Advanced Synthesis of 2-Bromo-6-Chlorobenzonitrile: A Cost-Effective Route for Pharmaceutical Intermediates
Advanced Synthesis of 2-Bromo-6-Chlorobenzonitrile: A Cost-Effective Route for Pharmaceutical Intermediates
The pharmaceutical industry constantly demands more efficient pathways for critical building blocks, and the recent disclosure in patent CN116003294A presents a transformative approach to synthesizing 2-bromo-6-chlorobenzonitrile. This specific aromatic nitrile serves as a pivotal intermediate in the construction of complex drug molecules, where the cyano group acts as a crucial bioisostere to enhance binding affinity and metabolic stability. The patented methodology departs from traditional, hazardous routes by utilizing a streamlined three-step sequence starting from readily available 2-bromo-6-chlorobenzoic acid. By leveraging thionyl chloride for activation, aqueous ammonia for amidation, and cyanuric chloride for the final dehydration, this process achieves exceptional purity levels of 99% while maintaining remarkably stable yields. For R&D directors and procurement specialists alike, this innovation represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates without compromising on quality or safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated benzonitriles has been plagued by significant operational challenges that impact both cost and environmental compliance. Traditional methods often rely on the Rosenmund-von Braun reaction, which necessitates the use of stoichiometric amounts of cuprous cyanide at elevated temperatures. This approach introduces severe complications, including the generation of toxic heavy metal waste that requires expensive and rigorous removal processes to meet pharmaceutical grade specifications. Furthermore, direct dehydration of benzamides using aggressive reagents like phosphorus oxychloride or thionyl chloride often leads to harsh reaction conditions that promote side reactions, resulting in lower yields and difficult-to-remove impurities. These legacy processes not only inflate the cost of goods sold due to extensive purification requirements but also pose substantial risks to supply chain continuity due to the regulatory scrutiny surrounding cyanide handling and heavy metal disposal.
The Novel Approach
In stark contrast, the methodology outlined in CN116003294A introduces a gentle yet highly effective strategy that circumvents these historical bottlenecks. By converting the starting benzoic acid into an acyl chloride and subsequently into a benzamide, the process sets the stage for a mild dehydration step using cyanuric chloride. This reagent acts as a powerful dehydrating agent under neutral to slightly acidic conditions, typically around 20°C, which preserves the integrity of the sensitive bromo and chloro substituents on the aromatic ring. The elimination of heavy metal catalysts and the use of moderate temperatures drastically simplify the downstream processing, allowing for straightforward filtration and recrystallization. This shift not only enhances the safety profile of the manufacturing process but also ensures a cleaner impurity profile, making it an ideal candidate for the cost reduction in pharmaceutical intermediates manufacturing where purity is paramount.
Mechanistic Insights into Cyanuric Chloride-Mediated Dehydration
The core of this synthetic innovation lies in the strategic use of cyanuric chloride (2,4,6-trichloro-1,3,5-triazine) as a dehydrating agent, a mechanism that offers superior control compared to thermal dehydration. In the final step, the 2-bromo-6-chlorobenzamide reacts with cyanuric chloride in a polar aprotic solvent like DMF. The mechanism involves the activation of the amide oxygen by the electron-deficient triazine ring, facilitating the elimination of water to form the carbon-nitrogen triple bond. This reaction proceeds efficiently at temperatures between 15°C and 35°C, with optimal results observed at 20°C, preventing the thermal degradation often seen in high-temperature processes. The byproduct of this reaction is cyanuric acid, which is easily removed during the aqueous workup, leaving the desired nitrile in high purity. This mechanistic pathway ensures that the halogen atoms remain intact, avoiding the debromination or dechlorination side reactions that can occur with more aggressive reagents.
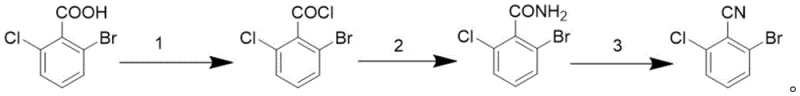
Furthermore, the precursor steps are equally optimized for impurity control. The initial conversion of the carboxylic acid to the acyl chloride using thionyl chloride is catalyzed by a small amount of DMF, which forms a reactive Vilsmeier-Haack intermediate that accelerates the reaction at low temperatures (5-10°C). This controlled addition prevents the formation of anhydrides or other oligomeric byproducts. Subsequently, the amidation step utilizes a biphasic system of ammonia water and ice water. The exothermic nature of the acyl chloride ammonolysis is managed by the ice bath, keeping the temperature near 0°C during addition and allowing it to warm to 20-30°C for the overnight stir. This thermal profile ensures complete conversion to the amide while minimizing hydrolysis back to the acid, resulting in a crude amide purity of over 97% before the final dehydration even begins.
How to Synthesize 2-Bromo-6-Chlorobenzonitrile Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the patented method. The process begins with the activation of 2-bromo-6-chlorobenzoic acid, where maintaining the temperature between 5°C and 10°C during the dropwise addition of thionyl chloride is critical to prevent runaway exotherms and ensure high conversion. Following the removal of excess thionyl chloride, the resulting acyl chloride is introduced to the ammonia solution without isolation, though the patent describes a workup to isolate the solid amide for better quality control. The final dehydration step demands strict temperature maintenance around 20°C; deviations above 35°C can lead to decreased purity, while temperatures below 15°C may slow the reaction kinetics unnecessarily. For a detailed breakdown of the standardized operating procedures and specific molar ratios required for commercial scale-up, please refer to the technical guide below.
- React 2-bromo-6-chlorobenzoic acid with thionyl chloride and catalytic DMF at 5-10°C, followed by reflux to form the acyl chloride intermediate.
- Add the crude acyl chloride to a cooled mixture of ammonia water and ice water, stirring overnight at room temperature to precipitate the benzamide.
- Dehydrate the benzamide using cyanuric chloride in DMF at 20°C, followed by aqueous workup and recrystallization to obtain high-purity nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling advantages that extend beyond simple chemical yield. The primary benefit lies in the drastic simplification of the purification workflow. By eliminating the need for heavy metal scavengers and complex chromatographic separations associated with copper-catalyzed methods, manufacturers can significantly reduce the consumption of specialized resins and solvents. This streamlining translates directly into lower operational expenditures and a reduced environmental footprint, aligning with modern green chemistry initiatives. Additionally, the use of commodity chemicals like thionyl chloride, ammonia, and cyanuric chloride ensures that the raw material supply is robust and less susceptible to the volatility often seen with specialized organometallic reagents, thereby enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the avoidance of expensive transition metal catalysts and the reduction in waste treatment costs. Traditional methods involving cuprous cyanide generate hazardous waste streams that require costly disposal protocols, whereas the byproducts of the cyanuric chloride method are benign and easily managed. Furthermore, the high yield stability across all three steps minimizes the loss of valuable starting materials, ensuring that the theoretical mass balance is closely approached in practice. This efficiency allows for a substantial reduction in the cost per kilogram of the final API intermediate, providing a competitive edge in pricing strategies for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Sourcing consistency is a critical factor for long-term production planning, and this route excels by relying on widely available bulk chemicals. Unlike specialized catalysts that may have single-source suppliers or long lead times, thionyl chloride and cyanuric chloride are produced globally at massive scales. This abundance mitigates the risk of supply disruptions and allows for flexible procurement strategies. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain against upstream fluctuations and ensuring continuous availability of the high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden bottlenecks, but this methodology is inherently designed for scalability. The exothermic steps are manageable with standard cooling infrastructure, and the absence of high-pressure or high-temperature requirements reduces the capital expenditure needed for reactor hardware. From an environmental perspective, the process generates minimal hazardous waste, simplifying compliance with increasingly stringent global environmental regulations. This ease of compliance reduces the administrative burden on EHS teams and lowers the risk of production stoppages due to regulatory audits, ensuring a smooth and uninterrupted flow of materials to the market.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its implementation. The following questions address common concerns regarding the practical application of patent CN116003294A, focusing on the specific advantages of the cyanuric chloride dehydration method and the quality of the resulting product. These insights are derived directly from the experimental data provided in the patent documentation, offering a clear picture of what partners can expect regarding yield, purity, and operational safety.
Q: Why is cyanuric chloride preferred over traditional dehydrating agents for this synthesis?
A: Cyanuric chloride operates under mild conditions (around 20°C) compared to harsh reagents like POCl3 or SOCl2 used in direct dehydration, significantly reducing side reactions and improving final product purity to 99%.
Q: Does this process involve heavy metal catalysts like copper?
A: No, unlike traditional Rosenmund-von Braun type reactions that require toxic cuprous cyanide, this patented route avoids heavy metals entirely, simplifying purification and waste treatment.
Q: What is the overall yield stability of this new method?
A: The patent data demonstrates consistent molar yields exceeding 98% for each step, ensuring a robust and scalable process suitable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-6-Chlorobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists has thoroughly analyzed the pathway described in CN116003294A and is fully prepared to execute this synthesis with precision and scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-bromo-6-chlorobenzonitrile meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to discuss your specific needs,索取 specific COA data, and review our comprehensive route feasibility assessments to ensure the seamless integration of this intermediate into your supply chain.
