Advanced Synthesis of N-Phenylalkyl Alpha-Aminocarboxamide Derivatives for CNS Therapeutics
Advanced Synthesis of N-Phenylalkyl Alpha-Aminocarboxamide Derivatives for CNS Therapeutics
The pharmaceutical landscape continuously demands innovative small molecules capable of addressing complex central nervous system (CNS) disorders with high efficacy and safety profiles. Patent CN1027588C introduces a significant advancement in this domain by disclosing a robust process for the preparation of N-phenylalkyl substituted alpha-aminocarboxamide derivatives. These compounds are not merely theoretical constructs but represent a class of potent therapeutic agents demonstrated to be effective as antiepileptic, antiparkinsonian, neuroprotective, antidepressant, antispasmodic, and hypnotic agents in mammalian models. The core innovation lies in the versatile synthetic methodology that allows for the precise modulation of substituents on the phenylalkyl backbone, thereby optimizing pharmacokinetic properties and biological activity. This technical disclosure provides a foundational blueprint for manufacturing high-purity intermediates that can be further developed into final drug substances, offering a reliable pathway for pharmaceutical companies seeking to expand their CNS pipeline with novel chemical entities.
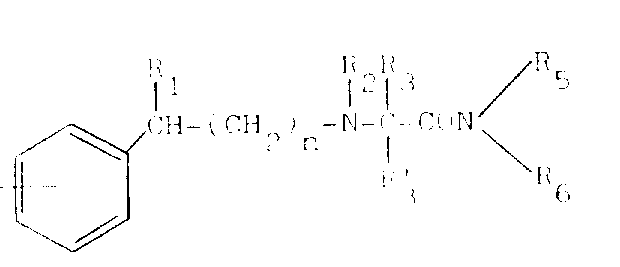
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aminocarboxamide derivatives often relied on multi-step sequences involving harsh reaction conditions that could compromise the integrity of sensitive functional groups. Traditional approaches frequently necessitated the use of strong acids or bases for amide bond formation or alkylation, which could lead to racemization of chiral centers or decomposition of thermally labile substituents such as halogens or alkoxy groups. Furthermore, older methodologies described in prior art, such as those referenced in British Patent No. 1140748, often resulted in crude products requiring extensive and costly purification procedures to remove toxic heavy metal catalysts or persistent organic impurities. These inefficiencies not only inflated the cost of goods sold but also introduced significant variability in batch-to-batch consistency, posing a challenge for regulatory compliance and supply chain reliability in the manufacture of high-purity pharmaceutical intermediates.
The Novel Approach
In contrast, the methodology outlined in CN1027588C employs a sophisticated reductive amination strategy that operates under remarkably mild conditions, typically ranging from 0°C to 40°C in solvents like methanol or acetonitrile. This approach utilizes sodium cyanoborohydride as a selective reducing agent, which effectively converts imine intermediates into stable amine bonds without affecting other reducible functionalities present in the molecule. The process is characterized by its operational simplicity, often requiring only stirring at room temperature followed by straightforward workup procedures such as filtration and solvent evaporation. By eliminating the need for extreme temperatures or pressures, this novel route significantly reduces energy consumption and enhances the overall safety profile of the manufacturing process, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing while maintaining exceptional product quality.
Mechanistic Insights into Reductive Amination and Alkylation Pathways
The core chemical transformation driving the synthesis of these bioactive molecules involves the nucleophilic attack of an amino group on a carbonyl carbon, followed by dehydration to form an imine or iminium ion, which is subsequently reduced in situ. In the preferred embodiment, an aminoamide hydrochloride salt is suspended in dry methanol under an inert nitrogen atmosphere to prevent oxidation, after which sodium cyanoborohydride is added to facilitate the hydride transfer. The presence of molecular sieves can further accelerate the reaction by sequestering water produced during imine formation, thus driving the equilibrium towards the desired product. This mechanism ensures high chemoselectivity, allowing for the incorporation of diverse substituents such as chloro, fluoro, or trifluoromethyl groups on the aromatic rings without side reactions, which is critical for tuning the lipophilicity and blood-brain barrier penetration of the final CNS active pharmaceutical ingredient.
Additionally, the patent describes alternative alkylation pathways where secondary amines react with alkyl halides or aldehydes to introduce N-methyl or other alkyl groups, expanding the structural diversity of the library. For instance, the reaction of a secondary amine intermediate with methyl iodide in the presence of anhydrous potassium carbonate allows for the precise installation of N-methyl groups, which can significantly alter the metabolic stability and receptor binding affinity of the compound. Understanding these mechanistic nuances is vital for process chemists aiming to control impurity profiles, as over-alkylation or incomplete reduction can lead to difficult-to-remove byproducts. The ability to switch between reductive amination and direct alkylation provides a flexible toolkit for optimizing the synthesis of specific analogues, ensuring that the manufacturing process can be tailored to meet the stringent purity specifications required for clinical-grade materials.
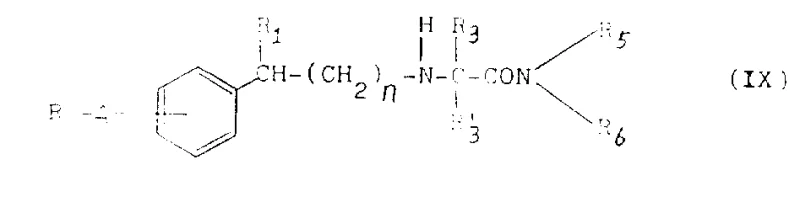
How to Synthesize N-Phenylalkyl Alpha-Aminocarboxamide Efficiently
The synthesis protocol detailed in the patent offers a reproducible framework for producing these valuable intermediates, beginning with the careful selection of high-quality starting materials such as substituted benzaldehydes and aminoamides. The process emphasizes the importance of maintaining anhydrous conditions and controlling the stoichiometry of the reducing agent to maximize yield and minimize waste. Operators are advised to monitor the reaction progress via thin-layer chromatography or HPLC to determine the optimal endpoint before proceeding to the isolation phase, which typically involves acid-base extraction to separate the free base from inorganic salts. The detailed standardized synthesis steps see below guide provides a comprehensive walkthrough for implementing this chemistry in a pilot or production setting.
- Prepare the reaction mixture by suspending the appropriate aminoamide hydrochloride salt in dry methanol under a nitrogen atmosphere.
- Add sodium cyanoborohydride as the reducing agent and introduce the substituted benzaldehyde derivative to initiate reductive amination.
- Stir the mixture at room temperature for 8 to 18 hours, followed by filtration, solvent evaporation, and purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers substantial benefits regarding cost efficiency and supply chain resilience. The reliance on commodity chemicals such as methanol, sodium cyanoborohydride, and readily available substituted benzaldehydes means that raw material sourcing is not constrained by geopolitical bottlenecks or single-supplier dependencies. This accessibility translates directly into enhanced supply chain reliability, as manufacturers can secure multiple sources for key inputs, thereby mitigating the risk of production stoppages due to material shortages. Furthermore, the mild reaction conditions reduce the burden on facility infrastructure, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, which significantly lowers capital expenditure requirements for scale-up.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of energy-intensive heating or cooling cycles result in a drastically simplified production workflow that lowers operational expenditures. By streamlining the purification process through efficient crystallization or chromatography techniques, the overall yield is optimized, reducing the amount of raw material required per kilogram of final product. This efficiency gain allows for competitive pricing structures without compromising on the quality or purity of the pharmaceutical intermediate, providing a clear economic advantage for partners looking to optimize their cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of the chemical process ensures consistent batch-to-batch quality, which is essential for maintaining regulatory filings and avoiding costly delays in drug development timelines. The use of stable intermediates and straightforward reaction protocols minimizes the likelihood of batch failures, ensuring a steady flow of materials to downstream formulation teams. This predictability is crucial for long-term supply agreements, enabling procurement managers to forecast inventory needs accurately and maintain optimal stock levels to support continuous commercial manufacturing operations.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods, as the primary byproducts are inorganic salts that can be easily treated or disposed of according to standard environmental protocols. The scalability of the reductive amination reaction has been demonstrated from gram to multi-kilogram scales without loss of efficiency, indicating a smooth path from laboratory discovery to commercial production. This environmental compatibility aligns with modern green chemistry principles, reducing the ecological footprint of the manufacturing process and ensuring compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these N-phenylalkyl substituted alpha-aminocarboxamide derivatives. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of this technology. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing development pipelines.
Q: What are the primary therapeutic applications of these derivatives?
A: These compounds exhibit potent activity against the central nervous system, serving as effective antiepileptic, antiparkinsonian, neuroprotective, and antidepressant agents.
Q: How does the reductive amination method improve process safety?
A: By utilizing mild reaction temperatures between 0-40°C and avoiding harsh acidic or basic conditions, the process minimizes thermal hazards and degradation risks.
Q: Are the starting materials commercially scalable?
A: Yes, the synthesis relies on readily available substituted benzaldehydes and aminoamides, ensuring a robust and continuous supply chain for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phenylalkyl Alpha-Aminocarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex patent chemistry into commercial reality with precision and speed. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest industry standards for identity, potency, and impurity control. Our commitment to quality assurance means that you can rely on us to deliver materials that are fully compliant with global regulatory requirements, minimizing the risk of delays in your drug approval process.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthetic route to your specific volume and timeline needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall development costs. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to evaluate the technical merits of our manufacturing capabilities firsthand. Let us be your trusted ally in bringing these promising CNS therapeutics from the laboratory bench to the patients who need them most.
