Advanced Manufacturing of 1-Beta-D-Ribofuranosyl Cytosine via Polymer-Supported Catalysis
The pharmaceutical industry constantly seeks robust synthetic pathways for critical nucleoside intermediates, and the technology disclosed in patent CN102911229B represents a significant leap forward in the manufacturing of 1-Beta-D-ribofuranosyl cytosine, commonly known as Cytidine. This compound serves as a fundamental structural fragment of RNA and acts as a pivotal precursor for a wide array of antiviral, antitumor, and anti-AIDS therapeutics, including Lamivudine and Cytarabine. Traditional synthetic methodologies have long been plagued by the use of hazardous homogeneous catalysts and complex purification protocols that hinder efficient scale-up. The innovation presented in this patent addresses these systemic bottlenecks by introducing a dual-protection strategy facilitated by N,N-dimethylformamide (DMF) and employing a recyclable polymer-supported Lewis acid catalyst. This approach not only enhances the chemical purity of the final product but also aligns with modern green chemistry principles by minimizing waste and eliminating toxic metal residues, thereby offering a compelling value proposition for global supply chains seeking reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of 1-Beta-D-ribofuranosyl cytosine has relied heavily on glycosylation reactions catalyzed by strong Lewis acids such as tin tetrachloride (SnCl4) or titanium tetrachloride (TiCl4). As illustrated in prior art references like the ultrasonic-assisted route shown below, these conventional methods present severe operational challenges for industrial manufacturers. The catalysts employed are often highly corrosive, moisture-sensitive, and dangerous to handle, requiring stringent safety measures that inflate operational expenditures. Furthermore, the homogenous nature of these catalysts makes their removal from the reaction mixture exceptionally difficult, often necessitating complex work-up procedures that can lead to product loss. A critical chemical deficiency in these older routes is the tendency for incomplete silylation protection of the cytosine base. This incomplete protection generates a spectrum of mono-protected impurities that are structurally similar to the desired product, making downstream purification arduous and resulting in lower overall yields and compromised purity profiles that fail to meet the rigorous standards of modern API manufacturing.
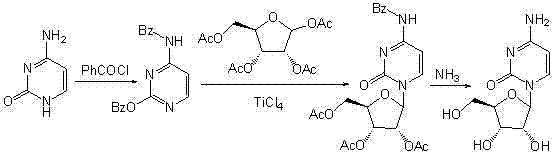
The Novel Approach
In stark contrast to the hazardous and inefficient legacy processes, the methodology outlined in CN102911229B introduces a transformative workflow that prioritizes both safety and chemical efficiency. The core innovation lies in the strategic addition of DMF during the silylation step, which acts as a promoter to ensure complete double silylation of the cytosine molecule, effectively shutting down the formation of problematic mono-protected byproducts. Following this protection step, the glycosylation is conducted using a macroporous styrene-based cation exchange resin loaded with a Lewis acid, such as titanium tetrachloride or tin tetrachloride. This heterogeneous catalytic system allows for a clean reaction environment where the catalyst can be physically separated from the product mixture via simple filtration. The reaction scheme depicted below highlights this streamlined progression from protected cytosine to the acetylated intermediate and finally to the high-purity target molecule. By replacing dangerous homogeneous catalysts with a stable, reusable solid support, this novel approach drastically simplifies the post-reaction processing, reduces environmental impact, and ensures a consistent, high-quality output suitable for sensitive pharmaceutical applications.
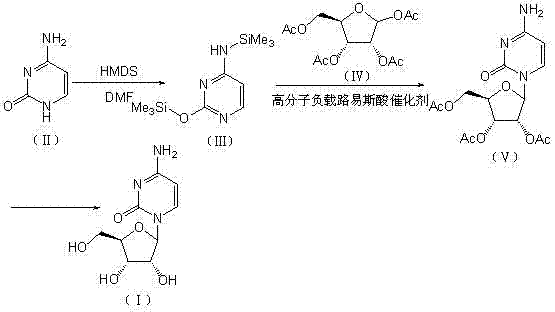
Mechanistic Insights into Polymer-Supported Lewis Acid Catalyzed Glycosylation
The mechanistic superiority of this synthesis lies in the precise control over the nucleophilic attack during the glycosylation phase. In the presence of the polymer-supported Lewis acid catalyst, the anomeric acetate group of the tetraacetylribose is activated, generating an oxocarbenium ion intermediate that is highly electrophilic. The double silylated cytosine, having been rendered more nucleophilic and soluble through the DMF-assisted protection step, attacks this activated sugar moiety with high regioselectivity to form the N-glycosidic bond. The steric environment provided by the macroporous resin support plays a crucial role in stabilizing the transition state and preventing side reactions that typically occur in bulk solution. Furthermore, the polymer matrix prevents the aggregation of catalyst active sites, ensuring uniform catalytic activity throughout the reaction vessel. This controlled environment minimizes the formation of alpha-anomers and other stereoisomeric impurities, which are common pitfalls in nucleoside synthesis. The result is a reaction pathway that favors the formation of the desired beta-configuration with exceptional fidelity, directly contributing to the high purity specifications observed in the final isolated product.
Impurity control is further enhanced by the specific role of the DMF additive during the initial silylation stage. In traditional silylation protocols using hexamethyldisilazane (HMDS), the reaction equilibrium can sometimes favor mono-silylated species, which subsequently participate in the glycosylation to form N3-substituted byproducts or other regioisomers. The introduction of DMF shifts this equilibrium decisively towards the formation of the N4,O-bis-silylated cytosine derivative. This fully protected intermediate is chemically inert at the exocyclic amine position, ensuring that glycosylation occurs exclusively at the N1 position of the pyrimidine ring. By eliminating the possibility of competing nucleophilic sites on the base, the process inherently reduces the complexity of the crude reaction mixture. Consequently, the subsequent deprotection and crystallization steps become far more efficient, requiring fewer solvent exchanges and purification cycles to achieve the target purity of greater than 99.5%, a critical metric for any high-purity pharmaceutical intermediate intended for clinical use.
How to Synthesize 1-Beta-D-Ribofuranosyl Cytosine Efficiently
The execution of this synthesis protocol requires careful attention to stoichiometry and reaction conditions to maximize the benefits of the polymer-supported catalyst system. The process begins with the suspension of cytosine in an anhydrous solvent such as toluene, followed by the addition of HMDS and the critical DMF promoter. Heating this mixture to reflux ensures complete conversion to the bis-silylated intermediate, which is then concentrated before being subjected to the glycosylation step. The choice of solvent for the glycosylation, typically dichloromethane or dichloroethane, is vital for maintaining the swelling of the polymer resin and ensuring optimal mass transfer between the solid catalyst and the liquid reactants. Following the coupling reaction, the solid catalyst is removed by filtration, allowing the filtrate to be washed and concentrated to yield the acetylated nucleoside. The final step involves mild alkaline hydrolysis to remove the acetyl protecting groups, followed by pH adjustment and crystallization to isolate the pure product. For detailed operational parameters and specific reagent quantities, please refer to the standardized guide below.
- Perform double silylation protection of cytosine using hexamethyldisilazane with DMF as an additive to prevent single protection.
- Conduct glycosylation with tetraacetylribose using a recoverable polymer-supported Lewis acid catalyst to form the acetylated intermediate.
- Execute alkaline deprotection followed by separation and purification to isolate high-purity 1-Beta-D-ribofuranosyl cytosine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic advantages beyond mere chemical yield. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of manufacturing by eliminating the need for expensive and complex metal scavenging processes. In traditional routes, removing trace tin or titanium from the final API often requires specialized resins or extensive washing, which adds time, cost, and waste to the production cycle. By utilizing a filterable polymer-supported catalyst, the new method removes this entire unit operation, leading to substantial cost savings in both materials and labor. Additionally, the ability to recover and reuse the catalyst multiple times without significant degradation in performance means that the effective cost of the catalyst per kilogram of product is drastically reduced. This efficiency gain allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy margins, a crucial factor in the highly price-sensitive generic drug market.
- Cost Reduction in Manufacturing: The elimination of hazardous homogeneous catalysts like SnCl4 removes the necessity for costly neutralization and heavy metal removal steps, significantly lowering the operational expenditure associated with waste treatment and safety compliance. The reusability of the polymer-supported catalyst further amortizes the initial investment over numerous batches, driving down the variable cost of goods sold and enhancing overall profitability for large-scale production runs.
- Enhanced Supply Chain Reliability: By simplifying the purification process and reducing the reliance on sensitive, moisture-intolerant reagents, the manufacturing timeline becomes more predictable and robust. The reduced risk of batch failure due to impurity spikes or catalyst deactivation ensures a steady flow of material, mitigating the risk of stockouts for downstream drug manufacturers who depend on a consistent supply of this critical antiviral and antitumor intermediate.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard filtration equipment rather than specialized reactors required for handling corrosive liquids. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, reducing the regulatory burden on manufacturing sites and facilitating smoother audits and inspections, thereby securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. Understanding these details is essential for R&D teams evaluating process transfer and procurement officers assessing supplier capabilities. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers in the pharmaceutical sector.
Q: How does the new method improve impurity control compared to traditional tin tetrachloride catalysis?
A: The novel method utilizes N,N-dimethylformamide (DMF) as an additive to promote complete double silylation of cytosine. This prevents the formation of mono-protected byproducts that typically complicate purification in traditional routes, thereby significantly reducing the overall impurity profile of the final API intermediate.
Q: What are the advantages of using a polymer-supported Lewis acid catalyst in this synthesis?
A: Unlike homogeneous catalysts like SnCl4 or TiCl4 which are corrosive and difficult to remove, the polymer-supported catalyst is heterogeneous. It can be easily separated via simple filtration, reused multiple times without significant loss of activity, and eliminates heavy metal contamination risks in the final product.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It operates under milder conditions, avoids hazardous reagents, simplifies post-processing operations, and has demonstrated high yields (up to 87% for the final product) with purity exceeding 99.5%, making it economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Beta-D-Ribofuranosyl Cytosine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN102911229B can be fully realized in an industrial setting. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1-Beta-D-Ribofuranosyl Cytosine meets the exacting standards required for API synthesis. Our commitment to quality and consistency makes us the preferred choice for multinational pharmaceutical companies seeking a dependable source for high-value nucleoside intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced polymer-supported catalytic technology can enhance your production efficiency and product quality.
