Revolutionizing Organosulfur Chemistry: Scalable Electrochemical Oxidation for High-Purity Intermediates
The landscape of organosulfur chemistry is undergoing a significant transformation driven by the urgent need for greener, safer, and more atom-economical synthetic methodologies. Patent CN116219450A introduces a groundbreaking electrochemical synthesis method for producing sulfoxide and sulfone compounds, two structural motifs that are ubiquitous in the scaffolds of modern pharmaceuticals, agrochemicals, and functional materials. Unlike traditional approaches that rely heavily on stoichiometric chemical oxidants, this innovation leverages electricity as the primary reagent, utilizing water as the oxygen source to achieve selective oxidation. This paradigm shift not only addresses critical environmental concerns regarding waste generation but also offers a robust platform for the commercial scale-up of complex organosulfur intermediates. By replacing hazardous reagents with electrons, the technology provides a pathway to high-purity sulfoxide and sulfone derivatives that meet the stringent quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of sulfoxides and sulfones has been plagued by significant safety and environmental challenges inherent to traditional oxidation chemistries. The most common methods involve the use of strong, stoichiometric oxidizing agents such as meta-chloroperoxybenzoic acid (mCPBA), hydrogen peroxide with catalysts, or high-valence iodine salts. These reagents are often thermally unstable, posing severe explosion risks during storage and handling, particularly on a multi-ton scale. Furthermore, the atom economy of these reactions is frequently poor, generating substantial amounts of organic waste that require complex and costly disposal procedures. Another critical drawback is the issue of over-oxidation; controlling the reaction to stop at the sulfoxide stage without proceeding to the sulfone is notoriously difficult, often leading to mixtures that require energy-intensive purification steps. These factors collectively inflate the cost of goods sold (COGS) and extend the reducing lead time for high-purity organosulfur intermediates, creating bottlenecks in the supply chains of major pharmaceutical manufacturers.
The Novel Approach
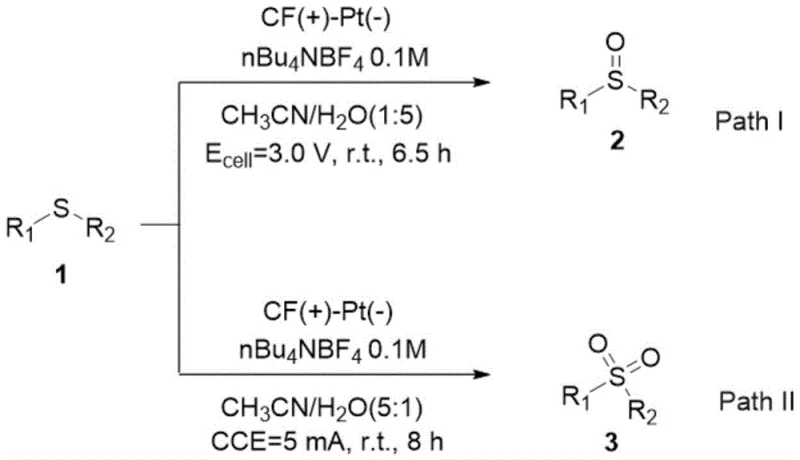
The electrochemical methodology disclosed in the patent represents a decisive break from these legacy constraints by introducing a tunable, reagent-free oxidation system. In this novel approach, the thioether substrate is dissolved in an aqueous electrolyte solution, and the oxidation state is dictated entirely by the applied electrical potential or current. For the synthesis of sulfoxides, the system operates under constant voltage conditions (2.8-3.2V), which provides just enough energy to effect mono-oxidation while preventing over-oxidation to the sulfone. Conversely, switching to constant current electrolysis or higher voltage ranges (5.0-10.0V) drives the reaction to completion, yielding the sulfone product with high selectivity. This precise electronic control eliminates the variability associated with chemical reagent addition rates and temperatures. Moreover, the use of water as the oxygen source means that the only byproduct is hydrogen gas, which can be safely vented or captured, rendering the process exceptionally clean. This level of control and simplicity makes it an ideal candidate for a reliable pharmaceutical intermediate supplier looking to modernize their production capabilities.
Mechanistic Insights into Electrochemical Anodic Oxidation
The core mechanism driving this transformation is the anodic oxidation of the sulfur atom within the thioether structure. When a potential is applied across the undivided electrolytic cell, the thioether molecules migrate to the anode surface (typically carbon cloth), where they undergo single-electron transfer to form a radical cation intermediate. In the presence of water molecules from the solvent system, this highly reactive intermediate is rapidly trapped, leading to the formation of the sulfur-oxygen bond. The beauty of this mechanism lies in its dependence on the electrode potential; by maintaining the cell voltage within the specific window of 2.8V to 3.2V, the system energetically favors the formation of the sulfoxide radical intermediate but lacks the driving force to oxidize the resulting sulfoxide further. This kinetic control is far superior to thermodynamic control achieved by chemical reagents, which often push reactions to the most stable oxidized state regardless of the desired product.
Furthermore, the choice of electrolyte and solvent plays a pivotal role in stabilizing these intermediates and ensuring efficient charge transfer. The patent specifies the use of tetrabutylammonium tetrafluoroborate in a mixture of acetonitrile and water. This specific solvent system ensures adequate solubility for both the organic thioether substrate and the inorganic electrolyte, facilitating a homogeneous reaction environment that minimizes side reactions. The absence of transition metal catalysts is another crucial mechanistic feature; unlike metal-catalyzed oxidations which risk contaminating the final product with trace heavy metals—a critical failure point for API manufacturing—this electrochemical method is inherently metal-free. This intrinsic purity profile simplifies downstream processing, as there is no need for expensive scavenging resins or complex filtration steps to remove metal residues, directly contributing to cost reduction in pharmaceutical intermediate manufacturing.
How to Synthesize Sulfoxide and Sulfone Compounds Efficiently
The practical implementation of this electrochemical protocol is remarkably straightforward, requiring standard electrolysis equipment rather than specialized high-pressure reactors. The process begins by dissolving the target thioether in the optimized acetonitrile-water solvent system containing the supporting electrolyte. The reaction is then initiated by applying the specific electrical parameters defined for the desired oxidation state. Because the reaction proceeds at room temperature and atmospheric pressure, the thermal load on the system is minimal, reducing the need for extensive cooling infrastructure. The detailed standardized synthesis steps, including exact molar ratios, electrode dimensions, and workup procedures, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve the thioether substrate in a mixed solvent system of acetonitrile and water containing a supporting electrolyte such as tetrabutylammonium tetrafluoroborate.
- For sulfoxide production, apply a constant cell voltage between 2.8V and 3.2V at room temperature to achieve selective mono-oxidation.
- For sulfone production, switch to constant current electrolysis (approx. 5mA) or higher voltage conditions (5.0-10.0V) to drive the reaction to the di-oxidized state.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic advantages that go beyond mere technical novelty. The elimination of hazardous chemical oxidants fundamentally alters the risk profile of the manufacturing process, removing the need for specialized storage facilities for peracids and reducing insurance premiums associated with handling explosive materials. Additionally, the simplicity of the workup procedure—often requiring only extraction and evaporation due to the lack of metal catalysts and organic byproducts—drastically reduces the consumption of auxiliary solvents and silica gel for chromatography. This streamlining of the post-reaction process leads to significant operational efficiencies and a smaller environmental footprint, aligning perfectly with the sustainability goals of modern multinational corporations.
- Cost Reduction in Manufacturing: The economic impact of switching to this electrochemical method is profound, primarily driven by the replacement of expensive stoichiometric oxidants with electricity and water. Traditional oxidants like mCPBA are not only costly to purchase but also generate equivalent masses of waste acid that must be neutralized and disposed of, adding hidden costs to the process. By contrast, the electrons used in this reaction are inexpensive, and the supporting electrolyte can potentially be recycled, creating a closed-loop system that minimizes raw material expenditure. Furthermore, the high selectivity of the reaction reduces the loss of valuable starting materials to over-oxidized byproducts, maximizing the yield per batch and improving the overall mass balance of the production line.
- Enhanced Supply Chain Reliability: Relying on external suppliers for specialized oxidizing agents introduces vulnerability into the supply chain, as these reagents often have limited shelf lives and strict transportation regulations. By generating the oxidative power in situ using electricity, manufacturers decouple their production capacity from the volatility of the reagent market. This self-sufficiency ensures consistent production schedules and protects against supply disruptions caused by logistical delays or regulatory changes in the transport of hazardous chemicals. The robustness of the method across a wide range of thioether substrates also means that a single production line can be easily adapted to manufacture diverse intermediates, enhancing flexibility and responsiveness to market demand.
- Scalability and Environmental Compliance: Scaling electrochemical processes is increasingly feasible with modern flow chemistry technologies, allowing for the seamless transition from laboratory gram-scale to multi-ton commercial production. The mild reaction conditions (room temperature, ambient pressure) mean that scaling up does not require exponentially larger cooling systems or reinforced pressure vessels, keeping capital expenditure (CAPEX) manageable. From an environmental compliance perspective, the process generates virtually no hazardous waste, with hydrogen gas being the only byproduct. This 'green' profile simplifies the permitting process for new manufacturing sites and helps companies meet increasingly rigorous global environmental, social, and governance (ESG) targets without compromising on output volume or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical oxidation technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this method for your specific application. Understanding these nuances is essential for R&D teams planning pilot runs and for procurement teams assessing the long-term viability of the supply source.
Q: What is the primary advantage of this electrochemical method over traditional oxidation?
A: The primary advantage is the use of water as the sole oxygen source, eliminating the need for toxic, hazardous, and expensive stoichiometric oxidants like peracids or high-valence iodine salts, thereby significantly reducing waste and safety risks.
Q: How is selectivity between sulfoxide and sulfone controlled?
A: Selectivity is precisely controlled by the electrical parameters: constant voltage electrolysis at lower potentials (2.8-3.2V) favors sulfoxide formation, while constant current or higher voltage conditions drive the reaction further to produce sulfones exclusively.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process operates under mild conditions (room temperature, atmospheric pressure) without requiring specialized high-pressure equipment or cryogenic cooling, making it highly amenable to commercial scale-up and continuous flow adaptation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfoxide Supplier
The electrochemical synthesis of sulfoxides and sulfones represents a mature and highly viable technology for the production of high-value fine chemicals. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green chemistry approach and have integrated similar advanced electrochemical capabilities into our CDMO infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the unique analytical requirements of electrochemically synthesized compounds, guaranteeing that every batch meets the highest international standards.
We invite you to explore how this innovative technology can optimize your supply chain and reduce your environmental impact. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our electrochemical capabilities can serve as a strategic asset for your organization's growth and sustainability goals.
