Advanced Synthesis of 6-Carboxyfluorescein: Overcoming Isomer Challenges for High-Purity Bio-Labeling Intermediates
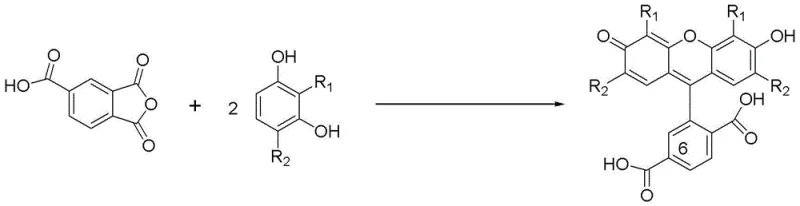
The landscape of fluorescent labeling reagents for biological macromolecules has long been dominated by the critical need for high-purity 6-carboxyfluorescein (6-FAM). As detailed in the groundbreaking patent CN102942553A, a novel preparation method has emerged that fundamentally addresses the historical bottlenecks associated with regioselectivity and yield in fluorescein synthesis. This technology leverages a specific metal catalyst system involving stannic chloride (SnCl4) within a methanesulfonic acid medium to drive the condensation of trimellitic anhydride with resorcinol derivatives. Unlike conventional approaches that struggle with inseparable isomeric by-products, this innovation achieves a dramatic shift in product distribution, elevating the ratio of the target 6-isomer against the unwanted 5-carboxyfluorescein from a problematic 1:1 to a highly favorable 2.6:1. For R&D directors and procurement specialists in the life sciences sector, this represents a pivotal advancement, transforming a low-efficiency process with a mere 32% separation yield into a robust manufacturing route capable of delivering 61% isolated yield. The implications for cost reduction in pharmaceutical intermediates manufacturing are profound, as the enhanced selectivity directly translates to reduced raw material consumption and simplified purification workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-carboxyfluorescein via Friedel-Crafts acylation has been plagued by a persistent and costly technical challenge: the formation of positional isomers. When trimellitic anhydride reacts with resorcinol under standard Lewis acid conditions, the electrophilic attack occurs with poor regioselectivity, generating approximately equivalent amounts of the desired 6-carboxy isomer and the undesired 5-carboxyfluorescein (5-CF). Because these two compounds are structural isomers with remarkably similar physicochemical properties, separating them requires extensive and expensive chromatographic techniques or multiple recrystallization steps, which inevitably lead to significant product loss. In many traditional protocols, the overall separation yield hovers around 32%, meaning that nearly two-thirds of the potential product value is lost to waste streams or remains trapped in difficult-to-process mother liquors. This inefficiency not only inflates the cost of goods sold (COGS) but also creates substantial environmental burdens due to the large volumes of solvents and silica gel required for purification, making scale-up for commercial production economically unviable for many suppliers.
The Novel Approach
The methodology disclosed in patent CN102942553A introduces a paradigm shift by utilizing tin tetrachloride (SnCl4) as a selective catalyst in a methanesulfonic acid solvent system. This specific combination creates a unique reaction environment that kinetically favors the formation of the 6-position substitution over the 5-position. By optimizing the molar ratios of trimellitic anhydride, resorcinol, and the tin catalyst to approximately 1:2:0.2, the process achieves a product ratio of 2.6:1, drastically reducing the burden on downstream purification. Furthermore, the workup procedure is elegantly simple: the reaction mixture is poured into ice water to precipitate the crude product, which is then purified using a specific methanol and hexane solvent mixture. This streamlined approach not only boosts the isolated yield to 61% but also eliminates the need for complex column chromatography, thereby enhancing the commercial scale-up of complex fluorescent intermediates. The result is a process that is not only chemically superior but also operationally simpler and more environmentally sustainable.
Mechanistic Insights into SnCl4-Catalyzed Regioselective Acylation
The success of this synthetic route lies in the precise modulation of Lewis acidity and solvent effects provided by the SnCl4/methanesulfonic acid system. Mechanistically, tin tetrachloride acts as a potent Lewis acid that coordinates with the carbonyl oxygen of the trimellitic anhydride, increasing the electrophilicity of the carbonyl carbon and facilitating the attack by the electron-rich resorcinol ring. However, the key differentiator is the solvent environment; methanesulfonic acid is a strong organic acid that likely protonates intermediate species or stabilizes specific transition states that favor attack at the 4-position of the resorcinol (which corresponds to the 6-position in the final xanthene dye structure). This selective activation suppresses the formation of the thermodynamic 5-isomer, which is typically favored in less controlled environments. The reaction proceeds through a cyclization step to form the xanthene core, and the presence of the carboxyl group is preserved throughout the acidic conditions, eventually forming a stable methanesulfonic acid adduct that can be easily isolated.
Impurity control is inherently built into this mechanism through the initial selectivity of the acylation step. By minimizing the generation of the 5-carboxy isomer at the source, the subsequent crystallization step becomes highly effective. The patent describes a recrystallization using a methanol and hexane mixture (1:4 volume ratio), which exploits the solubility differences between the desired 6-FAM adduct and remaining impurities. This physical purification is far more efficient when the crude mixture is already enriched with the target isomer. Finally, the hydrolysis step using aqueous NaOH cleaves the methanesulfonic acid adduct to release the sodium salt of 6-FAM, which is then acidified to precipitate the pure free acid. This multi-stage control ensures that the final product meets stringent purity specifications required for sensitive applications like PCR and DNA sequencing, where trace impurities can interfere with fluorescence signals.
How to Synthesize 6-Carboxyfluorescein Efficiently
The synthesis of 6-carboxyfluorescein via this patented route involves a carefully controlled sequence of acylation, crystallization, and hydrolysis steps designed to maximize regioselectivity and yield. The process begins with the dissolution of resorcinol in methanesulfonic acid, followed by the addition of trimellitic anhydride and the tin catalyst, and requires precise temperature management between 90°C and 110°C to ensure complete conversion while maintaining selectivity. Following the reaction, the product is isolated through precipitation in ice water and purified via a specific solvent recrystallization protocol before undergoing base-mediated hydrolysis to yield the final high-purity acid. For detailed operational parameters, stoichiometry, and safety considerations, please refer to the standardized synthesis guide below.
- Dissolve resorcinol derivatives in methanesulfonic acid and add trimellitic 1,2-anhydride under stirring at room temperature.
- Add tin tetrachloride (SnCl4) catalyst and heat the mixture to 90-110°C for 6-10 hours to complete the regioselective acylation.
- Precipitate the product in ice water, purify via methanol/hexane recrystallization, and hydrolyze the adduct with NaOH followed by acidification to obtain the free acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this SnCl4-catalyzed process offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage is the drastic simplification of the purification train; by shifting the isomer ratio from 1:1 to 2.6:1, the need for resource-intensive chromatographic separation is effectively eliminated. This reduction in processing complexity translates directly into significant cost savings in manufacturing, as it lowers solvent consumption, reduces labor hours associated with column packing and running, and minimizes waste disposal costs. Furthermore, the higher isolated yield of 61% compared to the traditional 32% means that less raw material is required to produce the same amount of finished goods, effectively nearly doubling the material efficiency of the production line. This efficiency gain provides a buffer against fluctuations in the pricing of key starting materials like resorcinol and trimellitic anhydride, ensuring more stable long-term pricing for customers.
- Cost Reduction in Manufacturing: The elimination of expensive and time-consuming chromatographic purification steps results in substantial operational expenditure (OPEX) reductions. By relying on crystallization rather than column chromatography, the process reduces the consumption of high-purity solvents and silica gel, which are major cost drivers in fine chemical production. Additionally, the near-doubling of the yield means that the fixed costs of production—such as energy, equipment depreciation, and labor—are amortized over a much larger output of saleable product, driving down the unit cost significantly without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures associated with difficult separations. Traditional methods often suffer from variable yields due to the sensitivity of isomer separation, leading to unpredictable delivery schedules. In contrast, this catalytic method provides consistent, high-yielding batches that can be reliably scheduled. The use of readily available and stable reagents like tin tetrachloride and methanesulfonic acid further secures the supply chain against raw material shortages, ensuring that production timelines are met consistently for critical biotech applications.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without the engineering challenges posed by large-scale chromatography. The simplified workup involving precipitation and filtration is easily adapted to standard reactor vessels, facilitating rapid scale-up. Moreover, the reduction in solvent waste and the avoidance of silica gel disposal align with modern green chemistry principles and environmental regulations, reducing the facility's environmental footprint and simplifying compliance with increasingly strict waste management protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 6-carboxyfluorescein using this advanced catalytic method. These insights are derived directly from the technical specifications and experimental data provided in patent CN102942553A, offering clarity on process capabilities and product quality. Understanding these details is essential for partners looking to integrate high-purity fluorescence labeling reagents into their supply chains.
Q: How does the SnCl4 catalytic system improve the isomer ratio compared to traditional methods?
A: Traditional Friedel-Crafts acylation often yields a nearly 1:1 mixture of 6-carboxyfluorescein and its 5-carboxy isomer, making separation difficult. The patented SnCl4/methanesulfonic acid system enhances regioselectivity, shifting the product ratio to approximately 2.6:1 in favor of the desired 6-isomer, significantly simplifying downstream purification.
Q: What are the key advantages of using methanesulfonic acid as the solvent in this process?
A: Methanesulfonic acid serves as both a solvent and a proton source that stabilizes the reaction intermediates. It allows for effective dissolution of the resorcinol starting materials and facilitates the activity of the tin tetrachloride Lewis acid catalyst, leading to higher conversion rates and easier product isolation upon quenching in ice water.
Q: Can this synthesis method be adapted for substituted fluorescein derivatives?
A: Yes, the protocol is versatile and applicable to various resorcinol derivatives, such as 4-chlororesorcinol and 4-methylresorcinol. The patent demonstrates successful synthesis of 2',7'-dichloro and 2',7'-dimethyl analogs with similarly high yields and purity, indicating broad substrate scope for specialized fluorescence labeling applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Carboxyfluorescein Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity 6-carboxyfluorescein plays in the advancement of diagnostic technologies and biological research. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this essential intermediate. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise isomer ratios and fluorescence quantum yields, guaranteeing that every batch meets the demanding requirements of the global life sciences industry. We are committed to leveraging innovative technologies, such as the SnCl4-catalyzed route, to deliver superior value to our partners.
We invite you to collaborate with us to optimize your supply chain for fluorescence labeling reagents. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our advanced manufacturing processes can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data for our 6-carboxyfluorescein inventory and to discuss route feasibility assessments for your next project. Let us be your trusted partner in delivering high-quality chemical solutions that drive innovation.
