Industrial Scale-Up of 3,4-Thiophenedicarboxaldehyde via Mild Vilsmeier-Haack Formylation
The global demand for high-performance heterocyclic building blocks has intensified, particularly for compounds serving as critical precursors in the development of advanced pharmaceutical agents and organic electronic materials. Patent CN102558139B introduces a transformative methodology for the synthesis of 3,4-thiophenedicarboxaldehyde (CAS: 1073-31-0), addressing long-standing inefficiencies in traditional production routes. This technical breakthrough shifts the paradigm from hazardous cryogenic lithiation to a robust, two-step sequence utilizing accessible reagents like sodium methoxide and bis(trichloromethyl)carbonate. For R&D directors and procurement strategists, this patent represents a pivotal opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality while mitigating the risks associated with volatile organometallic reagents. The disclosed process not only enhances the safety profile of the manufacturing workflow but also aligns with modern green chemistry principles by enabling solvent recovery and operating under significantly milder thermal conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN102558139B, the prevailing industrial standard for synthesizing 3,4-thiophenedicarboxaldehyde relied heavily on direct lithiation strategies involving n-butyllithium. This conventional approach necessitates extremely harsh reaction environments, specifically requiring temperatures as low as -78°C to maintain the stability of the reactive organolithium species. Such cryogenic conditions impose a substantial burden on manufacturing infrastructure, demanding specialized cooling equipment and rigorous energy consumption that drastically inflates operational expenditures. Furthermore, the inherent pyrophoric nature of n-butyllithium introduces severe safety hazards, complicating logistics and requiring extensive safety protocols that can slow down production throughput. The complexity of handling these moisture-sensitive reagents often leads to batch-to-batch variability, making it difficult to achieve the stringent purity standards required for high-value applications in the electronics and medicinal chemistry sectors without costly additional purification steps.
The Novel Approach
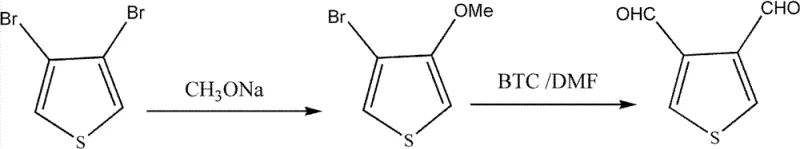
In stark contrast to the limitations of lithiation, the novel approach outlined in the patent employs a strategic two-step pathway that begins with a copper-catalyzed nucleophilic substitution followed by a Vilsmeier-Haack formylation. By utilizing 3,4-dibromothiophene as the starting material, the process first converts one bromine atom into a methoxy group using sodium methoxide under moderate heating, typically between 65°C and 75°C. This initial transformation sets the stage for the subsequent formylation, where bis(trichloromethyl)carbonate (BTC) and N,N-dimethylformamide (DMF) generate the active Vilsmeier reagent in situ. This methodology eliminates the need for cryogenic cooling, allowing reactions to proceed at manageable temperatures ranging from 50°C to 80°C. The shift to these milder conditions not only simplifies the engineering requirements for reactor design but also significantly reduces the risk profile of the operation, thereby facilitating cost reduction in fine chemical manufacturing and enabling smoother scale-up from laboratory to commercial production volumes.
Mechanistic Insights into Copper-Catalyzed Substitution and Vilsmeier Formylation
The first stage of this synthesis involves a sophisticated copper-catalyzed nucleophilic aromatic substitution, where the electron-deficient thiophene ring undergoes selective displacement of a bromine atom by a methoxide ion. The presence of catalysts such as cuprous chloride, cuprous bromide, or a specific mixture of potassium iodide and cupric oxide is critical for activating the carbon-bromine bond, lowering the activation energy required for the substitution to occur efficiently. This catalytic cycle ensures high regioselectivity, favoring the formation of 3-methoxy-4-bromothiophene while minimizing the formation of unwanted di-substituted byproducts like 3,4-dimethoxythiophene. The mechanistic precision here is vital for maintaining the integrity of the remaining bromine handle, which serves as the directing group for the subsequent formylation step. By optimizing the catalyst loading to between 2.5% and 10% of the total reaction mass, the process achieves a delicate balance between reaction rate and selectivity, ensuring that the intermediate is produced with a content purity exceeding 98% before moving to the next stage.
Following the isolation of the methoxy-bromo intermediate, the second stage leverages the classic Vilsmeier-Haack reaction mechanism to introduce the aldehyde functionalities. The reaction initiates with the formation of an iminium salt complex through the interaction of DMF and BTC, creating a potent electrophile capable of attacking the electron-rich positions on the thiophene ring. Upon heating to 50-80°C, this electrophilic species attacks the thiophene core, eventually leading to the hydrolysis of the iminium intermediate to reveal the aldehyde groups. This step is particularly efficient because the methoxy group acts as an activating substituent, enhancing the nucleophilicity of the adjacent positions and driving the formylation to completion. The result is a high-yielding conversion to 3,4-thiophenedicarboxaldehyde, with the patent reporting isolated yields for this specific step reaching upwards of 90%. The robustness of this mechanism allows for the use of common halogenated solvents like 1,2-dichloroethane or dichloromethane, which can be easily recovered and recycled, further enhancing the economic viability of the process.
How to Synthesize 3,4-Thiophenedicarboxaldehyde Efficiently
To implement this synthesis effectively in a pilot or production setting, operators must adhere to precise stoichiometric ratios and thermal profiles as defined in the patent examples. The process begins by charging a reactor with 3,4-dibromothiophene and a methanol solution of sodium methoxide, followed by the addition of the selected copper catalyst system. The mixture is then heated to the specified range of 65-75°C and maintained for a duration of 25 to 40 hours to ensure complete conversion to the mono-methoxy intermediate. After filtration and aqueous workup, the crude intermediate is subjected to the formylation conditions using the pre-formed Vilsmeier reagent. Detailed standardized operating procedures regarding quenching, pH adjustment to neutrality, and recrystallization techniques are essential to maximize the final purity and yield.
- Perform nucleophilic substitution on 3,4-dibromothiophene using sodium methoxide and a copper catalyst at 65-75°C to yield 3-methoxy-4-bromothiophene.
- Generate Vilsmeier reagent in situ using bis(trichloromethyl)carbonate and DMF at low temperature, then react with the intermediate at 50-80°C.
- Quench the reaction in ice water, adjust pH to neutral, extract with organic solvents, and purify via recrystallization to obtain the final dialdehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthetic route offers profound strategic benefits that extend beyond simple chemical efficiency. The elimination of n-butyllithium removes a major bottleneck in the supply chain, as this reagent often requires special hazardous material shipping and storage protocols that can delay production schedules. By replacing it with stable, commodity chemicals like sodium methoxide and BTC, manufacturers can streamline their raw material sourcing, reducing lead time for high-purity intermediates and insulating the supply chain from volatility in the market for specialty organometallics. Furthermore, the ability to operate at near-ambient or moderately elevated temperatures rather than deep cryogenic levels translates directly into lower utility costs and reduced wear on refrigeration infrastructure. These factors combine to create a manufacturing process that is not only safer but also significantly more resilient to disruptions, ensuring a steady flow of material to downstream customers in the pharmaceutical and electronic sectors.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of expensive, hazardous reagents with cost-effective alternatives and the optimization of energy usage. By avoiding the continuous energy drain associated with maintaining -78°C reaction environments, facilities can achieve substantial cost savings on electricity and cryogenic coolants. Additionally, the high selectivity of the copper-catalyzed step minimizes the generation of difficult-to-separate impurities, reducing the load on downstream purification units and lowering the overall cost of goods sold. The patent also highlights the recyclability of organic solvents such as dichloromethane and ethyl acetate, which further diminishes waste disposal costs and raw material consumption, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for maintaining uninterrupted production lines in the drug discovery and materials science industries. This synthetic route enhances reliability by utilizing reagents that are widely available from multiple global suppliers, reducing the risk of single-source dependency. The mild reaction conditions also mean that the process can be executed in a broader range of standard chemical reactors without the need for specialized cryogenic modifications, increasing the number of qualified contract manufacturing organizations (CMOs) capable of producing the material. This flexibility allows buyers to diversify their supply base and negotiate better terms, ensuring that project timelines are met without compromise due to equipment limitations or reagent shortages.
- Scalability and Environmental Compliance: Scaling chemical processes from grams to tons often exposes hidden inefficiencies, but this methodology is inherently designed for commercial scale-up of complex heterocycles. The use of biphasic workups and standard extraction techniques scales linearly, avoiding the mixing and heat transfer issues often encountered with cryogenic slurries. From an environmental perspective, the process aligns with increasingly strict regulatory frameworks by minimizing the use of pyrophoric materials and enabling solvent recovery loops. The reduction in hazardous waste generation and the lower energy footprint make this route more attractive for companies aiming to meet sustainability goals, facilitating smoother regulatory approvals and enhancing the corporate social responsibility profile of the final product supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,4-thiophenedicarboxaldehyde based on the disclosed patent technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines. The answers provided are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is the Vilsmeier-Haack method preferred over lithiation for 3,4-thiophenedicarboxaldehyde?
A: The Vilsmeier-Haack method avoids the use of cryogenic conditions (-78°C) and pyrophoric n-butyllithium required in traditional lithiation routes, significantly improving operational safety and reducing energy costs for industrial scale-up.
Q: What are the critical purity specifications for this intermediate?
A: High-purity specifications are essential for downstream applications in OLED materials and pharmaceuticals. The patented process achieves content levels exceeding 98-99% through optimized recrystallization and solvent recovery steps.
Q: Can the solvents used in this synthesis be recycled?
A: Yes, the patent explicitly highlights that organic solvents such as dichloromethane and ethyl acetate used in the extraction and reaction phases can be recovered and reused, contributing to a more sustainable and cost-effective manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Thiophenedicarboxaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality thiophene derivatives play in the advancement of next-generation pharmaceuticals and organic electronics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3,4-thiophenedicarboxaldehyde meets the exacting standards required by our global partners. Our facility is equipped to handle the specific thermal and safety requirements of the Vilsmeier-Haack process, providing a secure and compliant environment for the manufacture of this valuable intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your R&D and manufacturing operations.
