Advanced Carbonylation Strategy for Scalable Production of Fluorinated Pyridine Intermediates
Advanced Carbonylation Strategy for Scalable Production of Fluorinated Pyridine Intermediates
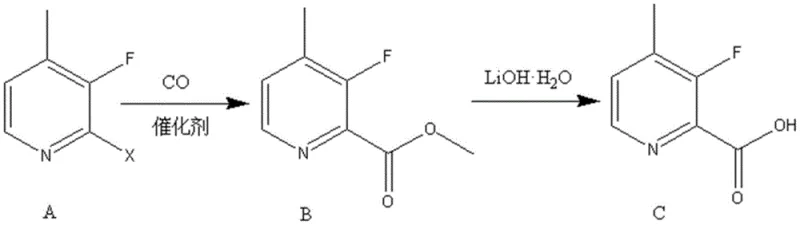
The rapid evolution of fluorinated heterocycles in modern medicinal chemistry has necessitated the development of more efficient synthetic pathways for key building blocks. Patent CN110642781A introduces a groundbreaking methodology for the preparation of 3-fluoro-4-methylpyridine-2-carboxylic acid, a critical scaffold in the design of novel bioactive agents. This technology leverages a streamlined two-step sequence involving palladium-catalyzed carbonylation followed by mild hydrolysis, effectively bypassing the multi-step complexities often associated with functionalizing electron-deficient pyridine rings. By integrating carbon monoxide insertion directly onto the halogenated precursor, the process achieves exceptional atom economy and operational simplicity. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process intensification, offering a route that is not only chemically elegant but also inherently scalable for industrial manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of substituted pyridine carboxylic acids has been plagued by harsh reaction conditions and poor regioselectivity, often requiring cryogenic temperatures or highly reactive organometallic reagents that are difficult to handle on a large scale. Conventional routes frequently involve lithiation strategies which demand stringent moisture-free environments and pose significant safety risks due to the pyrophoric nature of the reagents involved. Furthermore, existing methods often suffer from low yields due to side reactions such as nucleophilic aromatic substitution or decomposition of the sensitive fluorine substituent under aggressive basic conditions. The purification of intermediates in these legacy processes is typically cumbersome, requiring extensive chromatographic separation which drastically increases production costs and waste generation. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and inconsistent quality batches that fail to meet the rigorous specifications required for GMP-grade active pharmaceutical ingredient manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct carbonylative coupling strategy that transforms readily available 2-halo-3-fluoro-4-methylpyridines into the target ester with remarkable efficiency. This approach eliminates the need for unstable organolithium species, replacing them with a robust catalytic system that operates under moderate thermal conditions. The use of carbon monoxide as a C1 building block allows for the direct installation of the carboxyl functionality without altering the existing substitution pattern on the pyridine ring, thereby preserving the integrity of the fluorine atom. Subsequent hydrolysis is performed under mild alkaline conditions using lithium hydroxide, ensuring that the final acid is obtained in high purity without the formation of difficult-to-remove byproducts. This streamlined workflow significantly reduces the number of unit operations, translating directly into lower capital expenditure and reduced operational overhead for manufacturers aiming for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this synthetic innovation lies in the palladium-catalyzed methoxycarbonylation mechanism, which proceeds through a well-defined catalytic cycle involving oxidative addition, CO insertion, and nucleophilic attack. Initially, the zero-valent palladium species undergoes oxidative addition into the carbon-halogen bond of the starting pyridine derivative, forming a stable organopalladium complex. This step is crucial as it activates the relatively inert aryl halide towards subsequent transformation, with the electron-withdrawing nature of the pyridine nitrogen and the fluorine substituent facilitating this process. Following oxidative addition, carbon monoxide coordinates to the metal center and inserts into the palladium-carbon bond, generating an acyl-palladium intermediate. This insertion step is highly exothermic and drives the reaction forward, effectively locking the carbonyl group into the molecular framework with perfect regiocontrol at the 2-position.
Subsequent nucleophilic attack by methanol, facilitated by the presence of a base such as triethylamine or potassium acetate, releases the methyl ester product and regenerates the active palladium catalyst. The choice of base is critical in neutralizing the hydrogen halide byproduct, preventing catalyst poisoning and ensuring high turnover numbers throughout the reaction course. The second stage involves the hydrolysis of the ester, where lithium hydroxide acts as a potent nucleophile to cleave the ester bond. This saponification proceeds smoothly at ambient temperature, avoiding the thermal stress that could lead to decarboxylation or defluorination. The mechanistic elegance of this pathway ensures that impurity profiles remain clean, with minimal formation of homocoupling products or dehalogenated species, thus simplifying downstream purification and enhancing the overall quality of the high-purity pharmaceutical intermediate.
How to Synthesize 3-Fluoro-4-Methylpyridine-2-Carboxylic Acid Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and safety, particularly during the high-pressure carbonylation step. Operators must ensure that the autoclave is properly sealed and purged before introducing carbon monoxide to maintain the specified pressure of 1.5MPa. The reaction temperature should be carefully monitored within the 50°C to 100°C range to balance reaction kinetics with catalyst stability. Following the carbonylation, the workup involves standard extraction and distillation techniques to isolate the ester intermediate, which serves as a stable precursor for the final hydrolysis. The detailed standardized synthesis steps see the guide below.
- Perform carbonylation of 2-halo-3-fluoro-4-methylpyridine using a palladium catalyst and carbon monoxide under alkaline conditions to form the methyl ester intermediate.
- Dissolve the resulting ester in tetrahydrofuran and cool in an ice-water bath before adding lithium hydroxide solution.
- Maintain the hydrolysis reaction at ambient temperature, then adjust pH to isolate the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial strategic advantages for organizations focused on optimizing their supply chain resilience and reducing total cost of ownership. By condensing the synthesis into just two high-yielding steps, the method drastically reduces the consumption of solvents and reagents compared to traditional multi-step routes. This reduction in material usage directly correlates to lower waste disposal costs and a smaller environmental footprint, aligning with modern green chemistry initiatives and regulatory compliance standards. Furthermore, the reliance on commodity chemicals like carbon monoxide and methanol, rather than exotic custom reagents, ensures that raw material sourcing remains stable and不受 market volatility. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and expensive organometallic reagents results in significant operational savings. The process utilizes common laboratory equipment such as high-pressure reactors and standard filtration setups, avoiding the need for specialized infrastructure. Additionally, the high conversion rates minimize the loss of valuable fluorinated starting materials, ensuring that every kilogram of input translates efficiently into saleable product. The simplified purification protocol further reduces labor costs and processing time, allowing for faster batch turnover and improved asset utilization within the production facility.
- Enhanced Supply Chain Reliability: The versatility of the starting materials, which can be chloro, bromo, or iodo derivatives, provides procurement managers with multiple sourcing options to mitigate supply risks. If one halogenated precursor faces availability issues, the process can be adapted to utilize an alternative without compromising the final product quality. This flexibility creates a robust supply buffer, ensuring that production never halts due to a single point of failure in the raw material supply chain. Moreover, the stability of the intermediate ester allows for stockpiling if necessary, adding another layer of security to the overall logistics strategy.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, having been demonstrated to work effectively in high-pressure vessels suitable for ton-scale production. The absence of heavy metal contaminants in the final product, thanks to efficient catalyst removal and purification steps, simplifies the regulatory filing process for new drug applications. The aqueous workup in the hydrolysis step generates benign salt byproducts that are easy to treat, reducing the burden on wastewater treatment plants. This environmental compatibility facilitates smoother permitting processes and strengthens the company's position as a sustainable partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs.
Q: What are the critical reaction parameters for the carbonylation step?
A: The process requires a pressure of approximately 1.5MPa of carbon monoxide and temperatures ranging between 50°C and 100°C, utilizing palladium catalysts such as [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride.
Q: How is the final purity of the acid ensured during hydrolysis?
A: High purity is achieved by conducting the hydrolysis at mild temperatures (25°C) using lithium hydroxide monohydrate, followed by precise pH adjustment to 2-3 for extraction, minimizing thermal degradation.
Q: Can this route accommodate different halogen starting materials?
A: Yes, the method is robust across chloro, bromo, and iodo substrates, offering flexibility in raw material sourcing depending on availability and cost considerations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Fluoro-4-Methylpyridine-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-fluoro-4-methylpyridine-2-carboxylic acid meets the highest industry standards. Our commitment to technical excellence means we can rapidly adapt the patented carbonylation process to fit your specific manufacturing constraints while maintaining optimal yield and quality.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this streamlined method. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us partner with you to accelerate your timeline to market with a supply chain that is both efficient and reliable.
