Advanced Protected Amino Acid Halides for High-Efficiency Polyamide Manufacturing
Advanced Protected Amino Acid Halides for High-Efficiency Polyamide Manufacturing
The landscape of synthetic organic chemistry, particularly within the realm of biotechnology and pharmaceutical intermediates, is constantly evolving to meet the demands for higher purity and greater efficiency. Patent CN1561333A introduces a transformative approach to the synthesis of polyamides, specifically focusing on the generation of protected amino acid halides. These compounds serve as critical building blocks for creating sequence-specific DNA-binding molecules, such as those containing N-methylimidazole and N-methylpyrrole subunits. The traditional reliance on peptide coupling agents has long been a bottleneck in terms of speed and purity, but this patented methodology leverages alpha-haloenamine reagents to overcome these historical limitations. By shifting the paradigm from slow, equilibrium-driven couplings to rapid, high-yield halide activations, the technology offers a robust pathway for producing complex heteroaromatic oligomers. For industry leaders seeking a reliable polyamide intermediate supplier, understanding the mechanistic advantages of this acid halide generation is crucial for optimizing supply chains and reducing time-to-market for gene regulation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyamides, especially those incorporating heteroaromatic rings like imidazoles and pyrroles, has relied heavily on carbodiimides and uranium-based coupling reagents such as HBTU. While these reagents are standard in peptide chemistry, they present significant drawbacks when applied to the specific electronic environments of heteroaromatic systems. The formation of amide linkages between heteroaromatic units is notoriously sluggish, often requiring extended reaction times that exceed one hour per residue, which drastically reduces throughput in combinatorial library synthesis. Furthermore, these coupling agents frequently generate difficult-to-remove byproducts, such as urea derivatives, which contaminate the final product and necessitate laborious purification steps like preparative HPLC. The harsh electrochemical conditions sometimes required to activate carboxylic acids can also compromise sensitive protecting groups, leading to premature deprotection and side reactions that lower overall yield. Consequently, the conventional methods limit the ability to produce high-purity polyamides in the quantities necessary for high-throughput screening and clinical development.
The Novel Approach
In stark contrast to traditional coupling, the methodology described in CN1561333A utilizes alpha-haloenamine reagents to convert protected carboxylic acids directly into highly reactive acid halides. This shift in strategy bypasses the slow kinetics of direct amide bond formation by creating a superior electrophile that reacts rapidly with amine nucleophiles. The process can be executed in solution or, more advantageously, using immobilized reagents on solid supports, which simplifies the workup procedure to a mere filtration step. This novel approach not only accelerates the coupling reaction to completion in less than 30 minutes but also ensures that the resulting polyamide chains are formed with exceptional fidelity and minimal racemization. The ability to use stoichiometric amounts of monomers rather than large excesses further enhances the economic viability of the process. For procurement managers focused on cost reduction in pharmaceutical manufacturing, this efficiency translates directly into lower raw material consumption and reduced waste disposal costs, making it a superior choice for large-scale operations.
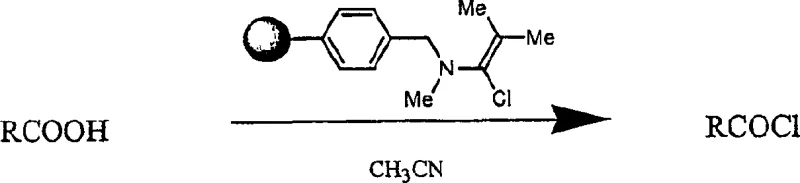
Mechanistic Insights into Alpha-Haloenamine Activation
The core innovation of this technology lies in the unique reactivity of the alpha-haloenamine species, which acts as a potent chlorinating or brominating agent under mild conditions. The mechanism involves the nucleophilic attack of the carboxylic acid oxygen on the electrophilic carbon of the alpha-haloenamine, facilitated by the leaving group ability of the amine moiety. This interaction results in the formation of a mixed anhydride-like intermediate that rapidly collapses to release the desired acid halide and a stable urea byproduct. When the alpha-haloenamine is immobilized on a polymer support, such as cross-linked polystyrene, the reaction becomes heterogeneous, allowing for easy separation of the reagent from the product mixture. This solid-phase variation is particularly advantageous for automation, as it eliminates the need for aqueous workups and solvent exchanges that typically plague solution-phase synthesis. The stability of the generated acid halides, particularly when protected with groups like Boc or Fmoc, ensures that they can be stored or transported without significant degradation, providing flexibility in the manufacturing schedule.
Furthermore, the structural integrity of the heteroaromatic ring system is preserved throughout the activation process, which is critical for maintaining the DNA-binding specificity of the final polyamide. The mild reaction conditions, typically conducted at room temperature in solvents like acetonitrile, prevent the decomposition of sensitive functional groups that might occur under the acidic or thermal stress of traditional thionyl chloride methods. The use of pentavalent phosphorus halides in the preparation of the alpha-haloenamine precursor ensures a high degree of halogenation efficiency, leading to reagents with consistent activity batches. For R&D directors concerned with impurity profiles, this controlled activation mechanism minimizes the formation of side products, resulting in intermediates that meet stringent purity specifications required for biological applications. The versatility of the system allows for the synthesis of both monomeric units and pre-formed oligomers, enabling a modular approach to constructing complex polyamide architectures.
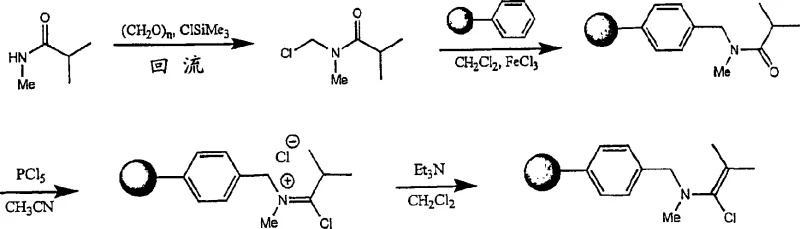
How to Synthesize Protected Amino Acid Halides Efficiently
The practical implementation of this synthesis route involves a straightforward sequence that begins with the preparation of the alpha-haloenamine reagent, followed by its reaction with the protected carboxylic acid substrate. The process is designed to be compatible with standard laboratory equipment and can be easily adapted for industrial scale-up using batch reactors or flow chemistry systems. The key to success lies in maintaining anhydrous conditions to prevent the hydrolysis of the reactive acid halide intermediate. Detailed protocols for the preparation of the immobilized reagent and its subsequent use in activating various heteroaromatic acids are provided in the patent examples, demonstrating the robustness of the method across different substrates. For technical teams looking to implement this technology, the following guide outlines the critical operational parameters.
- Preparation of the alpha-haloenamine reagent, optionally immobilized on a solid support like polystyrene, via reaction of a tertiary amide with a pentavalent phosphorus halide.
- Contacting the protected amino carboxylic acid (e.g., Boc-pyrrole-COOH) with the alpha-haloenamine reagent in a suitable solvent like acetonitrile at room temperature.
- Filtering off the solid support (if immobilized) to isolate the pure amino acid halide solution, ready for immediate coupling in polyamide chain elongation.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthesis methodology offers substantial strategic benefits for organizations managing the supply of complex chemical intermediates. The transition from traditional coupling agents to alpha-haloenamine activation addresses several pain points related to cost, timeline, and environmental compliance. By streamlining the synthetic route, companies can achieve a more predictable and resilient supply chain, reducing the risk of production delays caused by difficult purifications or low-yielding steps. The elimination of expensive coupling reagents and the reduction in solvent usage contribute to a leaner manufacturing process that aligns with modern sustainability goals. For supply chain heads, the ability to source high-quality intermediates produced via this efficient route ensures a steady flow of materials for downstream drug development programs.
- Cost Reduction in Manufacturing: The primary economic driver for this technology is the significant reduction in reagent costs associated with eliminating proprietary coupling agents like HBTU and carbodiimides. Additionally, the high conversion rates mean that less starting material is wasted, and the simplified purification process reduces the burden on downstream processing equipment. The ability to recover and potentially reuse the solid-supported reagent further enhances the cost-effectiveness of the operation. These factors combine to lower the overall cost of goods sold (COGS) for the final polyamide products, making them more competitive in the marketplace.
- Enhanced Supply Chain Reliability: The robustness of the alpha-haloenamine reagents ensures consistent batch-to-batch quality, which is essential for maintaining regulatory compliance in pharmaceutical production. The shortened reaction times allow for faster turnaround on custom synthesis orders, enabling suppliers to respond more agilely to fluctuating market demands. Furthermore, the stability of the intermediates reduces the logistical challenges associated with transporting sensitive chemicals, expanding the potential geographic reach of the supply network. This reliability fosters stronger partnerships between chemical manufacturers and their biotech clients.
- Scalability and Environmental Compliance: The method is inherently scalable, moving seamlessly from milligram-scale library synthesis to kilogram-scale production without significant process re-engineering. The reduction in hazardous waste generation, particularly the avoidance of heavy metal catalysts or toxic byproducts, simplifies waste management and lowers disposal costs. This alignment with green chemistry principles not only mitigates environmental impact but also future-proofs the manufacturing process against increasingly stringent regulatory standards. Companies adopting this technology demonstrate a commitment to sustainable practices that resonate with modern stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial applicability of the protected amino acid halides synthesized via this patented method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing workflows and quality assurance frameworks.
Q: What are the advantages of using alpha-haloenamine reagents over carbodiimides?
A: Alpha-haloenamine reagents offer significantly faster reaction times (often under 30 minutes), higher purity profiles by avoiding urea byproducts, and compatibility with automated solid-phase synthesis systems, unlike traditional carbodiimides which often require long coupling times and complex purification.
Q: Can this method be scaled for industrial production of polyamides?
A: Yes, the method is highly scalable. The use of immobilized reagents allows for simple filtration steps instead of complex extractions, making it ideal for large-scale batch processing and continuous flow chemistry in pharmaceutical manufacturing.
Q: What types of protecting groups are compatible with this synthesis?
A: The process is compatible with standard acid-labile and base-labile protecting groups, specifically tert-butoxycarbonyl (Boc) and 9-fluorenyl methoxy carbonyl (Fmoc), allowing for flexible orthogonal protection strategies in complex oligomer synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Protected Amino Acid Halides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production positions us as an ideal partner for bringing this advanced polyamide synthesis technology to the market. We possess the rigorous QC labs and stringent purity specifications necessary to ensure that every batch of protected amino acid halides meets the exacting standards required for biological applications. Our team is dedicated to supporting your R&D efforts with materials that enable faster discovery and more reliable data.
We invite you to contact our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this efficient synthesis route. We are prepared to provide specific COA data and route feasibility assessments to help you validate the quality and viability of our intermediates for your projects. Let us collaborate to accelerate your pipeline with superior chemical solutions.
