Revolutionizing Chiral Intermediate Synthesis with Safe Asymmetric Aldehyde Cyanation Technology
Revolutionizing Chiral Intermediate Synthesis with Safe Asymmetric Aldehyde Cyanation Technology
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for safer, more cost-effective synthetic routes for critical chiral intermediates. Patent CN1468212A presents a groundbreaking methodology for the asymmetric cyanation of aldehydes, addressing long-standing safety and economic challenges associated with traditional cyanohydrin synthesis. This technology enables the production of enantiomerically pure O-acyl cyanohydrins, which serve as versatile building blocks for alpha-hydroxy acids, alpha-amino alcohols, and potent pyrethroid insecticides. By shifting away from hazardous hydrogen cyanide and expensive silylated reagents, this process offers a robust platform for the reliable pharmaceutical intermediates supplier seeking to optimize their supply chain. The innovation lies in the strategic combination of chiral transition metal catalysts with inexpensive alkali metal cyanide sources, fundamentally altering the economic feasibility of producing high-value chiral compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric synthesis of cyanohydrins has been plagued by severe operational hazards and prohibitive costs that limit their industrial applicability. Traditional academic protocols often rely on trimethylsilyl cyanide (TMSCN) as the cyanide source, which, while effective in laboratory settings, is extremely volatile, expensive, and poses significant handling risks on a commercial scale. Furthermore, many established catalytic systems require cryogenic conditions, often necessitating temperatures as low as minus 80 degrees Celsius to achieve acceptable enantiomeric selectivity, which drastically increases energy consumption and equipment complexity. The reliance on such extreme conditions not only inflates operational expenditures but also introduces bottlenecks in batch processing times, making continuous manufacturing difficult to implement. Additionally, the resulting free cyanohydrins are often unstable and prone to autoracemization, leading to product degradation and reduced overall yield during storage and downstream processing. These cumulative factors render many conventional cyanation strategies economically unviable for the mass production of agrochemical intermediates and active pharmaceutical ingredients.
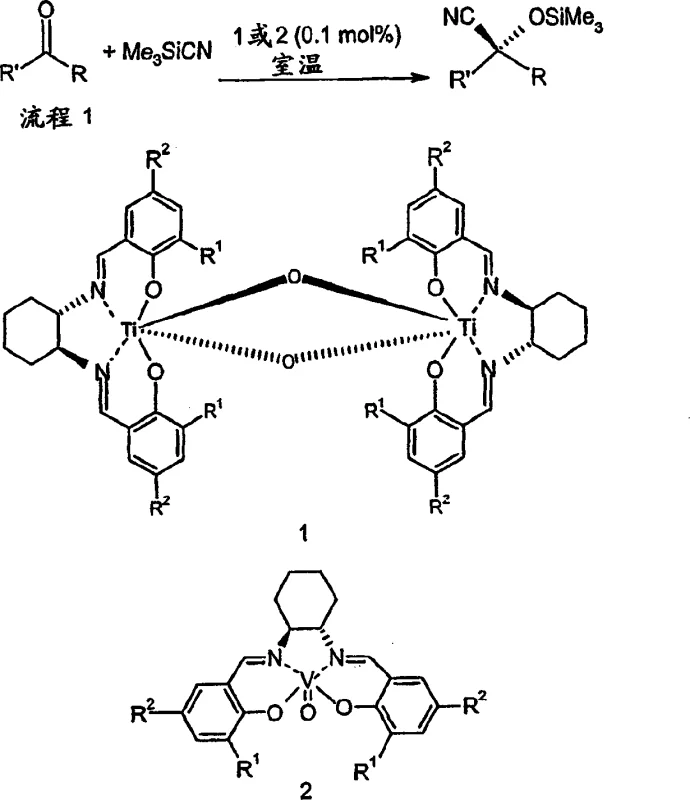
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a heterogeneous system comprising inexpensive alkali metal cyanides, such as potassium cyanide, alongside carboxylic anhydrides. This paradigm shift eliminates the need for silicon-based cyanide sources, thereby achieving substantial cost savings in raw material procurement while simultaneously enhancing workplace safety profiles. The reaction proceeds efficiently at much milder temperatures, typically ranging from minus 40 degrees Celsius to room temperature, which significantly reduces the energy burden on cooling infrastructure and allows for more flexible reactor scheduling. Crucially, the in situ formation of O-acyl cyanohydrins ensures that the chiral center is immediately protected upon formation, preventing racemization and simplifying the isolation of stable, high-purity products. This methodology represents a major leap forward in cost reduction in pharmaceutical intermediates manufacturing, offering a scalable solution that aligns with modern green chemistry principles and regulatory safety standards.
Mechanistic Insights into Chiral Titanium-Catalyzed Cyanation
The core of this technological advancement relies on the precise coordination chemistry between chiral transition metal complexes and the reactants to induce high levels of stereocontrol. The preferred catalysts are chiral titanium or vanadium complexes featuring tetradentate ligands, such as modified salen or salicylimine derivatives, which create a rigid chiral environment around the metal center. During the catalytic cycle, the Lewis acidic metal center activates the carbonyl group of the aldehyde, facilitating the nucleophilic attack by the cyanide ion delivered from the metal cyanide source. The presence of additives like 2,6-lutidine or water plays a critical role in accelerating the reaction kinetics, likely by assisting in the proton transfer steps or stabilizing the transition state geometry. This intricate interplay ensures that the cyanide addition occurs exclusively from one face of the prochiral aldehyde, resulting in high enantiomeric excess values often exceeding 90 percent for a wide range of substrates. The robustness of these catalysts allows them to maintain activity even in heterogeneous mixtures, which is essential for practical industrial applications where solubility issues often arise.
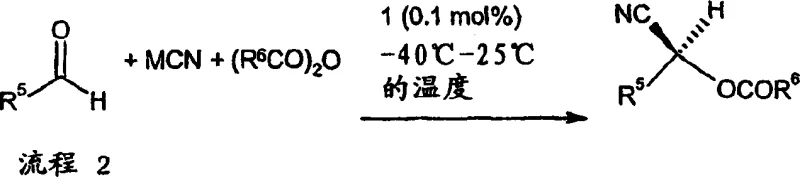
Furthermore, the mechanism inherently controls impurity profiles by rapidly trapping the reactive cyanohydrin intermediate as a stable O-acyl derivative. In traditional aqueous or protic conditions, free cyanohydrins can revert to the starting aldehyde and cyanide, leading to equilibrium issues and potential loss of optical purity over time. By employing carboxylic anhydrides as a co-reactant, the system drives the reaction to completion through the irreversible formation of the ester bond, effectively locking the stereochemistry in place. This kinetic trapping mechanism minimizes side reactions such as aldol condensations or over-reduction, which are common pitfalls in aldehyde chemistry. For R&D teams focused on impurity control, this means a cleaner crude reaction profile that requires less intensive purification downstream, directly translating to higher overall process efficiency. The ability to tune the steric and electronic properties of the ligand substituents further allows for the optimization of selectivity for specific challenging substrates, ensuring consistent quality across different batches.
How to Synthesize Chiral O-Acyl Cyanohydrins Efficiently
Implementing this synthesis route requires careful attention to the order of addition and temperature management to maximize both yield and enantioselectivity. The standard protocol involves preparing a mixture of the chiral catalyst, the alkali metal cyanide source, and the aldehyde in a suitable polar aprotic solvent such as dichloromethane. Once the initial mixture is homogenized, additives are introduced to activate the system before cooling the reaction vessel to the target temperature, typically between minus 30 and minus 40 degrees Celsius for optimal results. The final critical step involves the addition of the carboxylic anhydride, which initiates the trapping phase and drives the conversion to the desired O-acyl product. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare a heterogeneous mixture of alkali metal cyanide (e.g., KCN), chiral titanium catalyst, solvent, and aldehyde.
- Add additives such as 2,6-lutidine or water to accelerate the reaction rate and stabilize the transition state.
- Cool the mixture to the desired temperature (-40°C to room temperature) and add a carboxylic anhydride to trap the cyanohydrin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cyanation technology offers transformative benefits that extend far beyond simple chemical yield improvements. The substitution of high-cost specialty reagents with commodity chemicals fundamentally alters the cost structure of producing chiral building blocks, making previously marginal projects commercially viable. Moreover, the enhanced stability of the O-acyl cyanohydrin products simplifies logistics, allowing for longer storage periods and reduced waste due to decomposition during transit. This reliability is crucial for maintaining continuous supply lines to downstream formulation plants, ensuring that production schedules are not disrupted by raw material instability. The following points detail how this innovation translates into tangible business value for your organization.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the replacement of expensive trimethylsilyl cyanide with readily available alkali metal cyanides like potassium cyanide. This switch drastically lowers the raw material cost per kilogram of product, as commodity cyanide salts are significantly cheaper than their silylated counterparts. Additionally, the elimination of cryogenic requirements reduces utility costs associated with extreme cooling, allowing reactions to run at more energy-efficient temperatures. The simplified workup procedure, which often involves simple filtration to remove inorganic salts, further reduces labor and solvent consumption during the isolation phase. These combined factors result in a leaner manufacturing process with a substantially lower cost of goods sold, enhancing profit margins for high-volume intermediates.
- Enhanced Supply Chain Reliability: Sourcing specialized reagents like TMSCN can be subject to volatility and long lead times, whereas alkali metal cyanides are produced globally in massive quantities for various industries. By relying on these ubiquitous raw materials, manufacturers can mitigate the risk of supply disruptions and secure more favorable long-term contracts with vendors. The robustness of the catalyst system also means that production is less sensitive to minor variations in reagent quality, reducing the frequency of batch failures and rejected shipments. This stability ensures a consistent flow of high-purity intermediates to customers, strengthening the reputation of the manufacturer as a dependable partner in the global supply chain. Reducing lead time for high-purity pharmaceutical intermediates becomes achievable when the bottleneck of specialized reagent procurement is removed.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction mixture is well-suited for large-scale stirred tank reactors, facilitating the commercial scale-up of complex chiral intermediates without requiring exotic equipment. From an environmental perspective, the avoidance of volatile silicon byproducts simplifies waste treatment and reduces the burden on abatement systems, aligning with increasingly stringent global environmental regulations. The process generates fewer hazardous byproducts compared to HCN-based routes, lowering the costs associated with hazardous waste disposal and regulatory compliance reporting. Furthermore, the high atom economy of the anhydride trapping step ensures that reagents are efficiently converted into the final product, minimizing the generation of unreacted starting materials. This sustainable approach not only meets corporate social responsibility goals but also future-proofs the manufacturing site against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this asymmetric cyanation technology. These answers are derived directly from the experimental data and embodiments described in the patent literature to provide accurate guidance for process development teams. Understanding these nuances is essential for successfully transferring this chemistry from the laboratory bench to pilot and commercial production scales. We encourage technical stakeholders to review these details closely to assess the fit for their specific application requirements.
Q: What are the safety advantages of this cyanation method over traditional HCN routes?
A: This method eliminates the need for highly toxic hydrogen cyanide (HCN) gas and volatile trimethylsilyl cyanide (TMSCN), utilizing safer solid alkali metal cyanides instead.
Q: Can this process be scaled for commercial production of agrochemical intermediates?
A: Yes, the use of inexpensive reagents like potassium cyanide and acetic anhydride, combined with moderate temperature requirements, makes it highly suitable for large-scale manufacturing.
Q: What types of aldehydes are compatible with this chiral catalyst system?
A: The system demonstrates broad substrate scope, effectively processing aromatic, heteroaromatic, and aliphatic aldehydes with high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cyanohydrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this cyanation method are seamlessly translated into robust manufacturing operations. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art chiral analysis capabilities to guarantee the enantiomeric excess of every batch we produce. Our commitment to quality assurance means that clients receive intermediates that consistently meet the demanding requirements of regulated pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can optimize your supply chain for efficiency and reliability. Let us be your partner in delivering high-performance chiral solutions that drive your business forward.
