Advanced Synthesis of Chiral Amino Alcohols for High-Value Pharmaceutical Intermediates
Advanced Synthesis of Chiral Amino Alcohols for High-Value Pharmaceutical Intermediates
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant innovations in asymmetric synthesis, particularly for complex molecules like tachykinin antagonists. Patent CN1100410A introduces a groundbreaking methodology for the preparation of (+)-2-(3,4-dichlorophenyl)-4-hydroxybutylamine, a critical building block designated as Compound (I). This specific amino alcohol serves as a pivotal precursor in the construction of potent neurokinin receptor antagonists, which are essential for treating various inflammatory and pain-related disorders. The disclosed process represents a substantial leap forward from conventional resolution techniques, offering a pathway that combines high stereochemical control with operational simplicity. By leveraging a unique second-order asymmetric transformation followed by a robust enantioconservative reduction, this technology addresses the longstanding challenges of yield loss and optical purity often encountered in the manufacture of chiral arylalkylamines.
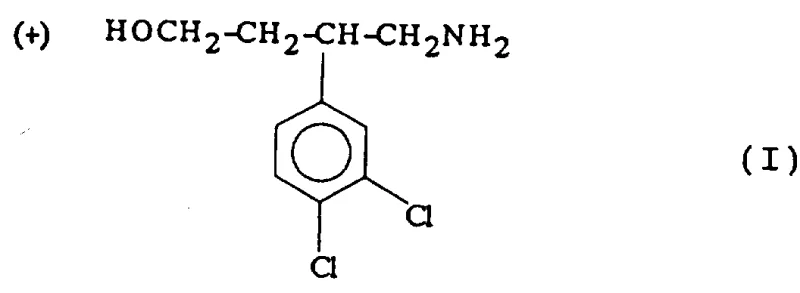
For procurement specialists and supply chain managers evaluating reliable pharmaceutical intermediate suppliers, understanding the underlying chemistry is crucial for assessing long-term viability. The method described in CN1100410A does not merely offer a laboratory curiosity; it provides a scalable industrial solution that significantly mitigates the risks associated with raw material volatility and processing inefficiencies. The ability to produce this key intermediate with consistent quality and improved throughput directly translates to cost reduction in API manufacturing for downstream clients. As we delve deeper into the technical specifics, it becomes evident that this process is designed to meet the rigorous demands of modern Good Manufacturing Practice (GMP) environments while maintaining the economic feasibility required for competitive market positioning.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure amino alcohols similar to Compound (I) relied heavily on the resolution of racemic mixtures using classical agents such as tartaric acid. In these traditional workflows, a racemic amine or acid is reacted with a chiral resolving agent to form diastereomeric salts, which are then separated via fractional crystallization. However, this approach suffers from an inherent theoretical ceiling: the maximum yield of the desired single enantiomer is strictly limited to 50% of the starting racemate. The remaining 50%, consisting of the unwanted enantiomer, is often discarded or subjected to costly and inefficient racemization-recycling loops that add significant complexity and waste to the process. Furthermore, classical resolution often requires multiple recrystallization steps to achieve acceptable enantiomeric excess, leading to substantial material losses and extended production cycles. For a commercial operation, these inefficiencies manifest as higher unit costs, increased solvent consumption, and a larger environmental footprint, all of which are critical pain points for supply chain heads focused on sustainability and margin optimization.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN1100410A employs a sophisticated strategy centered on a second-order asymmetric transformation using D-(-)-N-methylglucamine. Instead of merely separating existing enantiomers, this process dynamically converts the unwanted isomer into the desired form during the crystallization phase. By treating the racemic 3-cyano-3-(3,4-dichlorophenyl)propionic acid with D-(-)-N-methylglucamine, the system drives the equilibrium towards the formation of the less soluble salt of the levorotatory acid. This phenomenon allows for theoretical yields that far exceed the 50% barrier, with experimental data indicating yields based on the levorotatory enantiomer reaching as high as 190%. This breakthrough effectively utilizes the entire mass of the starting racemic material, drastically reducing waste and improving the overall atom economy. Additionally, the subsequent reduction step utilizes borane complexes to simultaneously reduce both the nitrile and carboxylic acid functionalities in a single operation, streamlining the synthesis and minimizing the number of isolation steps required to reach the final amino alcohol target.
Mechanistic Insights into Second-Order Asymmetric Transformation and Borane Reduction
The core of this innovative synthesis lies in the precise manipulation of stereochemistry during the resolution of the intermediate acid. The process begins with the alkylation of 3,4-dichlorophenylacetonitrile (Compound II) using an alkali metal haloacetate, such as sodium chloroacetate, in the presence of a strong base like sodium amide or sodium tert-butoxide. This reaction generates the racemic 3-cyano-3-(3,4-dichlorophenyl)propionic acid (Compound III), which serves as the substrate for the chiral resolution.
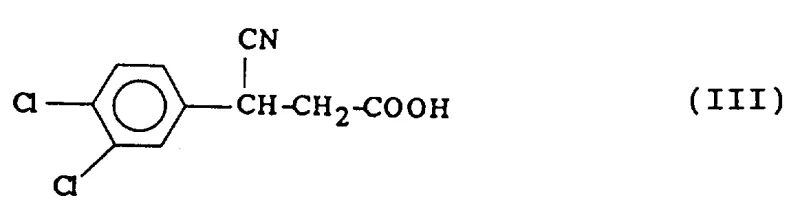
Upon treatment with D-(-)-N-methylglucamine, a second-order asymmetric transformation occurs. Unlike simple preferential crystallization, this mechanism involves the continuous equilibration of the enantiomers in solution. As the salt of the desired (-)-enantiomer crystallizes out of the solution, the equilibrium shifts to replenish it, effectively converting the (+)-enantiomer into the (-)-form until the entire batch is consumed. The resulting salt is then treated with a strong acid, such as hydrochloric acid, to liberate the free optically pure (-)-3-cyano-3-(3,4-dichlorophenyl)propionic acid (Compound IV). This intermediate is isolated with exceptional optical purity, setting the stage for the final transformation.
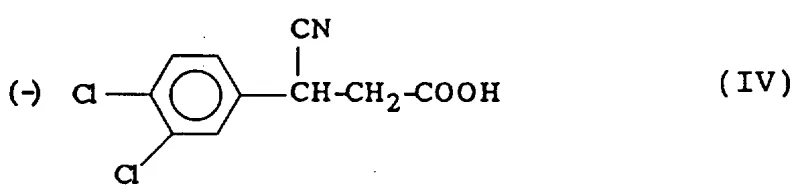
The final step involves an enantioconservative reduction using borane, typically supplied as a complex with tetrahydrofuran (THF) or dimethyl sulfide. Borane is a powerful reducing agent capable of converting carboxylic acids to primary alcohols and nitriles to primary amines. In this specific context, the reaction conditions are carefully controlled at room temperature to ensure that the chiral center at the benzylic position remains intact. The mechanism proceeds without racemization, preserving the 99% enantiomeric purity achieved in the previous step. The use of borane complexes, as depicted in the patent figures, offers a safer and more manageable alternative to handling gaseous diborane, further enhancing the operational safety profile of the process.
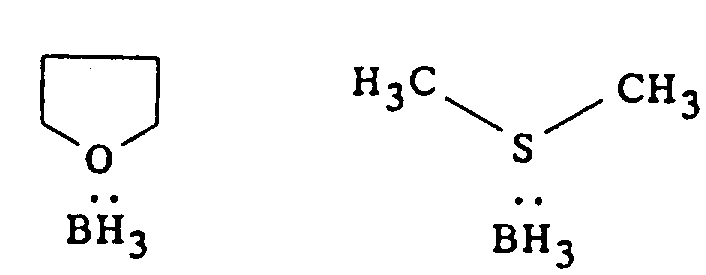
Impurity control is inherently built into this mechanistic pathway. The crystallization of the N-methylglucamine salt acts as a powerful purification step, excluding structurally related impurities and unreacted starting materials from the crystal lattice. Furthermore, the workup procedures involving acid-base extractions effectively remove the resolving agent and inorganic byproducts. The result is a final product that meets stringent purity specifications required for pharmaceutical applications, minimizing the burden on downstream purification processes.
How to Synthesize (+)-2-(3,4-dichlorophenyl)-4-hydroxybutylamine Efficiently
The synthesis of this high-value chiral intermediate is streamlined into four distinct operational phases that balance chemical efficiency with practical manufacturability. The process initiates with the formation of the carbon-carbon bond to create the racemic acid backbone, followed by the critical chiral resolution that defines the stereochemical outcome. The subsequent liberation of the free acid and the final dual-reduction complete the transformation. Each step has been optimized in the patent examples to demonstrate robustness across different scales, utilizing common solvents like ethanol, dichloromethane, and THF. For detailed operational parameters, stoichiometry, and specific workup instructions tailored to your facility's capabilities, please refer to the standardized synthesis guide below.
- React 3,4-dichlorophenylacetonitrile with an alkali metal haloacetate in the presence of a strong base at temperatures between -40°C and 25°C to form racemic 3-cyano-3-(3,4-dichlorophenyl)propionic acid.
- Treat the resulting racemic acid with D-(-)-N-methylglucamine to induce a second-order asymmetric transformation, crystallizing the salt of the desired levorotatory enantiomer.
- Liberate the free optically pure (-)-3-cyano-3-(3,4-dichlorophenyl)propionic acid from its salt using a strong acid followed by solvent extraction.
- Perform an enantioconservative reduction of the purified acid using borane (e.g., BH3-THF complex) at room temperature to simultaneously reduce the nitrile and carboxylic acid groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1100410A offers compelling strategic advantages that extend beyond mere chemical elegance. The shift from traditional resolution methods to this dynamic asymmetric transformation fundamentally alters the cost structure of producing chiral amino alcohols. By eliminating the 50% yield cap inherent in classical methods, the process significantly reduces the amount of raw materials required per kilogram of finished product. This improvement in atom economy directly correlates to lower variable costs and reduced waste disposal expenses, contributing to substantial cost savings in fine chemical manufacturing. Moreover, the use of D-(-)-N-methylglucamine, an inexpensive industrial raw material derived from glucose, replaces more costly or less efficient resolving agents, further enhancing the economic viability of the route.
- Cost Reduction in Manufacturing: The elimination of the theoretical yield limit means that manufacturers can produce nearly double the amount of product from the same quantity of starting racemate compared to traditional tartrate resolution. This efficiency gain removes the need for complex racemization and recycling loops, which often require additional reactors, catalysts, and energy inputs. Consequently, the overall production cost per unit is drastically simplified and lowered, allowing for more competitive pricing strategies in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically 3,4-dichlorophenylacetonitrile and sodium chloroacetate, are commodity chemicals with stable and robust supply chains. Unlike specialized chiral catalysts that may suffer from supply bottlenecks or long lead times, the reagents used here are readily available from multiple global sources. This diversity in sourcing options mitigates the risk of supply disruptions, ensuring consistent production schedules and reliable delivery timelines for downstream API manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, avoiding extreme conditions such as deep cryogenic temperatures for the resolution step, which can be performed between 0°C and the solvent boiling point. The workup involves standard liquid-liquid extractions and crystallizations, unit operations that are easily transferred from pilot plants to multi-ton commercial reactors. Furthermore, the high efficiency of the reaction reduces the volume of solvent waste and chemical byproducts generated, aligning with increasingly stringent environmental regulations and corporate sustainability goals regarding green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of using D-(-)-N-methylglucamine for resolution?
A: Unlike traditional tartrate resolution which is limited to a maximum 50% theoretical yield of the desired enantiomer, the use of D-(-)-N-methylglucamine facilitates a second-order asymmetric transformation. This dynamic process allows for the conversion of the unwanted enantiomer into the desired one during crystallization, potentially achieving yields up to 190% based on the initial levorotatory content, effectively utilizing the entire racemic mixture.
Q: How is high enantiomeric purity maintained during the reduction step?
A: The process utilizes an enantioconservative reduction using borane complexes such as BH3-THF or BH3-dimethyl sulfide. These reagents reduce both the cyano group to a primary amine and the carboxylic acid to a primary alcohol under mild conditions (room temperature), preserving the stereochemical integrity of the chiral center established in the previous resolution step, resulting in products with 99% enantiomeric purity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is highly scalable. It avoids cryogenic conditions for the resolution step (which can be performed between 0°C and the solvent boiling point) and uses inexpensive, industrially available resolving agents like N-methylglucamine. The workup procedures involve standard acid-base extractions and crystallizations, making it robust for manufacturing environments ranging from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (+)-2-(3,4-dichlorophenyl)-4-hydroxybutylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand the critical nature of chiral purity in drug development, which is why our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of (+)-2-(3,4-dichlorophenyl)-4-hydroxybutylamine we produce.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact us today to request specific COA data, route feasibility assessments, and samples to evaluate how our high-purity intermediates can accelerate your drug development timeline.
