Advanced Synthesis of 4-Indole Thiosemicarbazide Derivatives for Commercial Anti-Fibrosis Applications
Advanced Synthesis of 4-Indole Thiosemicarbazide Derivatives for Commercial Anti-Fibrosis Applications
The pharmaceutical industry is constantly seeking novel molecular entities to address unmet medical needs, particularly in the realm of chronic liver diseases where current therapeutic options remain limited. Patent CN112679409A introduces a significant breakthrough with the disclosure of 4-indole-substituted thiosemicarbazide derivatives, a new class of compounds exhibiting potent anti-hepatic fibrosis activity. These molecules function as agonists for the nuclear receptor Nur77, a critical checkpoint in controlling TGF-β signaling and fibrogenesis. The structural versatility of these derivatives, defined by Formula (I) where R can be hydrogen, halogens, or various alkyl and alkoxy groups, allows for extensive structure-activity relationship studies to optimize therapeutic efficacy. As a leading entity in fine chemical manufacturing, understanding the synthetic accessibility and biological potential of such scaffolds is paramount for developing reliable pharmaceutical intermediate supplier capabilities.
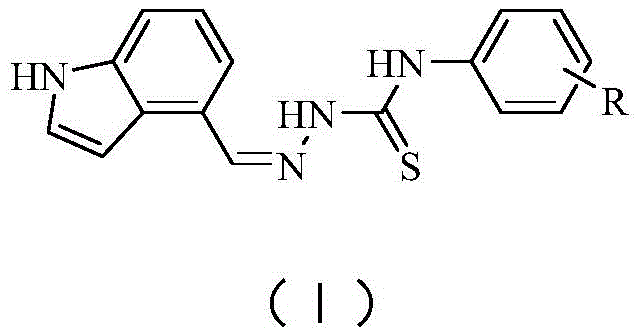
The significance of this technology extends beyond mere biological activity; it represents a robust platform for cost reduction in API manufacturing. Liver fibrosis, a precursor to cirrhosis, lacks approved pharmacological treatments, making the discovery of Nur77 agonists a high-value target for drug developers. The patented synthesis route offers a streamlined approach that avoids the pitfalls of traditional multi-step organic syntheses, which often suffer from low overall yields and complex purification requirements. By leveraging simple condensation reactions in common solvents like ethanol, this methodology aligns perfectly with the principles of green chemistry and industrial scalability. For procurement managers and supply chain heads, this translates to a reduced risk profile and enhanced continuity of supply for critical drug development programs focused on hepatic disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic compounds intended for biological screening has been plagued by inefficiencies that hinder commercial viability. Conventional routes often rely on harsh reaction conditions, toxic solvents, or expensive transition metal catalysts that necessitate rigorous removal steps to meet stringent purity specifications. In the context of thiosemicarbazide derivatives, traditional methods might involve multiple protection and deprotection steps, leading to significant material loss and increased waste generation. Furthermore, the use of non-polar or chlorinated solvents can complicate downstream processing and increase environmental compliance costs. These factors collectively contribute to extended lead times for high-purity pharmaceutical intermediates, creating bottlenecks in the drug discovery pipeline. For R&D directors, the presence of difficult-to-remove impurities or residual metals can invalidate preclinical data, forcing costly re-synthesis and delaying critical milestones.
The Novel Approach
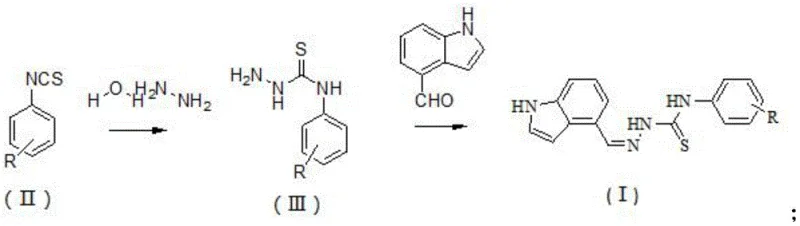
In stark contrast, the methodology described in the patent utilizes a direct and atom-economical two-step condensation strategy that dramatically simplifies the production landscape. The process begins with the reaction of substituted isothiocyanates and hydrazine hydrate to form a thiosemicarbazide intermediate, followed by a second condensation with 4-indole carbaldehyde. This approach eliminates the need for exotic reagents or complex catalytic systems, relying instead on thermodynamic driving forces to achieve high conversion rates. The use of ethanol as a universal solvent for both steps not only reduces solvent inventory costs but also facilitates easy product isolation through crystallization or filtration. This novel approach ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal capital expenditure on specialized equipment. The simplicity of the workflow allows for rapid iteration of R groups, accelerating the optimization of biological potency while maintaining a lean manufacturing footprint.
Mechanistic Insights into Condensation-Based Synthesis
The chemical elegance of this synthesis lies in its reliance on nucleophilic addition-elimination mechanisms that are highly predictable and controllable. In the first step, the nucleophilic nitrogen of hydrazine hydrate attacks the electrophilic carbon of the isothiocyanate group. This reaction proceeds smoothly at moderate temperatures ranging from 70°C to 80°C, forming the stable thiosemicarbazide backbone. The choice of hydrazine hydrate in slight molar excess ensures complete consumption of the isothiocyanate, minimizing the presence of unreacted starting materials in the crude mixture. This mechanistic clarity is crucial for R&D teams aiming to replicate results across different batches, as it reduces the sensitivity of the reaction to minor fluctuations in mixing or heating rates. The resulting intermediate serves as a versatile building block, retaining reactive amine functionality for the subsequent coupling step.
The second stage involves the condensation of this intermediate with 4-indole carbaldehyde, a reaction that forms the critical imine or hydrazone linkage connecting the indole moiety to the thiosemicarbazide core. Conducted under reflux conditions in ethanol at approximately 78°C, this step leverages the removal of water to drive the equilibrium towards product formation. The indole ring, being electron-rich, could potentially participate in side reactions under acidic or oxidative conditions, but the neutral to slightly basic environment of this protocol preserves the integrity of the heterocycle. Impurity control is inherently managed by the precipitation of the product; as the conjugated system forms, the molecule's solubility in ethanol decreases, causing it to crash out of solution. This self-purifying mechanism significantly reduces the burden on downstream purification units, ensuring that the final active pharmaceutical ingredient candidates meet high standards of chemical purity without extensive chromatographic intervention.
How to Synthesize 4-Indole-Substituted Thiosemicarbazide Efficiently
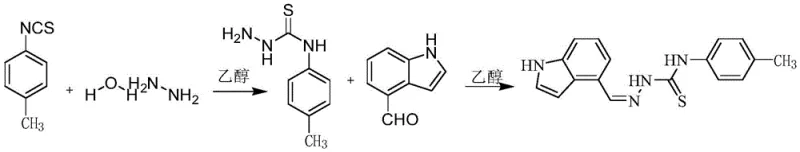
Implementing this synthesis in a pilot or production plant requires careful attention to stoichiometry and thermal management to maximize yield and safety. The process is designed to be operationally simple, making it accessible for contract development and manufacturing organizations (CDMOs) looking to expand their portfolio of hepatic disease therapeutics. The initial condensation requires precise temperature control to prevent the decomposition of hydrazine, while the second step benefits from efficient reflux condensers to maintain solvent volume. Detailed standard operating procedures would specify the exact addition rates and stirring speeds to ensure homogeneity, particularly as the solid product begins to form. For those interested in the specific operational parameters, the detailed standardized synthesis steps are outlined in the guide below, reflecting the optimized conditions found in the patent examples.
- Condense substituted phenyl isothiocyanate with hydrazine hydrate in ethanol at 70-80°C to form the thiosemicarbazide intermediate.
- React the resulting intermediate with 4-indole carbaldehyde in ethanol under reflux conditions at 78°C.
- Isolate the final product via filtration and washing with ethanol to obtain high-purity 4-indole-substituted thiosemicarbazide derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this technology offers compelling advantages that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts and hazardous solvents removes significant cost centers associated with raw material procurement and waste disposal. Traditional processes often require expensive scavengers to remove trace metals, a step that is entirely unnecessary here. This inherent simplicity translates to substantial cost savings in manufacturing, allowing for more competitive pricing of the final intermediates. Moreover, the reliance on commodity chemicals like ethanol and hydrazine hydrate insulates the supply chain from the volatility often seen with specialized reagents. Procurement managers can secure long-term contracts for these bulk materials with greater confidence, ensuring that production schedules remain uninterrupted even during global supply disruptions.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing a solvent system that serves dual purposes as both reaction medium and crystallization agent. By avoiding the need for complex workup procedures such as liquid-liquid extraction with multiple organic phases, the consumption of utilities and labor is drastically reduced. The high yield reported in the patent examples indicates that raw material utilization is optimized, meaning less waste is generated per kilogram of product. This efficiency lowers the cost of goods sold (COGS) and improves the margin profile for downstream drug manufacturers. Additionally, the absence of chromatography steps reduces the consumption of silica gel and eluents, further contributing to a leaner cost structure.
- Enhanced Supply Chain Reliability: The starting materials, specifically substituted isothiocyanates and indole aldehydes, are widely produced by the global chemical industry, ensuring a robust and diversified supply base. This availability reduces the risk of single-source dependency, a critical factor for supply chain heads managing risk portfolios. The synthetic route is short, consisting of only two main chemical transformations, which minimizes the number of potential failure points in the production line. Shorter synthesis times mean faster turnaround from order to delivery, enabling pharmaceutical partners to accelerate their preclinical and clinical timelines. The stability of the intermediates also allows for flexible production scheduling, where the thiosemicarbazide precursor can be stocked and reacted on demand.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the lack of exothermic hazards and the use of low-boiling, recyclable solvents. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. From an environmental standpoint, the use of ethanol aligns with green chemistry initiatives, reducing the facility's volatile organic compound (VOC) emissions profile. The solid waste generated is primarily organic in nature and can often be managed through standard incineration or recovery protocols. This environmental compatibility simplifies the permitting process for new manufacturing lines and supports the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their pipelines, we have compiled answers to common inquiries regarding the synthesis and application of these derivatives. The following information is derived directly from the technical disclosures and experimental data provided in the patent documentation. Understanding these nuances is essential for assessing the feasibility of integrating this chemistry into existing manufacturing workflows. We encourage potential partners to review these details closely to appreciate the depth of innovation involved.
Q: What is the primary therapeutic target of these 4-indole thiosemicarbazide derivatives?
A: These compounds act as Nur77 agonists. By inducing Nur77 expression, they rebalance TGF-β signaling pathways, effectively inhibiting hepatic stellate cell activation and reducing liver fibrosis progression.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, the synthesis utilizes readily accessible raw materials such as substituted phenyl isothiocyanates, hydrazine hydrate, and 4-indole carbaldehyde, ensuring a stable and reliable supply chain for large-scale production.
Q: Does this process require complex purification methods like column chromatography?
A: No, the process is designed for industrial simplicity. The products typically precipitate out of the ethanol solvent and can be isolated through simple filtration and washing, eliminating the need for expensive and time-consuming chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Indole-Substituted Thiosemicarbazide Derivative Supplier
The development of anti-fibrosis agents represents a frontier in modern medicine, and having a manufacturing partner capable of navigating the complexities of heterocyclic chemistry is essential. NINGBO INNO PHARMCHEM stands ready to support your drug discovery efforts with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs that ensure every batch meets stringent purity specifications required for IND-enabling studies. We understand that consistency is key in pharmaceutical development, and our process engineers are dedicated to optimizing every parameter to deliver high-quality intermediates reliably.
We invite you to collaborate with us to unlock the full potential of these Nur77 agonists for your therapeutic programs. Our team can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient synthesis route can lower your overall project costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your journey from benchtop discovery to clinical success with our superior manufacturing capabilities and commitment to excellence.
