Advanced Preparation of Monocyclic Beta-Lactam Antibiotic Intermediates for Global Pharma Supply Chains
Advanced Preparation of Monocyclic Beta-Lactam Antibiotic Intermediates for Global Pharma Supply Chains
The pharmaceutical landscape continuously demands more efficient and scalable routes for producing complex antibiotic intermediates, particularly those targeting resistant bacterial strains. Patent CN1067053C discloses a sophisticated preparation method for intermediates of heteroaroyl derivatives of monocyclic beta-lactam antibiotics, specifically focusing on compounds with potent anti-microbial activity against Gram-negative bacteria. This technology represents a significant leap forward in the synthesis of azetidinone-based therapeutics, offering a versatile platform for generating diverse analogues through the strategic manipulation of the quinoxaline side chain. The core innovation lies in the coupling of a specialized beta-lactam sulfonic acid core with a functionalized quinoxaline carboxylic acid derivative, enabling precise control over stereochemistry and substituent patterns. 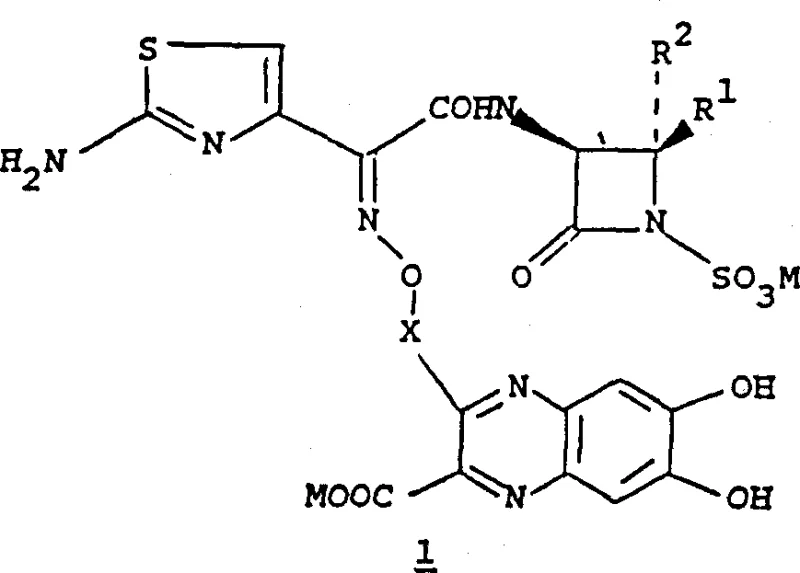 As a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is crucial for R&D teams aiming to optimize their API manufacturing pipelines. The disclosed compounds, represented generally by Formula I, incorporate a 4-hetaroylpyrazol or quinoxaline moiety linked via an oxyimino or alkyl spacer to the beta-lactam nucleus. This structural architecture is designed to enhance stability and bioavailability while maintaining the essential beta-lactam ring integrity required for bactericidal action. The patent details extensive experimental data, including specific embodiments for synthesizing various protected and unprotected forms, providing a robust blueprint for commercial scale-up of complex pharmaceutical intermediates.
As a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is crucial for R&D teams aiming to optimize their API manufacturing pipelines. The disclosed compounds, represented generally by Formula I, incorporate a 4-hetaroylpyrazol or quinoxaline moiety linked via an oxyimino or alkyl spacer to the beta-lactam nucleus. This structural architecture is designed to enhance stability and bioavailability while maintaining the essential beta-lactam ring integrity required for bactericidal action. The patent details extensive experimental data, including specific embodiments for synthesizing various protected and unprotected forms, providing a robust blueprint for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing monocyclic beta-lactam antibiotics often suffer from significant drawbacks related to yield, stereoselectivity, and the stability of key intermediates. Conventional acylation strategies frequently rely on highly reactive acid chlorides or mixed anhydrides that can lead to racemization at the chiral centers of the beta-lactam ring, particularly at the C3 and C4 positions. Furthermore, the introduction of complex heteroaromatic side chains, such as quinoxaline derivatives, typically requires harsh reaction conditions that may degrade the sensitive four-membered lactam ring. Many prior art processes involve multiple protection and deprotection steps that increase the overall step count, reduce atom economy, and generate substantial chemical waste, thereby escalating the cost reduction in API manufacturing. The inability to efficiently control the cis/trans isomerism during the coupling phase often results in difficult-to-separate mixtures, compromising the final purity specifications required for clinical applications.
The Novel Approach
The novel approach detailed in CN1067053C overcomes these hurdles by utilizing a modular coupling strategy that separates the construction of the side chain from the beta-lactam core assembly. By pre-forming the quinoxaline carboxylic acid derivative with appropriate protecting groups (such as tert-butyloxycarbonyl or benzyl), the process ensures that the sensitive hydroxylamine functionality remains stable until the final coupling stage. This method allows for the use of milder activation techniques, such as Mitsunobu conditions or nucleophilic substitution on activated leaving groups, which preserve the stereochemical integrity of the beta-lactam nucleus. The patent exemplifies this through the reaction of Formula 2 compounds with Formula 3 compounds, where the side chain is introduced as a pre-activated ester or via an oxyimino linkage. 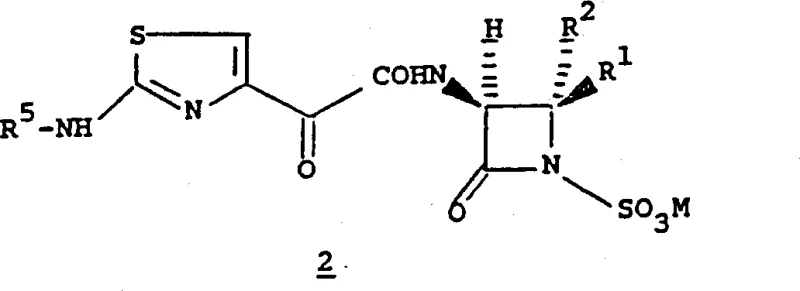 This decoupling of synthetic steps significantly simplifies the purification process, as impurities generated during side chain synthesis can be removed before the critical coupling reaction. Consequently, this approach facilitates the commercial scale-up of complex polymer additives and pharmaceutical intermediates by reducing the burden on downstream processing and improving overall process robustness.
This decoupling of synthetic steps significantly simplifies the purification process, as impurities generated during side chain synthesis can be removed before the critical coupling reaction. Consequently, this approach facilitates the commercial scale-up of complex polymer additives and pharmaceutical intermediates by reducing the burden on downstream processing and improving overall process robustness.
Mechanistic Insights into Quinoxaline Side Chain Coupling
The mechanistic pathway for constructing the target molecules involves a intricate sequence of condensation, functionalization, and coupling reactions centered around the quinoxaline ring system. The synthesis begins with the formation of the quinoxaline nucleus, typically achieved by the condensation of a 1,2-diaminobenzene derivative, such as 5,6-diamino-2,2-dimethyl-1,3-benzodioxole, with an alpha-keto ester. This cyclization establishes the aromatic core which is subsequently functionalized at the 2-position to introduce the linker arm. A critical aspect of the mechanism is the activation of the linker, which can be accomplished through halogenation (e.g., bromination) followed by nucleophilic displacement with a protected hydroxylamine species. 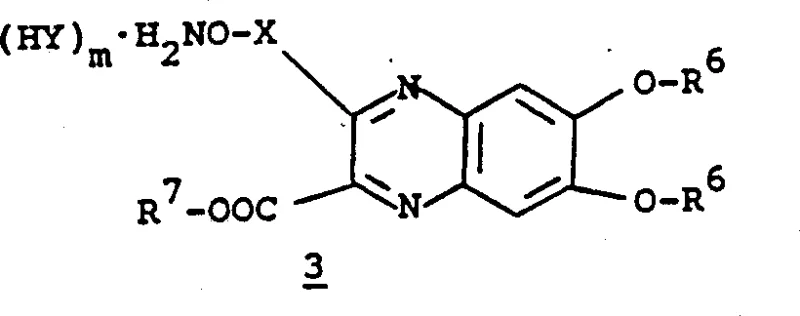 The patent describes alternative pathways involving the oxidation of methyl groups to aldehydes followed by Wittig olefination or direct phosphonate coupling, as illustrated in the reaction schemes for preparing Formula 12 and Formula 19 compounds. These mechanistic variations offer flexibility in tuning the length and saturation of the linker (represented by X in Formula I), allowing chemists to optimize the pharmacokinetic properties of the final antibiotic. The coupling with the beta-lactam core (Formula 7) is typically conducted in aqueous or mixed solvent systems at controlled pH levels (around pH 2.0 to 6.0) to ensure the solubility of the zwitterionic species while minimizing hydrolysis of the beta-lactam ring. The use of ion exchange resins, such as Dowex or XAD-2, in the final purification steps highlights the importance of removing inorganic salts and unreacted starting materials to achieve the high purity standards demanded by regulatory bodies.
The patent describes alternative pathways involving the oxidation of methyl groups to aldehydes followed by Wittig olefination or direct phosphonate coupling, as illustrated in the reaction schemes for preparing Formula 12 and Formula 19 compounds. These mechanistic variations offer flexibility in tuning the length and saturation of the linker (represented by X in Formula I), allowing chemists to optimize the pharmacokinetic properties of the final antibiotic. The coupling with the beta-lactam core (Formula 7) is typically conducted in aqueous or mixed solvent systems at controlled pH levels (around pH 2.0 to 6.0) to ensure the solubility of the zwitterionic species while minimizing hydrolysis of the beta-lactam ring. The use of ion exchange resins, such as Dowex or XAD-2, in the final purification steps highlights the importance of removing inorganic salts and unreacted starting materials to achieve the high purity standards demanded by regulatory bodies.
Impurity control is paramount in this synthesis, particularly regarding the formation of geometric isomers (E/Z) around the oxime ether linkage and the stereochemistry at the beta-lactam ring junction. The patent data indicates that the cis-isomer generally possesses higher biological activity, necessitating rigorous separation techniques. Mechanistic studies suggest that the choice of activating agent and reaction temperature plays a pivotal role in determining the isomeric ratio. For instance, the use of triphenylphosphine and diethyl azodicarboxylate (DEAD) in Mitsunobu-type couplings can influence the inversion of configuration, while acid-catalyzed deprotection steps must be carefully monitored to prevent epimerization. The detailed examples provided, such as Example 10 and Example 17, demonstrate that careful optimization of reaction times and stoichiometry can suppress the formation of trans-isomers and other regioisomeric byproducts, ensuring a cleaner impurity profile for the final active pharmaceutical ingredient.
How to Synthesize Monocyclic Beta-Lactam Intermediates Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction engineering, focusing on the sequential assembly of the quinoxaline side chain and its conjugation with the beta-lactam sulfonic acid core. The process generally initiates with the preparation of the protected hydroxylamine reagent, such as O-benzylhydroxylamine or N,N-di-BOC-hydroxylamine, which serves as the nucleophile for attaching the side chain. Subsequent steps involve the functionalization of the quinoxaline methyl group, either through radical bromination or oxidation to an aldehyde, followed by chain extension using phosphonate chemistry or Wittig reagents to establish the desired linker length (n=0 to 4). The final coupling is executed under mild acidic conditions to maintain the stability of the beta-lactam ring, followed by global deprotection using catalytic hydrogenation or acid hydrolysis.
- Preparation of the quinoxaline side chain precursor via condensation of diamino-benzodioxole derivatives with oxo-esters, followed by functionalization of the methyl group.
- Activation of the hydroxylamine moiety using protecting groups such as BOC or trityl to ensure stability during subsequent coupling reactions.
- Coupling the activated side chain with the beta-lactam sulfonic acid core under controlled pH conditions, followed by deprotection and purification via ion exchange chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic routes described in CN1067053C offers tangible benefits in terms of cost efficiency and supply reliability. The modular nature of the synthesis allows for the parallel production of key intermediates, such as the quinoxaline carboxylic acid derivatives and the beta-lactam sulfonic acid salts, which mitigates the risk of bottlenecks associated with linear synthesis paths. By utilizing robust protecting group strategies, the process minimizes the degradation of sensitive intermediates, leading to higher overall yields and reduced raw material consumption. This efficiency translates directly into cost reduction in pharmaceutical intermediates manufacturing, as fewer batches are required to meet production targets, and the load on waste treatment facilities is significantly lowered due to improved atom economy.
- Cost Reduction in Manufacturing: The elimination of harsh acylating agents and the use of stable, isolable intermediates reduce the need for specialized containment equipment and expensive reagents. The ability to purify intermediates via crystallization or standard chromatography before the final coupling step prevents the propagation of impurities, thereby reducing the cost associated with reprocessing off-spec material. Furthermore, the versatility of the quinoxaline scaffold allows for the potential sharing of common starting materials across different product lines, leveraging economies of scale to drive down unit costs substantially.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as substituted nitrobenzenes and standard amino acids, ensures a stable supply base that is less susceptible to geopolitical disruptions. The synthetic route avoids the use of scarce precious metal catalysts in the key coupling steps, relying instead on more abundant reagents like phosphines and carbodiimides, which enhances the resilience of the supply chain. Additionally, the robustness of the process allows for flexible manufacturing schedules, enabling rapid response to fluctuations in market demand for antibiotic APIs without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The processes described are amenable to large-scale production, with examples demonstrating successful execution on multi-gram scales that can be readily translated to kilogram and tonne levels. The use of aqueous workups and the avoidance of highly toxic solvents in the final purification stages align with modern environmental, health, and safety (EHS) standards, facilitating regulatory approval and reducing the environmental footprint of the manufacturing facility. This commitment to green chemistry principles not only ensures compliance with stringent global regulations but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these monocyclic beta-lactam intermediates. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What is the primary antibacterial spectrum of these monocyclic beta-lactam intermediates?
A: The compounds described in patent CN1067053C exhibit significant activity against Gram-negative bacteria, making them valuable precursors for treating urinary tract and respiratory tract infections in mammals.
Q: How does the quinoxaline side chain improve the synthesis process?
A: The quinoxaline carboxylic acid derivatives provide a robust and stable scaffold that allows for versatile coupling strategies, including Mitsunobu reactions and nucleophilic substitutions, facilitating easier scale-up compared to unstable acylating agents.
Q: What are the critical purity specifications for these intermediates?
A: High-performance liquid chromatography (HPLC) analysis in the patent examples indicates purity levels exceeding 95% for final zwitterionic compounds, requiring rigorous control of stereoisomers (cis/trans) during the coupling steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monocyclic Beta-Lactam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation antibiotics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence means we can navigate the complexities of stereochemical control and impurity management inherent in beta-lactam synthesis.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your specific drug development needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will empower your R&D and supply chain teams to make informed decisions. Let us be your trusted partner in delivering reliable, high-purity pharmaceutical intermediates that drive innovation and therapeutic success.
