Advanced Ruthenium-Catalyzed Synthesis of Optically Pure 4-Aryl-2-Hydroxy-Butyric Acid for Commercial Scale-Up
Advanced Ruthenium-Catalyzed Synthesis of Optically Pure 4-Aryl-2-Hydroxy-Butyric Acid for Commercial Scale-Up
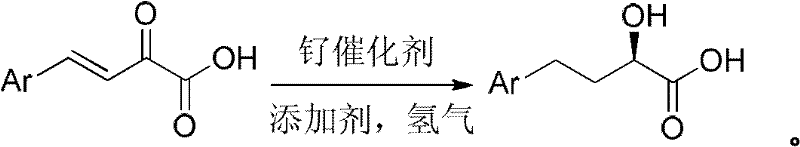
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for synthesizing chiral intermediates essential for life-saving medications. A pivotal advancement in this domain is detailed in patent CN101941900A, which discloses a highly efficient preparation method for optically pure 4-aryl-2-hydroxy-butyric acid. This compound serves as a critical building block for the synthesis of Puli-class antihypertensive drugs, which are angiotensin-converting enzyme (ACE) inhibitors representing a substantial portion of the global hypertension medication market. The disclosed technology leverages a sophisticated ruthenium complex featuring biaryl axial chiral phosphine ligands, such as SunPhos or BINAP, to catalyze the asymmetric hydrogenation of 4-aryl-2-oxo-3-butenoic acid. By operating in organic ether or alcohol solvents with the assistance of acidic additives, this novel route achieves exceptional stereocontrol. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate supplier options, this patent represents a paradigm shift from laborious, multi-step classical resolutions to a streamlined, direct catalytic process that promises superior purity profiles and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure 4-phenyl-2-hydroxy-butyric acid has been plagued by significant technical and economic inefficiencies inherent to traditional synthetic strategies. Prior art methods often relied on the reduction of corresponding ketones followed by the resolution of racemic esters or carboxylic acids, a process that inherently wastes at least fifty percent of the raw material due to the discard of the unwanted enantiomer. Alternative approaches involving enzymatic kinetic resolution or salification with chiral amines introduced further complexity, requiring extensive downstream processing to separate the resolving agents. Furthermore, conventional asymmetric reduction methods frequently utilized 4-phenyl-2-oxo-butyric acid or its ethyl ester as the starting substrate. These precursors present severe handling challenges; the free acid is a high-boiling liquid that is thermally unstable and notoriously difficult to purify to the high standards required for asymmetric catalysis. Impurities in these unstable liquid starting materials drastically reduce the turnover number of expensive catalysts and compromise the optical purity of the final product, creating a bottleneck for cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these legacy methodologies, the innovative approach described in the patent data utilizes 4-aryl-2-oxo-3-butenoic acid as the primary reaction substrate, offering a transformative solution to the stability and purity issues of the past. This unsaturated keto-acid exists as a stable solid at normal temperatures, which fundamentally simplifies logistics, storage, and quality control procedures compared to its unstable liquid predecessors. The new route eliminates the need for preliminary esterification steps that were previously mandatory to protect the carboxylic acid functionality during hydrogenation. By directly subjecting the acid to asymmetric catalytic hydrogenation in the presence of a specialized ruthenium catalyst and acidic additives, the process achieves high enantioselectivity in a single synthetic operation. This consolidation of steps not only drastically simplifies the workflow but also enhances the overall atom economy and yield, providing a compelling value proposition for supply chain heads focused on reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Ru-SunPhos Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the precise orchestration of the catalytic cycle mediated by the ruthenium-chiral phosphine complex. The catalyst system, preferably comprising [RuCl(benzene)(R)-SunPhos]Cl, operates through a mechanism where the chiral environment of the biaryl ligand dictates the facial selectivity of the hydrogen addition. The reaction involves the simultaneous or sequential reduction of the carbon-carbon double bond and the prochiral ketone group. The presence of acidic additives, such as hydrobromic acid, plays a crucial role in activating the catalyst species and potentially protonating intermediate alkoxides to facilitate product release and catalyst regeneration. This synergistic interaction between the metal center, the chiral ligand, and the acid additive ensures that the hydrogenation proceeds with high fidelity, minimizing the formation of undesired diastereomers or over-reduced byproducts. The robustness of this catalytic system allows it to tolerate a variety of aryl substituents, including halogens and alkoxy groups, demonstrating broad substrate scope which is vital for versatile commercial applications.
Controlling the impurity profile is paramount for any process intended for pharmaceutical grade production, and this mechanism offers inherent advantages in impurity suppression. Because the starting material is a stable solid that can be rigorously purified via crystallization prior to the reaction, the input stream into the reactor is of consistently high quality, unlike the variable quality of liquid ketone precursors. The high turnover frequency of the ruthenium catalyst means that lower catalyst loadings can be employed, which subsequently reduces the burden of heavy metal removal in the downstream purification stages. Furthermore, the reaction conditions, typically maintained between 70°C and 90°C, are mild enough to prevent thermal degradation of the sensitive hydroxy-acid product while being energetic enough to drive the reaction to completion. The resulting crude product possesses an enantiomeric excess that is already high, and a simple recrystallization step is sufficient to upgrade the optical purity to greater than 99% ee, effectively removing trace impurities and ensuring the final material meets the stringent specifications required for regulatory submission.
How to Synthesize (R)-4-Aryl-2-Hydroxy-Butyric Acid Efficiently
The implementation of this synthesis route in a production environment requires careful attention to the preparation of the catalyst and the control of reaction parameters to maximize efficiency. The process begins with the in-situ or ex-situ formation of the active ruthenium complex, followed by the charging of the stable solid substrate into a pressurized reactor system. Solvent selection is flexible, allowing for the use of common industrial solvents like tetrahydrofuran, dioxane, or ethanol, which facilitates easy solvent recovery and recycling. The reaction is conducted under a hydrogen atmosphere, with pressures optimized to balance reaction rate and safety. Detailed standard operating procedures regarding the specific molar ratios of substrate to catalyst, which can range widely from 100:1 to 50000:1 depending on the desired throughput, are critical for optimizing cost performance. For a comprehensive understanding of the exact experimental conditions and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction vessel by charging 4-aryl-2-oxo-3-butenoic acid substrate into an autoclave with an organic ether or alcohol solvent such as tetrahydrofuran or ethanol.
- Add the chiral ruthenium catalyst complex, specifically [RuCl(benzene)(R)-SunPhos]Cl, along with an acidic additive like hydrobromic acid to facilitate the activation of the catalytic cycle.
- Pressurize the system with high-purity hydrogen gas and maintain the reaction temperature between 70°C and 90°C, followed by purification via recrystallization to achieve >99% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend far beyond simple chemical conversion. The shift from unstable liquid starting materials to stable solid substrates fundamentally de-risks the supply chain by enabling long-term storage and bulk purchasing without the fear of material degradation. This stability ensures a consistent supply of high-quality raw materials, which is essential for maintaining uninterrupted production schedules for critical antihypertensive medications. Moreover, the elimination of multiple protection and deprotection steps, such as esterification and hydrolysis, significantly reduces the consumption of auxiliary chemicals and solvents. This reduction in material intensity directly correlates to a lower environmental footprint and decreased waste disposal costs, aligning with modern green chemistry initiatives and environmental compliance standards that are increasingly important for global pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route offers profound opportunities for cost optimization throughout the manufacturing value chain. By bypassing the need for separate esterification and hydrolysis units, the process reduces capital expenditure on equipment and lowers operational expenses related to energy consumption and labor. The ability to use the carboxylic acid form directly avoids the cost of alcohol reagents and the subsequent separation of water byproducts. Additionally, the high catalytic efficiency allows for the use of precious metal catalysts at very low loadings, minimizing the cost associated with ruthenium and chiral ligands. These cumulative efficiencies result in a significantly reduced cost of goods sold, making the final API intermediate more competitive in the global marketplace without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical process and the availability of the starting materials. The substrates used in this method are readily accessible and can be synthesized via simple, low-cost pathways, reducing dependency on niche suppliers. The solid state of the key intermediate simplifies logistics, allowing for safer and more economical transportation compared to hazardous or unstable liquids. This reliability ensures that downstream API manufacturers can rely on a steady flow of intermediates, mitigating the risk of production stoppages due to raw material shortages. The process's tolerance to various aryl substitutions also means that a single production line can potentially be adapted to produce a family of related intermediates, adding flexibility to the supply network.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the use of standard unit operations such as hydrogenation autoclaves and crystallization tanks. The reaction conditions are well within the safe operating limits of standard industrial equipment, reducing the need for specialized high-pressure or cryogenic infrastructure. From an environmental perspective, the reduction in solvent usage and the elimination of stoichiometric chiral resolving agents minimize the generation of hazardous waste streams. The high atom economy of the hydrogenation reaction ensures that most of the mass of the starting materials ends up in the final product, adhering to the principles of sustainable manufacturing. This alignment with environmental regulations simplifies the permitting process and enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of the route. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or commercial partnership. The answers reflect the consensus of the technical data regarding catalyst performance, substrate scope, and purification outcomes.
Q: What are the primary advantages of using 4-aryl-2-oxo-3-butenoic acid over traditional ketone substrates?
A: Unlike traditional 4-phenyl-2-oxo-butyric acid which is a high-boiling, unstable liquid difficult to purify, the 4-aryl-2-oxo-3-butenoic acid substrate is a stable solid at room temperature. This physical state significantly simplifies handling, storage, and purification processes, leading to higher reproducibility and lower raw material costs in industrial settings.
Q: How does the new ruthenium-catalyzed method improve optical purity compared to prior art?
A: The patented method utilizes a specific biaryl axial chiral phosphine ligand, such as SunPhos, coordinated with ruthenium. This system, combined with acidic additives, enables direct asymmetric hydrogenation that yields crude products with high enantiomeric excess. Subsequent simple recrystallization further elevates the ee value to greater than 99%, eliminating the need for complex resolution steps required by older enzymatic or chemical reduction methods.
Q: Is this synthesis route suitable for large-scale manufacturing of ACE inhibitor intermediates?
A: Yes, the process is explicitly designed for industrial suitability. By avoiding the multi-step esterification and hydrolysis sequences found in conventional routes, the method reduces the overall number of unit operations. The use of robust heterogeneous-compatible conditions and stable solid starting materials ensures that the process can be safely scaled from laboratory benchtop to multi-ton commercial production without compromising safety or quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aryl-2-Hydroxy-Butyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of life-saving cardiovascular therapies. Our team of expert chemists has thoroughly analyzed the potential of the ruthenium-catalyzed asymmetric hydrogenation route described in CN101941900A and is fully prepared to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical enantiomeric excess values required for chiral drug substances. We are committed to delivering materials that not only meet but exceed the expectations of our global partners.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our pilot batches and to discuss detailed route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of this essential chiral building block for your next-generation antihypertensive formulations.
