Industrial Scale-Up of High-Purity Secretin via Advanced Fmoc Solid-Phase Peptide Synthesis
Industrial Scale-Up of High-Purity Secretin via Advanced Fmoc Solid-Phase Peptide Synthesis
The pharmaceutical industry continuously seeks robust methodologies for the production of complex polypeptide hormones, and the synthesis of Secretin stands as a prime example of technological evolution in this sector. Patent CN103214568A introduces a groundbreaking solid-phase method that addresses the longstanding challenges of low yield and difficult purification associated with traditional synthetic routes. This innovation leverages the Fmoc-strategy combined with the strategic incorporation of pseudo-proline dipeptides to replace specific Serine residues within the peptide chain. By fundamentally altering the physicochemical properties of the growing chain, this approach effectively mitigates aggregation and enhances overall reaction efficiency. For R&D directors and procurement specialists seeking a reliable secretin supplier, understanding the mechanistic advantages of this patent is crucial for securing a stable supply of high-purity active pharmaceutical ingredients.
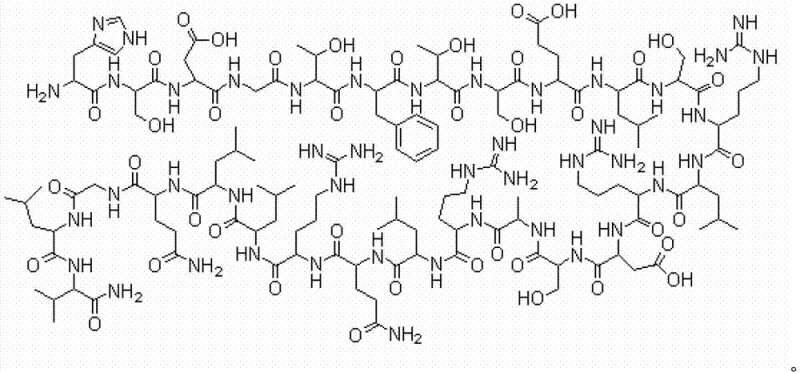
Secretin is a basic polypeptide consisting of 27 amino acids with a spiral structure, playing vital physiological roles in regulating pancreatic exocrine secretion and inhibiting gastric acid. The structural complexity, featuring a mix of hydrophobic residues like Leucine and Valine alongside hydrophilic residues such as Lysine and Arginine, historically posed significant solubility challenges during synthesis. The patent outlines a four-step process involving resin selection, sequential amino acid coupling, cleavage, and purification. This method is not merely an incremental improvement but a paradigm shift that facilitates the realization of industrialization for this critical hormone, ensuring that manufacturers can meet the stringent quality demands of the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for Secretin synthesis were plagued by inefficiencies that rendered large-scale production economically unviable and technically arduous. Traditional liquid-phase synthesis, as documented in early literature, suffered from extremely limited synthetic scope, typically concentrating on peptides of around 10 amino acids, necessitating tedious intermediate purifications that resulted in a total recovery of less than 10%. Furthermore, solid-phase synthesis utilizing the Boc-strategy introduced severe complications due to the requirement for repeated acid deprotection. Each cycle of removing the Boc group with 50% TFA caused approximately 1.4% of the peptide to detach prematurely from the resin, a loss that compounds drastically as the peptide chain lengthens. Additionally, acid catalysis frequently induced undesirable side reactions on sensitive side chains, making the Boc method particularly unsuitable for peptides containing acid-unstable residues like Tryptophan.
The Novel Approach
The novel approach detailed in the patent circumvents these historical bottlenecks by employing a solid-phase Fmoc strategy coupled with the innovative use of pseudo-proline derivatives. Unlike the harsh acidic conditions of the Boc method, the Fmoc strategy allows for rapid and complete deprotection under mild alkaline conditions, preserving the integrity of the peptide-resin linkage and minimizing side reactions. A key breakthrough is the replacement of specific Serine residues, particularly at positions 8 and 16, with pseudo-proline dipeptides. This modification disrupts the formation of rigid beta-sheet structures that typically cause peptide chain aggregation and poor solubility during elongation. Consequently, this method achieves a total recovery greater than 50% and an HPLC purity exceeding 99%, representing a substantial leap forward in cost reduction in peptide manufacturing and enabling feasible commercial scale-up of complex polypeptides.
Mechanistic Insights into Fmoc-Catalyzed Cyclization and Pseudo-Proline Substitution
The core of this synthetic success lies in the precise manipulation of peptide conformation during the elongation phase. In standard solid-phase peptide synthesis, the growing chain often adopts a beta-sheet conformation, leading to intermolecular hydrogen bonding that renders the resin-bound peptide insoluble and inaccessible to incoming activated amino acids. The introduction of pseudo-proline, a cyclic derivative of serine, introduces a kink in the peptide backbone due to its rigid oxazolidine ring structure. This steric constraint effectively breaks the regularity required for beta-sheet formation, thereby maintaining the peptide chain in a more soluble, random coil conformation. This enhanced solvation allows coupling reagents to access the reactive amine termini more efficiently, driving the reaction to completion and significantly reducing the formation of deletion sequences and other impurities that complicate downstream purification.
Furthermore, the choice of coupling agents and resin loading plays a pivotal role in the mechanistic success of this route. The patent specifies the use of coupling systems such as DIPCDI combined with additives like HOBt or HOAt, or alternatively DIPEA with uranium salts like HATU or HBTU. These reagents ensure rapid activation of the carboxyl group while minimizing racemization, a critical concern when handling chiral amino acids. The resin loading is meticulously optimized to a range of 0.1 to 0.6 mmol/g; loadings higher than this threshold increase steric crowding on the resin surface, hindering coupling efficiency, while lower loadings unnecessarily inflate material costs. The final cleavage step utilizes a sophisticated cocktail of TFA, Thioanisole (TA), Triisopropylsilane (TIS), Ethanedithiol (EDT), and water, where each component serves as a specific scavenger to trap reactive cationic species generated during the removal of acid-labile protecting groups, ensuring the final product remains free from alkylation byproducts.
How to Synthesize Secretin Efficiently
The synthesis of Secretin via this patented method requires strict adherence to protocol to maximize the benefits of the Fmoc strategy and pseudo-proline substitution. The process begins with the careful selection of the solid support, followed by the iterative cycle of deprotection and coupling using optimized molar ratios of activators. The strategic placement of pseudo-proline dipeptides at Ser-8 and Ser-16 is non-negotiable for achieving the reported high yields. While the general workflow is straightforward for experienced chemists, the specific nuances of resin swelling, washing protocols, and the composition of the cleavage cocktail are critical for reproducibility. For a detailed breakdown of the standardized operating procedures, including specific reaction times and solvent volumes, please refer to the technical guide below.
- Select an appropriate aminoresin carrier such as Rink Amide resin with a substitution degree between 0.1 and 0.6 mmol/g to ensure optimal coupling efficiency.
- Couple amino acids sequentially using Fmoc-strategy, strategically substituting Serine residues at positions 8 and 16 with pseudo-proline dipeptides to prevent beta-sheet formation.
- Cleave the full-length protected peptide from the resin using a TFA-based cocktail containing scavengers like TA, TIS, and EDT, followed by HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound economic and logistical benefits that extend beyond simple yield improvements. The elimination of harsh acid cycles and the reduction in purification complexity directly translate to a more streamlined manufacturing process with fewer unit operations. This simplification reduces the consumption of expensive solvents and reagents, leading to substantial cost savings in the overall production budget. Moreover, the robustness of the Fmoc strategy ensures a more predictable production schedule, mitigating the risks of batch failures that are common with older, less stable synthetic methods. This reliability is essential for maintaining continuous supply lines to pharmaceutical clients who demand consistent quality and timely delivery.
- Cost Reduction in Manufacturing: The implementation of pseudo-proline dipeptides significantly enhances the crude peptide purity, which drastically simplifies the downstream purification process. By reducing the burden on preparative HPLC and minimizing the loss of material during multiple purification steps, the overall cost of goods sold is effectively lowered. Additionally, the higher total recovery rate means that less starting material is required to produce the same amount of final API, optimizing raw material utilization and reducing waste disposal costs associated with failed batches or low-yield processes.
- Enhanced Supply Chain Reliability: The mild reaction conditions of the Fmoc strategy reduce the dependency on specialized corrosion-resistant equipment required for handling high concentrations of strong acids, thereby broadening the base of capable manufacturing partners. The improved solubility of the peptide chain during synthesis minimizes the risk of reactor blockages or incomplete reactions, ensuring that production timelines are met consistently. This operational stability allows for better capacity planning and reduces the lead time for high-purity polypeptides, providing a competitive edge in a market where speed to clinic is paramount.
- Scalability and Environmental Compliance: The process is designed with industrialization in mind, utilizing reagents and solvents that are manageable on a large scale without compromising safety or environmental standards. The reduction in side reactions means fewer toxic byproducts are generated, simplifying waste treatment and aligning with increasingly stringent global environmental regulations. The ability to scale from gram to kilogram quantities without significant re-optimization ensures that the supply chain can flexibly respond to market demand fluctuations, supporting both clinical trial materials and commercial launch volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Secretin using this advanced solid-phase methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent, providing clarity on how this technology resolves historical synthesis痛点. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process adoption.
Q: How does the use of pseudo-proline dipeptides improve Secretin synthesis yield?
A: Pseudo-proline dipeptides introduce a rigid structure that disrupts the formation of intermolecular beta-sheets during chain elongation. This significantly reduces peptide aggregation and racemization, particularly at Serine residues, leading to a total recovery greater than 50% compared to less than 10% in traditional liquid-phase methods.
Q: Why is the Fmoc strategy preferred over the Boc strategy for this specific polypeptide?
A: The Fmoc strategy utilizes mild alkaline conditions for deprotection, avoiding the repeated strong acid treatments required by the Boc strategy. This minimizes side reactions on acid-sensitive side chains and prevents the premature cleavage of the peptide from the resin, which is a common issue in long-chain synthesis using Boc chemistry.
Q: What are the critical parameters for resin selection in this synthesis?
A: The patent specifies using aminoresins like Rink Amide with a substitution degree strictly controlled between 0.1 and 0.6 mmol/g. Loading levels higher than 0.6 mmol/g can reduce coupling efficiency due to steric hindrance in long chains, while lower loading increases resin consumption and cost without proportional benefits.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Secretin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103214568A are fully realized in practical manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Secretin meets the highest international standards, providing our partners with the confidence needed to advance their drug development pipelines.
We invite you to engage with our technical procurement team to discuss how our advanced capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall expenditure. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
