Advanced Enzymatic Resolution Technology for High-Purity D-Biphenylalanine Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks more efficient pathways for producing chiral intermediates, particularly for complex molecules like neutral endopeptidase inhibitors. Patent CN101547893B introduces a transformative methodology for the recovery of L-biphenylalanine compound salts and the subsequent recycling of biphenylalanine ester compounds. This technology addresses a critical bottleneck in enzymatic resolution processes where the unwanted isomer is typically discarded or subjected to cumbersome recovery procedures involving acidification and crystallization. By leveraging a specific protease derived from the genus Bacillus in a biphasic solvent system, this invention enables the direct extraction of the L-isomer salt from the aqueous layer. This approach not only streamlines the purification of the desired D-enantiomer but also creates a closed-loop system for raw material utilization, representing a significant leap forward in sustainable pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of optically active biphenylalanine derivatives via enzymatic hydrolysis has been plagued by inefficiencies in handling the non-target isomer. In standard protocols, after the enzyme selectively hydrolyzes one enantiomer, the resulting mixture contains the desired ester and the salt of the unwanted amino acid. To recover this unwanted salt for potential reuse, manufacturers historically had to lower the pH significantly to convert the salt into its free acid form. This acidification step often necessitates the use of strong mineral acids, followed by complex crystallization procedures to isolate the free acid. These additional unit operations increase processing time, generate substantial acidic wastewater, and often result in yield losses due to the solubility characteristics of the free acid. Furthermore, converting the free acid back into an ester for recycling requires re-esterification, adding further cost and complexity to the supply chain.
The Novel Approach
The methodology disclosed in CN101547893B circumvents these traditional pitfalls by maintaining the L-biphenylalanine in its salt form throughout the recovery process. By carefully controlling the hydrolysis conditions within a pH range of 6 to 13 using a Bacillus-derived protease, the reaction mixture naturally separates into an organic layer containing the unreacted D-ester and an aqueous layer containing the L-salt. The innovation lies in the subsequent treatment of this aqueous layer: by adding specific inorganic salts and an organic solvent such as methyl tert-butyl ether (MTBE) or toluene, the L-salt can be directly extracted into the organic phase without ever becoming a free acid. 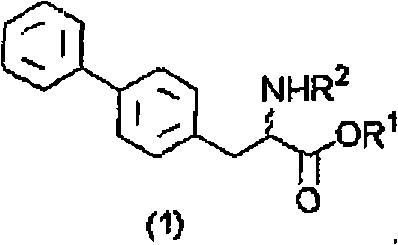 This direct extraction eliminates the need for acidification and crystallization, drastically reducing the number of processing steps and minimizing waste generation while preserving the optical integrity of the recovered material for downstream recycling.
This direct extraction eliminates the need for acidification and crystallization, drastically reducing the number of processing steps and minimizing waste generation while preserving the optical integrity of the recovered material for downstream recycling.
Mechanistic Insights into Bacillus Protease-Catalyzed Resolution
The core of this technological advancement relies on the high enantioselectivity of proteases from the genus Bacillus, particularly Alcalase derived from Bacillus licheniformis. These enzymes exhibit a profound preference for hydrolyzing the L-enantiomer of the biphenylalanine ester under mild alkaline conditions. The reaction is conducted in a mixed solvent system, typically comprising water and a hydrophobic organic solvent like MTBE, which facilitates the partitioning of products. The pH is meticulously maintained between 6.0 and 9.5, often using buffers such as taurine or phosphate, to ensure optimal enzyme activity and stability. Under these conditions, the L-ester is rapidly hydrolyzed to the corresponding carboxylate salt, which remains soluble in the aqueous phase, while the D-ester remains intact in the organic phase. This kinetic resolution is highly efficient, with experimental data indicating optical purities exceeding 99% ee for both the recovered D-ester and the generated L-salt, ensuring that the final pharmaceutical intermediate meets stringent regulatory standards for chirality.
Beyond the initial resolution, the patent details a robust mechanism for closing the material loop, which is critical for economic viability. Once the L-salt is extracted, it is not discarded but rather converted back into the starting ester. This is achieved through a two-step sequence: esterification followed by racemization. The L-salt is first esterified, preferably using dimethyl sulfate in the presence of a base like sodium bicarbonate, to form the L-ester. Subsequently, this L-ester is subjected to racemization using alkali metal alkoxides, such as sodium methoxide, in an alcoholic solvent. 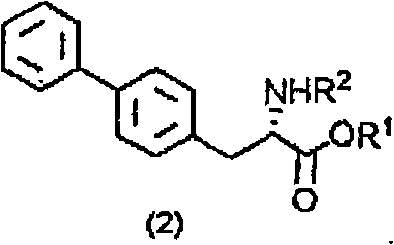 This base-catalyzed racemization effectively scrambles the chiral center, regenerating the racemic DL-ester mixture which can then be fed back into the enzymatic hydrolysis reactor. This cyclic process ensures that nearly 100% of the starting material is eventually converted into the desired D-isomer product, maximizing atom economy and minimizing raw material costs.
This base-catalyzed racemization effectively scrambles the chiral center, regenerating the racemic DL-ester mixture which can then be fed back into the enzymatic hydrolysis reactor. This cyclic process ensures that nearly 100% of the starting material is eventually converted into the desired D-isomer product, maximizing atom economy and minimizing raw material costs.
How to Synthesize D-Biphenylalanine Ester Efficiently
Implementing this resolution technology requires precise control over reaction parameters to achieve the high yields and optical purities described in the patent. The process begins with the preparation of the racemic biphenylalanine ester, which is then subjected to enzymatic hydrolysis in a biphasic system. Operators must monitor the pH closely, adjusting it with potassium hydroxide or similar bases to stay within the optimal window for the protease. Following the reaction, the phase separation is critical; the organic layer containing the D-product is separated, while the aqueous layer undergoes the salting-out extraction. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures for the esterification and racemization stages, are outlined below to guide process development teams in scaling this route.
- Hydrolyze DL-biphenylalanine ester in a mixed solvent of MTBE and water using Bacillus protease at pH 6-13 to separate L-salt and D-ester.
- Extract the L-biphenylalanine salt from the aqueous layer by adding inorganic salts and organic solvent, avoiding acidification steps.
- Recycle the recovered L-salt via esterification and base-catalyzed racemization to regenerate the DL-starting material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic resolution and recycling protocol offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the acidification and crystallization steps traditionally required to handle the L-isomer byproduct, manufacturers can significantly reduce the consumption of auxiliary chemicals such as strong acids and solvents. This reduction in reagent usage translates directly into lower variable costs per kilogram of produced intermediate. Furthermore, the ability to recycle the L-isomer back into the process means that the effective yield of the desired D-isomer from the initial racemic starting material approaches theoretical maximums, thereby reducing the overall demand for raw biphenylalanine precursors and insulating the supply chain from fluctuations in bulk chemical pricing.
- Cost Reduction in Manufacturing: The elimination of the free acid isolation step removes a major cost center associated with acid consumption, neutralization waste treatment, and energy-intensive crystallization processes. Additionally, the recycling loop ensures that no chiral material is wasted, effectively doubling the utility of the initial racemic feedstock without requiring additional synthetic steps to create new starting material from scratch. This inherent efficiency drives down the cost of goods sold (COGS) for the final D-biphenylalanine derivative, making it a more competitive option for generic drug manufacturers seeking to optimize their margin structures in a price-sensitive market environment.
- Enhanced Supply Chain Reliability: Relying on a process that maximizes raw material utilization reduces the dependency on external suppliers for bulk chiral starting materials. By internally recycling the L-isomer, the manufacturing facility becomes more self-sufficient, mitigating risks associated with supply disruptions or quality variability from upstream vendors. The use of robust, commercially available enzymes like Alcalase further stabilizes the supply chain, as these biocatalysts are produced at a global scale with consistent quality, ensuring that the resolution process can be sustained over long production campaigns without the risk of enzyme shortage or performance drift.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing common industrial solvents like MTBE and toluene which are easily recovered and reused. The avoidance of strong acid waste streams simplifies wastewater treatment requirements, aligning with increasingly stringent environmental regulations regarding effluent discharge. The simplified unit operations also facilitate easier technology transfer from pilot plant to commercial scale, reducing the time-to-market for new drug formulations that rely on this key intermediate. This environmental and operational efficiency positions the manufacturer as a responsible partner capable of meeting the rigorous sustainability audits required by top-tier multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biphenylalanine resolution technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process suitability for your specific application. Understanding these nuances is essential for R&D teams planning to integrate this methodology into their existing production lines or for procurement specialists assessing the long-term viability of suppliers utilizing this proprietary technique.
Q: What is the primary advantage of the salt recovery method in CN101547893B?
A: The method allows for the direct recovery of L-biphenylalanine as a salt from the aqueous layer without converting it to a free acid, significantly simplifying the workflow and reducing waste compared to traditional acidification and crystallization methods.
Q: Which enzyme is preferred for the hydrolysis of biphenylalanine esters?
A: Proteases derived from microorganisms belonging to the genus Bacillus, specifically Alcalase from Bacillus licheniformis, are preferred due to their excellent enantioselectivity and stability in the pH 6-13 range.
Q: How is the L-isomer recycled back to the starting material?
A: The recovered L-salt undergoes esterification using dimethyl sulfate and a base, followed by racemization using alkali metal alkoxides like sodium methoxide, effectively converting it back into the DL-biphenylalanine ester for reuse.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Biphenylalanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral resolution technologies in the modern pharmaceutical landscape. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the enzymatic resolution of biphenylalanine are executed with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including optical purity analysis via chiral HPLC, guaranteeing that every batch of D-biphenylalanine intermediate meets the highest global standards. We are committed to leveraging advanced process chemistry to deliver cost-effective solutions without compromising on quality or regulatory compliance.
We invite you to collaborate with our technical team to explore how this innovative recycling methodology can be adapted to your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of implementing this closed-loop resolution strategy. We encourage you to contact our technical procurement team today to request specific COA data for our biphenylalanine derivatives and to discuss comprehensive route feasibility assessments tailored to your development timeline and commercial goals.
