Advanced Manufacturing of 2-Bromo-5-Iodo-Benzyl Alcohol via Safe Halogenation Strategy
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways for halogenated aromatic building blocks, particularly those capable of undergoing diverse cross-coupling reactions. Patent CN113233957B introduces a groundbreaking preparation method for 2-bromo-5-iodo-benzyl alcohol, a versatile intermediate essential for constructing complex molecular architectures. This technology addresses critical safety and efficiency bottlenecks found in legacy synthesis routes by replacing hazardous reduction steps with a controlled hydrolysis strategy. For R&D directors and procurement specialists, this represents a significant opportunity to enhance supply chain resilience while mitigating operational risks associated with energetic reagents. The process leverages a logical four-step sequence starting from readily available o-toluidine, ensuring that the production of this high-value intermediate remains economically viable and environmentally compliant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated benzyl alcohols has relied heavily on the reduction of corresponding carboxylic acids using powerful reducing agents like lithium aluminum hydride (LiAlH4). While chemically effective, this approach presents severe industrial drawbacks, primarily the generation of large volumes of hydrogen gas which creates substantial explosion hazards in large reactors. Furthermore, alternative oxidative routes utilizing sodium percarbonate have been documented to produce excessive amounts of waste solvents and volatile organic compounds (VOCs), complicating waste treatment protocols. The handling of these dangerous oxidants requires rigorous safety measures, and any negligence in post-treatment can lead to catastrophic thermal events. Consequently, manufacturers face inflated operational costs due to the need for specialized containment equipment and extensive waste disposal procedures, making these conventional methods increasingly unsustainable for modern green chemistry standards.
The Novel Approach
The methodology disclosed in the patent circumvents these dangers by employing a constructive strategy that builds the alcohol functionality through nucleophilic substitution rather than destructive reduction. By utilizing a sequence of regioselective iodination, Sandmeyer bromination, radical benzylic bromination, and final hydrolysis, the process completely eliminates the need for lithium aluminum hydride. This shift not only removes the risk of hydrogen evolution but also avoids the generation of aluminum salt waste streams that are difficult to treat. The use of mild bases and standard halogenating reagents allows for simpler reaction control and easier workup procedures. This strategic pivot from reduction to substitution fundamentally alters the safety profile of the manufacturing process, enabling facilities to operate with lower risk exposure and reduced environmental footprint while maintaining high chemical fidelity.
Mechanistic Insights into Regioselective Halogenation and Substitution
The core of this synthetic success lies in the precise control of electrophilic aromatic substitution and radical mechanisms. The initial step involves the iodination of o-toluidine in an aqueous medium facilitated by a phase transfer catalyst, which ensures the iodine atom is directed exclusively to the para-position relative to the amino group. This high regioselectivity is crucial as it prevents the formation of isomeric impurities that would be difficult to separate later. Following this, the amino group is converted to a diazonium salt and subsequently replaced by a bromine atom via a Sandmeyer reaction using cuprous bromide, establishing the necessary halogen pattern on the aromatic ring. The subsequent radical bromination of the methyl group using N-bromosuccinimide (NBS) and AIBN initiator selectively targets the benzylic position without affecting the aromatic halogens. Finally, the benzylic bromide undergoes hydrolysis in dimethyl sulfoxide with a base to yield the target alcohol.
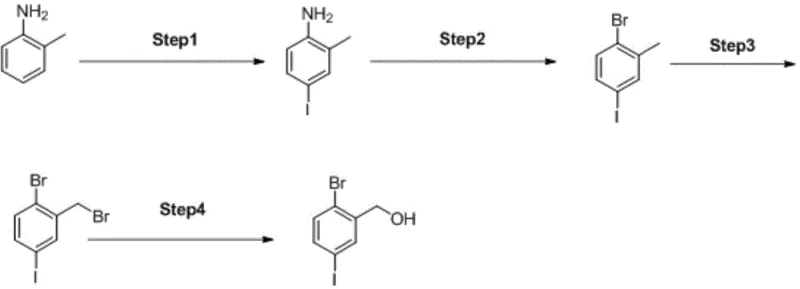
Understanding the impurity control mechanism is vital for ensuring the quality of the final pharmaceutical intermediate. The use of a phase transfer catalyst in the first step not only accelerates the reaction but also suppresses poly-iodination, ensuring a clean mono-iodinated product. In the radical bromination step, the stoichiometry of NBS is carefully controlled to prevent over-bromination or ring halogenation, which are common side reactions in free radical processes. The final hydrolysis step is conducted under mild alkaline conditions that favor SN2 substitution at the benzylic carbon while leaving the aryl bromide and iodide bonds intact, preserving the valuable coupling handles for downstream chemistry. This meticulous control over reaction parameters at each stage ensures that the impurity profile remains within stringent specifications required for GMP manufacturing environments.
How to Synthesize 2-Bromo-5-Iodo-Benzyl Alcohol Efficiently
Implementing this synthesis requires careful attention to temperature control and reagent addition rates to maximize yield and safety. The process begins with the suspension of o-toluidine and a weak carbonate base in water, where the temperature is maintained between 10°C and 15°C during the addition of the iodinating agent to manage exothermicity. Following isolation of the iodo-aniline, the diazotization is performed in sulfuric acid at low temperatures before warming for the Sandmeyer step. The radical bromination requires heating in a halogenated solvent with an initiator, followed by a final hydrolysis in DMSO. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Perform regioselective iodination of o-toluidine using a phase transfer catalyst and weak base to generate 2-methyl-4-iodoaniline.
- Convert the aniline derivative to 2-bromo-4-iodotoluene via diazotization followed by Sandmeyer bromination using cuprous bromide.
- Execute radical bromination of the methyl group using N-bromosuccinimide (NBS) and AIBN initiator to form the bromomethyl intermediate.
- Hydrolyze the bromomethyl group using a base in DMSO to yield the final 2-bromo-5-iodo-benzyl alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this novel synthetic route offers substantial advantages by simplifying the raw material portfolio and reducing dependency on hazardous specialty reagents. The elimination of lithium aluminum hydride removes the need for expensive inert atmosphere handling and specialized quenching protocols, directly translating to lower operational expenditures. Furthermore, the avoidance of sodium percarbonate reduces the burden on waste management systems, allowing facilities to allocate resources more efficiently towards production rather than compliance. The use of commodity chemicals like iodine, sodium nitrite, and NBS ensures a stable supply chain with minimal risk of vendor lock-in or price volatility. This stability is critical for long-term project planning and cost forecasting in the competitive pharmaceutical intermediate market.
- Cost Reduction in Manufacturing: The removal of hazardous reduction steps significantly lowers the capital expenditure required for safety infrastructure, such as hydrogen detection systems and explosion-proof reactors. By avoiding the generation of aluminum salts and excessive organic waste, the facility reduces its spending on waste treatment and disposal services. The simplified workup procedures, often involving straightforward filtration or extraction, minimize solvent consumption and energy usage during concentration steps. These cumulative efficiencies result in a leaner manufacturing process that delivers a more cost-effective product without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents like o-toluidine and standard halogenating agents ensures that production schedules are not disrupted by the scarcity of exotic catalysts. The robustness of the aqueous iodination step and the solid-state stability of intermediates facilitate easier storage and transportation logistics. This reliability allows supply chain managers to maintain lower safety stocks while ensuring continuous availability for downstream customers. The predictable reaction outcomes reduce the incidence of batch failures, further stabilizing the supply flow and strengthening relationships with key stakeholders in the value chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in multi-purpose chemical plants, such as stirred tank reactors and filtration units. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, future-proofing the manufacturing site against regulatory changes. The absence of high-pressure hydrogenation steps simplifies the engineering requirements for scaling from pilot to commercial tonnage. This ease of scale-up ensures that the supplier can rapidly respond to increased market demand without requiring extensive plant modifications or lengthy validation cycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate. These answers are derived directly from the patented technology to provide accurate guidance for potential partners. Understanding these details helps in assessing the feasibility of integrating this material into your specific drug substance or agrochemical synthesis pipelines. We encourage technical teams to review these points to fully appreciate the operational benefits offered by this advanced manufacturing route.
Q: Why is this new method safer than traditional lithium aluminum hydride reduction?
A: Traditional routes often utilize lithium aluminum hydride (LiAlH4) to reduce carboxylic acids, which generates significant amounts of hydrogen gas and poses explosion risks. This novel protocol eliminates the reduction step entirely by constructing the alcohol functionality through hydrolysis of a benzylic bromide, thereby removing the hazard of hydrogen evolution and simplifying safety protocols.
Q: How does this process improve waste management compared to oxidation methods?
A: Previous methods utilizing sodium percarbonate for oxidation generate substantial organic waste and volatile organic compounds (VOCs), alongside potential explosion hazards during post-treatment. The disclosed method employs aqueous workups and avoids dangerous oxidants, significantly reducing the volume of hazardous three-waste discharge and simplifying environmental compliance for large-scale production.
Q: What is the regioselectivity profile of the initial iodination step?
A: The process utilizes a phase transfer catalyst system that ensures high regioselectivity, directing the iodine atom exclusively to the para-position relative to the amino group. This specificity prevents the formation of isomeric byproducts, which streamlines downstream purification and ensures a consistent impurity profile critical for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-5-Iodo-Benzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality halogenated intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-bromo-5-iodo-benzyl alcohol meets the exacting standards required for pharmaceutical applications. Our commitment to safety and environmental stewardship mirrors the advantages of the patented process we employ, making us a responsible partner for your long-term projects.
We invite you to contact our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this safer, more efficient route. Our experts are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing planning. Let us collaborate to drive innovation and efficiency in your chemical supply chain.
