Scalable Synthesis of Optically Pure 4-Aryl-2-Hydroxybutyric Acid via Novel Ruthenium Catalysis
The pharmaceutical industry continuously seeks robust pathways for synthesizing chiral building blocks, particularly for antihypertensive agents. Patent CN101941900A introduces a groundbreaking preparation method for optically pure 4-aryl-2-hydroxy-butyric acid, a critical intermediate in the manufacture of ACE inhibitors. This technology leverages a sophisticated asymmetric catalytic hydrogenation strategy, utilizing ruthenium complexes coordinated with biaryl axial chiral phosphine ligands. By shifting the synthetic focus from unstable liquid esters to stable solid acid substrates, this innovation addresses long-standing challenges in purity and process efficiency. The method demonstrates exceptional versatility across various aryl substitutions, providing a reliable foundation for the production of high-purity pharmaceutical intermediates. For global procurement teams, this represents a significant opportunity to secure a more stable and cost-effective supply chain for essential cardiovascular medication precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-4-phenyl-2-hydroxybutyric acid has been plagued by inefficiencies inherent in traditional resolution and reduction techniques. Conventional approaches often relied on the kinetic resolution of racemic mixtures, a process that theoretically caps the maximum yield at 50%, resulting in substantial waste of valuable starting materials. Alternative methods involved the asymmetric reduction of ketone esters, which required the substrate to be in a liquid state. These liquid precursors, such as 4-phenyl-2-oxobutyrate esters, are notoriously unstable and possess high boiling points, making their purification via distillation energy-intensive and technically difficult. Furthermore, these legacy routes frequently necessitated a multi-step sequence involving initial esterification, followed by asymmetric hydrogenation, and finally hydrolysis to retrieve the free acid. This convoluted pathway not only increased operational costs but also introduced multiple opportunities for impurity generation and yield loss, complicating the supply chain for reliable pharmaceutical intermediate suppliers.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by employing 4-aryl-2-oxo-3-butenoic acid as the direct substrate. Unlike its ester counterparts, this substrate exists as a stable solid at ambient temperatures, drastically simplifying storage, handling, and purification protocols. The novel approach bypasses the need for esterification and subsequent hydrolysis, enabling a direct transformation to the target chiral acid. By utilizing a ruthenium catalyst system augmented with acidic additives, the reaction achieves high enantioselectivity directly on the acid functionality. This streamlining of the synthetic route eliminates several unit operations, thereby reducing the overall processing time and equipment requirements. The ability to produce the target molecule with high optical purity through a simple recrystallization step further underscores the superiority of this direct hydrogenation strategy over complex multi-step alternatives.
Mechanistic Insights into Ru-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise coordination chemistry of the ruthenium catalyst system. The active species is generated from ruthenium precursors complexed with bulky, electron-rich biaryl axial chiral phosphine ligands, such as SunPhos or SegPhos. These ligands create a highly defined chiral environment around the metal center, which is crucial for discriminating between the prochiral faces of the olefinic substrate during the hydrogenation event. The presence of acidic additives, specifically hydrogen halides like HBr, plays a pivotal role in activating the catalyst and stabilizing the transition state. This synergistic interaction between the metal complex and the acid additive ensures that the hydrogen addition occurs with high regio- and stereoselectivity. The mechanism avoids the formation of racemic by-products that typically plague non-catalytic reduction methods, ensuring that the crude product already possesses a high enantiomeric excess before any purification steps are undertaken.
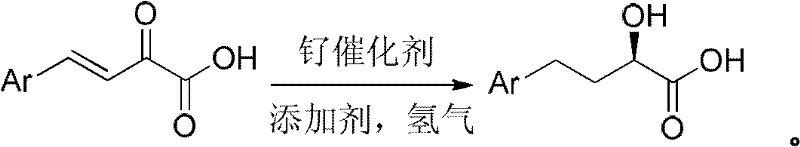
Following the catalytic cycle, the control of impurities is managed through the physical properties of the product rather than complex chromatographic separations. The patent highlights that while the crude reaction mixture may exhibit an ee value around 90%, the specific crystallization behavior of the (R)-enantiomer allows for significant enrichment. By selecting appropriate solvent systems, such as ether or dichloroethane mixtures, the desired enantiomer preferentially crystallizes out of the solution. This recrystallization process effectively rejects minor impurities and the opposite enantiomer, pushing the optical purity to greater than 99% ee. This mechanism of purification is particularly advantageous for cost reduction in API manufacturing, as it replaces expensive chiral HPLC or extensive column chromatography with a scalable, low-energy crystallization step. The robustness of this purification strategy ensures that the final product meets the stringent purity specifications required for regulatory submission and commercial drug production.
How to Synthesize (R)-4-Aryl-2-Hydroxybutyric Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating laboratory success into industrial reality. The process begins with the careful preparation of the catalyst system, ensuring that the ruthenium precursor and chiral ligand are thoroughly mixed under inert conditions to prevent oxidation. The reaction is conducted in common organic solvents like tetrahydrofuran or alcohols, which are readily available and easy to recover. The detailed standardized synthesis steps below outline the precise molar ratios, temperature controls, and pressure settings required to maximize turnover numbers and ensure consistent batch-to-batch quality. This level of procedural detail is essential for process chemists aiming to implement this technology for the commercial scale-up of complex pharmaceutical intermediates.
- Prepare the reaction system by combining 4-aryl-2-oxo-3-butenoic acid substrate with a ruthenium complex catalyst containing biaryl axial chiral phosphine ligands in an organic ether or alcohol solvent.
- Introduce acidic additives such as hydrogen bromide to the mixture and seal the autoclave, then pressurize with high-purity hydrogen gas to initiate the asymmetric catalytic hydrogenation.
- After the reaction completes, isolate the crude product and perform a simple recrystallization step to elevate the enantiomeric excess (ee) value to greater than 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical elegance. The shift from liquid, unstable esters to solid, stable acid substrates fundamentally alters the risk profile of the supply chain. Solid raw materials are less prone to degradation during transport and storage, reducing the incidence of rejected shipments and ensuring a more reliable flow of goods. Furthermore, the elimination of esterification and hydrolysis steps translates directly into a simplified manufacturing workflow. Fewer processing steps mean reduced consumption of auxiliary chemicals, lower energy usage for heating and cooling, and decreased waste generation. These factors collectively contribute to substantial cost savings and a smaller environmental footprint, aligning with modern green chemistry initiatives and corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the truncation of the synthetic sequence. By removing the need for separate esterification and hydrolysis stages, manufacturers can significantly reduce the consumption of reagents such as alcohols, coupling agents, and bases. Additionally, the high turnover number of the ruthenium catalyst means that less precious metal is required per kilogram of product, lowering the raw material cost burden. The ability to achieve high purity through simple recrystallization rather than expensive chromatographic techniques further drives down the cost of goods sold, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of 4-aryl-2-oxo-3-butenoic acid, a substrate that is easier to synthesize and purify than its predecessors. Its solid state facilitates easier handling in bulk quantities, minimizing the risks associated with pumping and transferring viscous or volatile liquids. This stability ensures that production schedules are less likely to be disrupted by raw material quality issues. Moreover, the robustness of the catalytic system allows for consistent performance even at larger scales, reducing the variability that often plagues the transition from pilot plant to full commercial production.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers distinct advantages. The reaction conditions, while requiring hydrogen pressure, operate within standard industrial autoclave capabilities. The solvents used are common and can be efficiently recycled, minimizing volatile organic compound (VOC) emissions. The high atom economy of the hydrogenation reaction, combined with the avoidance of stoichiometric reducing agents like borohydrides, results in a cleaner waste stream. This simplifies wastewater treatment and helps facilities maintain compliance with increasingly stringent environmental regulations, securing the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of the process. Understanding these details is crucial for technical teams evaluating the potential for technology transfer and for procurement specialists assessing the long-term value proposition of this synthetic route.
Q: What are the key advantages of using 4-aryl-2-oxo-3-butenoic acid over traditional ester substrates?
A: Unlike traditional liquid ester substrates which are unstable and difficult to purify, 4-aryl-2-oxo-3-butenoic acid is a stable solid at room temperature. This physical state significantly simplifies purification processes, ensures consistent raw material quality, and eliminates the need for additional esterification and hydrolysis steps found in older methodologies.
Q: How does the new catalytic system achieve high optical purity?
A: The process utilizes a specialized ruthenium complex with biaryl axial chiral phosphine ligands, such as SunPhos, alongside acidic additives. This combination facilitates highly enantioselective hydrogenation. Furthermore, the resulting crude product can be easily upgraded to greater than 99% ee through a straightforward recrystallization procedure, ensuring pharmaceutical-grade purity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial suitability. It operates under manageable temperatures and pressures, utilizes stable solid substrates that are easy to handle in bulk, and achieves high turnover numbers (TON) for the catalyst. The elimination of complex protection and deprotection steps further enhances its viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aryl-2-Hydroxybutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of life-saving medications. Our technical team has extensively analyzed the potential of the ruthenium-catalyzed route described in CN101941900A and is fully prepared to leverage this chemistry for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, including the verification of enantiomeric excess values exceeding 99% as required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for antihypertensive drug intermediates. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient catalytic process can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to bring safer, more affordable cardiovascular therapies to the global market through superior chemical manufacturing.
