Advanced Chiral Rhodium Complexes for High-Efficiency Asymmetric Hydrogenation in Pharma Manufacturing
Advanced Chiral Rhodium Complexes for High-Efficiency Asymmetric Hydrogenation in Pharma Manufacturing
The landscape of asymmetric synthesis in the pharmaceutical industry is continually evolving, driven by the relentless demand for higher enantiomeric purity and more efficient catalytic systems. A pivotal advancement in this domain is documented in patent CN85103245A, which introduces a novel class of chiral rhodium-diphosphine complexes designed specifically for asymmetric hydrogenation processes. This technology represents a significant departure from conventional catalysts by incorporating perfluorinated carboxylate anions into the coordination sphere of the rhodium center. The general formula of these complexes, depicted as 〔Rh(x)(y)(L0,1,2)〕, showcases a sophisticated architecture where 'y' denotes a chiral diphosphine ligand and 'x' represents a perfluorinated carboxylate group such as trifluoroacetate or perfluorobutyrate. This structural innovation is not merely academic; it addresses critical industrial pain points regarding catalyst loading, reaction time, and ultimately, the optical purity of the resulting pharmaceutical intermediates.
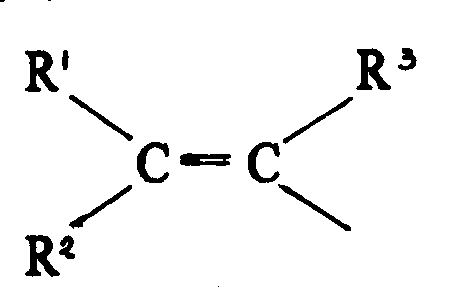
For procurement specialists and supply chain managers evaluating reliable pharmaceutical intermediate suppliers, the implications of this patent are profound. The ability to achieve yields of optically active substances up to 94% or more, as explicitly demonstrated in the patent examples, contrasts sharply with the 80% to 84% yields typical of earlier generation halide-based complexes. This improvement in efficiency translates directly into cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material waste and reducing the burden on downstream purification processes. Furthermore, the patent highlights that these novel complexes allow for a significant reduction in the amount of catalyst required, which is a crucial factor given the high cost of rhodium metal. By optimizing the ligand environment with electron-withdrawing fluorine groups, the catalyst exhibits greater activity, thereby shortening reaction cycles and enhancing overall plant throughput without compromising on the stringent quality standards required for API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of key precursors like ketopantolactone relied heavily on rhodium complexes containing halide ligands such as chlorine, bromine, or iodide. While these traditional catalysts laid the groundwork for chiral synthesis, they suffer from inherent limitations that impact commercial viability. The most significant drawback is the moderate enantioselectivity, which often caps optical purity at around 80% to 84%. In the context of large-scale commercial scale-up of complex pharmaceutical intermediates, this level of impurity necessitates extensive and costly recrystallization or chromatographic purification steps to meet regulatory specifications. Additionally, halide ligands can sometimes lead to catalyst deactivation or slower reaction kinetics, requiring higher catalyst loadings to drive the reaction to completion. This not only increases the direct material cost due to the precious metal content but also complicates the removal of residual rhodium from the final product, a critical quality attribute for any active pharmaceutical ingredient.
The Novel Approach
The innovative strategy outlined in CN85103245A overcomes these barriers by replacing the traditional halide anions with perfluorinated carboxylates. This substitution fundamentally alters the electronic properties of the rhodium center, rendering it more electrophilic and thus more active towards the hydrogenation substrate. The patent data reveals that this novel approach enables the production of R-(-)-Pantolactone with optical purities reaching 93.8% and even higher in optimized examples, a substantial leap forward in stereochemical control. Moreover, the reaction times are notably shortened, with some examples showing complete conversion in as little as 1 to 2 hours under moderate pressure conditions. For a high-purity pharmaceutical intermediate supplier, this means a more robust and predictable manufacturing process. The use of ligands such as BPPM and POPPM in conjunction with these unique anions creates a highly specific chiral pocket that effectively discriminates between enantiotopic faces of the substrate, ensuring consistent product quality batch after batch.
Mechanistic Insights into Perfluorinated Anion-Enhanced Catalysis
To fully appreciate the technical superiority of this system, one must delve into the mechanistic nuances of the catalytic cycle. The chiral diphosphine ligands, such as the pyrrolidine-based structures shown below, coordinate tightly to the rhodium atom, establishing the chiral environment necessary for asymmetric induction. The presence of the perfluorinated carboxylate anion (X) acts as a weakly coordinating counter-ion that is easily displaced by the substrate and hydrogen, facilitating rapid turnover. Unlike strongly coordinating halides which can poison the active site or slow down the oxidative addition of hydrogen, the fluorinated anions maintain a delicate balance of stability and lability. This ensures that the catalytic species remains active throughout the reaction duration, allowing for lower catalyst-to-substrate ratios, often in the range of 0.0001% to 0.01% by weight of rhodium relative to the compound to be hydrotreated.
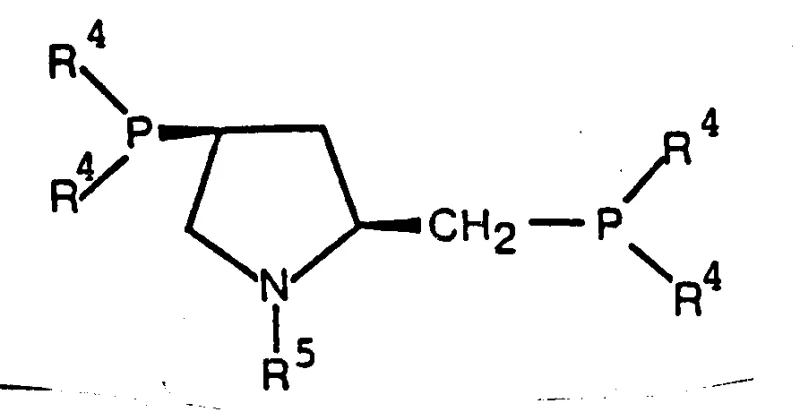
Furthermore, the mechanism involves a precise interplay between the steric bulk of the ligand substituents and the electronic effects of the fluorine atoms. The ligands described, featuring aryl groups like m-tolyl or p-tolyl on the phosphorus atoms, create a rigid conformational lock that directs the approach of the hydrogen molecule to the prochiral double bond of the substrate. The electron-withdrawing nature of the perfluoro-groups on the anion enhances the Lewis acidity of the rhodium center, making it more receptive to the pi-electrons of the olefinic or keto substrate. This synergistic effect results in the high enantioselectivity observed in the reduction of ketopantolactone to pantolactone. From an impurity control perspective, this high selectivity minimizes the formation of the unwanted S-enantiomer, simplifying the impurity profile and reducing the risk of genotoxic impurities or other stereochemical contaminants that could derail regulatory approval processes for the final drug product.
How to Synthesize Chiral Rhodium-Diphosphine Catalyst Efficiently
The preparation of these high-performance catalysts is designed to be operationally straightforward, facilitating easy adoption in existing manufacturing facilities. The synthesis typically involves the reaction of a rhodium precursor, such as bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate, with the chosen chiral diphosphine ligand and a salt containing the perfluorinated anion, such as tetrabutylammonium trifluoroacetate. This process is conducted under strictly inert conditions to prevent oxidation of the sensitive rhodium species. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry and environmental controls required to generate the active orange catalyst solution ready for immediate use in hydrogenation reactors.
- Prepare the reaction environment by establishing an inert atmosphere (oxygen < 1 ppm) using a glove box to prevent catalyst oxidation.
- Combine the rhodium precursor (e.g., bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate) with the chiral diphosphine ligand (e.g., mCH3-POPPM) and the perfluorinated carboxylate salt.
- Stir the mixture in an anhydrous organic solvent like toluene at ambient temperature (approx. 22°C) for 24 hours to form the active orange catalyst solution.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers focused on the bottom line and supply continuity, the adoption of this catalytic technology offers compelling strategic advantages beyond mere technical performance. The shift to perfluorinated anion-based catalysts directly addresses the economic pressures of modern chemical manufacturing by optimizing resource utilization. By achieving higher yields and optical purities in fewer steps, manufacturers can significantly reduce the consumption of expensive starting materials and solvents. This efficiency gain is critical in the volatile market of fine chemicals, where margin compression is a constant threat. Furthermore, the robustness of the catalyst system implies fewer batch failures and less variability, leading to a more predictable supply chain that can reliably meet the demanding delivery schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The implementation of this novel catalyst system drives cost efficiency primarily through the drastic reduction in catalyst loading required to achieve full conversion. Since rhodium is a precious metal with fluctuating and often high market prices, minimizing its usage per kilogram of product results in substantial cost savings. Additionally, the enhanced enantioselectivity reduces the need for costly downstream purification processes such as chiral resolution or repeated recrystallization, which are often the most expensive stages in the production of chiral intermediates. The qualitative improvement in process efficiency means that the overall cost of goods sold (COGS) is lowered, providing a competitive pricing advantage in the marketplace without sacrificing quality margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplified operational profile of this hydrogenation process. The reaction conditions, operating at moderate temperatures between 20°C and 70°C and pressures up to 50 bar, are compatible with standard stainless steel autoclaves found in most multipurpose chemical plants. This compatibility eliminates the need for specialized, high-cost reactor infrastructure, thereby reducing capital expenditure barriers and speeding up technology transfer. Moreover, the shorter reaction times observed with these catalysts increase the asset turnover rate of the manufacturing equipment, allowing for greater production capacity within the same timeframe. This agility ensures that suppliers can respond more rapidly to fluctuations in market demand, securing the continuity of supply for critical vitamin and pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the homogeneous nature of the catalytic system and the use of common organic solvents like toluene and methanol. The high activity of the catalyst allows for smaller reactor volumes to produce the same output, reducing the physical footprint of the manufacturing operation. From an environmental standpoint, the higher atom economy and reduced waste generation align with green chemistry principles. The elimination of halide ligands also mitigates the formation of corrosive byproducts and simplifies waste stream treatment, ensuring compliance with increasingly stringent environmental regulations. This sustainability profile is increasingly important for multinational corporations seeking to minimize their carbon footprint and adhere to responsible sourcing guidelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-diphosphine technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is essential for R&D teams planning process validation and for procurement officers negotiating supply agreements.
Q: What is the primary advantage of using perfluorinated carboxylate anions in rhodium catalysts?
A: The use of perfluorinated carboxylate anions (such as trifluoroacetate) instead of traditional halides significantly increases both the catalytic activity and enantioselectivity, achieving optical purities up to 94% compared to the 80-84% typical of prior art methods.
Q: Can this catalyst system be used for substrates other than ketopantolactone?
A: Yes, the patent demonstrates successful application in the asymmetric hydrogenation of alpha,beta-unsaturated acids, alpha-keto-carboxylic acids, and specifically methyl pyruvate to methyl lactate with high enantiomeric excess.
Q: What are the typical reaction conditions for this hydrogenation process?
A: The process typically operates at temperatures between 20°C and 70°C under hydrogen pressures ranging from 2 to 50 bar, utilizing solvents such as toluene, methanol, or tetrahydrofuran.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Rhodium Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like those described in CN85103245A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical, industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise measurement of enantiomeric excess and residual metal content, guaranteeing that every batch meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this asymmetric hydrogenation technology can be integrated into your supply chain to drive efficiency and quality. By engaging with us, you gain access to a Customized Cost-Saving Analysis that evaluates the specific economic impact of switching to this novel catalyst system for your product portfolio. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering superior chemical solutions that empower your drug development and manufacturing goals.
