 mol%) favors the formation of N-acylamino-acid esters, while lower amounts (50-250 mol%) selectively produce N-acylamino-acetals.
Q: Is solvent required for this reaction on an industrial scale?
A: No, the patent explicitly highlights that the reaction can be performed solvent-free, which drastically simplifies downstream processing, reduces solvent recovery costs, and enhances the overall environmental profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acylamino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN1167671C for maintaining a competitive edge in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity N-acylamino acid esters that meet the most rigorous quality specifications, supported by our state-of-the-art rigorous QC labs and analytical capabilities. Our expertise allows us to optimize reaction parameters for maximum yield and minimal impurity formation, guaranteeing a consistent supply of critical intermediates for your drug development pipelines.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and target specifications. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our optimized processes can enhance your supply chain resilience and drive down your overall manufacturing costs. Let us be your trusted partner in navigating the complexities of modern pharmaceutical intermediate synthesis.
