Scalable Copper-Catalyzed Alkyne Coupling for Commercial Pharmaceutical Intermediate Production
The chemical manufacturing landscape is constantly evolving, driven by the need for more sustainable and cost-effective synthetic routes. Patent CN1288117C introduces a significant advancement in the field of organic synthesis, specifically targeting the coupling reaction of terminal alkynes and aryl halides. This technology represents a pivotal shift away from traditional palladium-dependent methodologies, offering a robust alternative that utilizes copper iodide (CuI) in conjunction with amino acid additives. For R&D directors and process chemists, this patent data suggests a pathway to synthesize complex aryl alkyne structures under remarkably mild conditions. The core innovation lies in the ability to achieve high conversion rates at temperatures averaging 20°C lower than comparable literature methods, which translates directly into energy savings and improved safety profiles during scale-up. By leveraging inexpensive and air-stable catalysts, this method addresses critical pain points in the production of fine chemical intermediates, ensuring that high-purity targets can be met without the prohibitive costs associated with precious metal catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl alkynes has been dominated by the Sonogashira coupling reaction, which typically relies on a dual catalytic system involving both palladium and copper. While effective, this conventional approach presents substantial drawbacks for large-scale industrial applications. The primary concern is the reliance on palladium, a precious metal that is not only expensive but also subject to volatile market pricing and supply chain constraints. Furthermore, the removal of trace palladium residues from the final active pharmaceutical ingredient (API) or intermediate is a rigorous and costly process, often requiring specialized scavengers or multiple purification steps to meet stringent regulatory limits. Additionally, traditional protocols often necessitate harsh reaction conditions, including elevated temperatures and the use of sensitive phosphine ligands that degrade upon exposure to air, complicating the operational workflow and increasing the risk of batch-to-batch variability.
The Novel Approach
In contrast, the methodology described in the patent data offers a streamlined solution by eliminating palladium entirely. This novel approach utilizes a copper-only catalytic system augmented by simple amino acid ligands, such as N,N-dimethylglycine hydrochloride. As illustrated in the reaction scheme below, this system effectively couples terminal alkynes with aryl halides (iodides or bromides) to form the desired carbon-carbon triple bond linkage. The use of amino acids as ligands is a game-changer; they are inexpensive, commercially available in bulk, and stable in air, removing the need for inert handling during the setup phase. This shift not only drastically simplifies the raw material procurement process but also enhances the overall robustness of the synthesis. The reaction proceeds efficiently in polar aprotic solvents like DMF or DMSO, tolerating a wide range of functional groups including esters, nitro groups, and halogens, which is crucial for the modular synthesis of diverse pharmaceutical scaffolds.
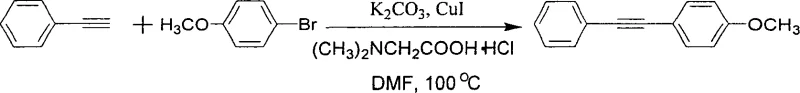
Mechanistic Insights into CuI-Catalyzed Alkyne Coupling
From a mechanistic perspective, the success of this reaction hinges on the synergistic interaction between the copper catalyst and the amino acid ligand. The amino acid acts as a bidentate or monodentate ligand that coordinates with the copper center, stabilizing the active catalytic species against aggregation and decomposition. This stabilization is critical for maintaining catalytic turnover over the extended reaction periods typically required for less reactive aryl bromides. The proposed mechanism likely involves the formation of a copper-acetylide intermediate, followed by oxidative addition of the aryl halide and subsequent reductive elimination to release the coupled product. The presence of the base, typically potassium carbonate (K2CO3), is essential for deprotonating the terminal alkyne to generate the nucleophilic acetylide species. Importantly, the mild nature of this catalytic cycle minimizes side reactions such as homocoupling of the alkyne (Glaser coupling), which is a common impurity profile challenge in copper-mediated reactions. By carefully tuning the ligand-to-copper ratio, typically between 2:1 and 4:1, the process ensures high selectivity for the cross-coupled product.
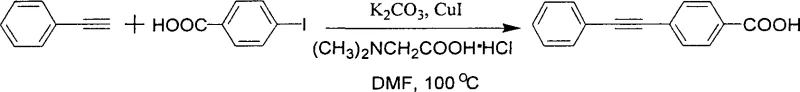
Impurity control is another area where this mechanism offers distinct advantages. The absence of palladium eliminates the risk of Pd-related impurities, which are notoriously difficult to purge. Furthermore, the reaction conditions are sufficiently mild to preserve sensitive functional groups on the aromatic ring. For instance, the patent data demonstrates successful coupling with substrates containing nitro groups and carboxylic acids without the need for extensive protecting group strategies. This chemoselectivity reduces the total number of synthetic steps required to reach the target molecule, thereby improving the overall atom economy and reducing waste generation. The ability to run the reaction at 100°C, rather than the higher temperatures often demanded by ligand-free copper systems, further protects thermally labile intermediates from degradation, ensuring a cleaner crude reaction profile and simplifying downstream purification efforts.
How to Synthesize Phenylacetylene Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The general procedure involves charging a reaction vessel with the terminal alkyne, aryl halide, base, catalyst, and ligand in the chosen solvent. The mixture is then heated under an inert atmosphere to prevent oxidation of the copper species, although the ligand itself provides significant air stability. Detailed standard operating procedures regarding stoichiometry, addition order, and workup protocols are critical for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent examples, ensuring that technical teams can replicate the high yields reported in the intellectual property documentation.
- Combine terminal alkyne, aryl halide, K2CO3 base, CuI catalyst, and amino acid ligand in DMF solvent under inert atmosphere.
- Heat the reaction mixture to 100°C in an oil bath and maintain stirring for approximately 24 hours to ensure complete conversion.
- Cool the mixture, perform aqueous workup with ethyl acetate extraction, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers tangible strategic benefits beyond mere technical feasibility. The most immediate impact is seen in the reduction of raw material costs. By substituting expensive palladium catalysts and specialized phosphine ligands with commodity chemicals like copper iodide and amino acid salts, the direct material cost of the synthesis is significantly lowered. This cost structure is more resilient to market fluctuations, as copper and amino acids are produced in massive volumes for other industries, ensuring a stable and reliable supply chain. Moreover, the simplified purification process resulting from the absence of heavy metal contaminants reduces the consumption of specialty scavengers and solvents, contributing to further operational expenditure savings. These factors combined create a more predictable cost model for long-term production contracts.
- Cost Reduction in Manufacturing: The elimination of palladium from the catalytic cycle removes one of the most significant cost drivers in traditional cross-coupling reactions. Palladium prices are historically volatile and can spike due to geopolitical factors or mining constraints. By relying on copper, which is orders of magnitude cheaper and more abundant, manufacturers can secure a substantial reduction in catalyst-related expenses. Additionally, the amino acid ligands used are bulk chemicals with low price points, unlike custom-synthesized phosphines. This shift allows for a drastic simplification of the bill of materials, leading to overall lower production costs per kilogram of the final intermediate without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the pharmaceutical industry, where disruptions can halt production lines. The reagents required for this process—CuI, K2CO3, DMF, and simple amino acids—are widely available from multiple global suppliers. This multi-sourcing capability mitigates the risk of single-source dependency often associated with proprietary ligands or precious metals. Furthermore, the air stability of the catalyst system means that storage and handling requirements are less stringent, reducing the risk of material degradation during transit or warehousing. This robustness ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental and safety challenges. This methodology addresses these concerns by operating at moderate temperatures and avoiding toxic heavy metals. The reduction in reaction temperature by approximately 20°C compared to conventional methods lowers energy consumption for heating and cooling, aligning with green chemistry principles. From a waste management perspective, the absence of palladium simplifies effluent treatment, as there is no need for complex heavy metal recovery or disposal protocols. This ease of waste handling facilitates smoother regulatory approvals for commercial scale-up, enabling faster time-to-market for new drug candidates relying on these aryl alkyne motifs.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this coupling technology is essential for stakeholders evaluating its integration into existing manufacturing workflows. The following questions address common inquiries regarding the operational parameters, substrate compatibility, and economic implications of the process. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Whether you are assessing the feasibility for a specific target molecule or evaluating the broader economic impact, these insights clarify the capabilities and limitations of the copper-amino acid catalytic system.
Q: What are the advantages of using amino acid ligands in this coupling reaction?
A: Amino acid ligands, such as N,N-dimethylglycine, stabilize the copper catalyst, allowing the reaction to proceed under milder conditions (approx. 20°C lower than traditional methods) and are significantly cheaper and more air-stable than phosphine ligands.
Q: What is the optimal temperature range for this Cu-catalyzed coupling?
A: The patent recommends a temperature range of 60°C to 110°C, with 100°C being the further recommended optimal condition for balancing reaction rate and energy consumption.
Q: Does this method support diverse substrate scopes?
A: Yes, the method is highly versatile, successfully coupling various aryl iodides and bromides containing functional groups like nitro, carboxyl, methoxy, and halogens with terminal alkynes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylacetylene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global fine chemicals market. Our technical team has extensively analyzed the potential of copper-catalyzed coupling reactions and possesses the expertise to translate this patent knowledge into commercial reality. We offer comprehensive CDMO services tailored to the production of complex aryl alkyne intermediates, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and state-of-the-art reactors capable of maintaining the precise temperature and atmospheric controls required for these sensitive transformations, ensuring stringent purity specifications are met for every batch delivered.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective synthesis route can be applied to your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this palladium-free methodology for your supply chain. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our manufactured intermediates against your internal standards. Let us help you optimize your production costs and secure a reliable supply of high-quality chemical building blocks for your next generation of pharmaceutical products.
