Revolutionizing Ephedrine Intermediate Production via Advanced Diastereomeric Salt Resolution
Introduction to Advanced Chiral Resolution Technologies
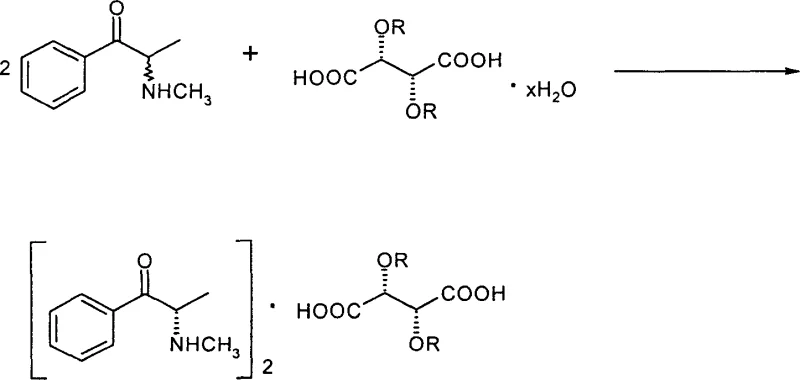
The pharmaceutical industry continuously seeks robust methodologies for the production of high-value chiral intermediates, particularly those serving as precursors for critical active pharmaceutical ingredients (APIs) such as ephedrine. A significant technological breakthrough in this domain is documented in Chinese Patent CN1293039C, which details a highly efficient preparation method for [(S)-(-)-alpha-methylamino phenylketone]2.(2R,3R)-tartaric acid derivatives. This specific intermediate is pivotal for the synthesis of (1R, 2S)-(-)-ephedrine, a compound with widespread therapeutic applications. The patent introduces a novel resolution strategy that overcomes the thermodynamic and kinetic limitations inherent in traditional separation techniques. By leveraging specific (2R,3R)-tartaric acid derivatives in optimized mixed solvent systems, the process achieves not only high diastereomeric purity but also optical yields that remarkably exceed theoretical expectations through in-situ asymmetric transformation. This report analyzes the technical merits and commercial implications of this innovation for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of alpha-methylaminopropiophenone has relied heavily on resolving agents such as (1R, 3S)-(+)-camphoric acid or (1R)-(-)-camphorsulfonic acid, as evidenced by earlier patents like CN1298867A and CN1267664A. These conventional processes suffer from significant operational inefficiencies and chemical instability issues. For instance, the salts formed between the ketone and camphoric acids exhibit high sensitivity to temperature; heating above 50°C often triggers deleterious side reactions that drastically reduce the optical yield of the desired enantiomer. Furthermore, these legacy methods typically require complex thermal cycling, involving heating to dissolve the crude salt followed by careful cooling to induce crystallization. This energy-intensive approach not only complicates process control but also limits the scalability of the operation. Additionally, the solubility profiles of these camphor-based salts are often insufficiently distinct between diastereomers, necessitating multiple recrystallization steps to achieve acceptable purity, which further erodes overall process yield and increases solvent consumption.
The Novel Approach
In stark contrast, the methodology disclosed in CN1293039C utilizes (2R,3R)-tartaric acid derivatives, such as dibenzoyl tartaric acid or di-p-toluyl tartaric acid, to form diastereomeric salts with a distinct 2:1 stoichiometry. This shift in chemistry fundamentally alters the process dynamics. The new approach eliminates the need for aggressive thermal cycling; the reaction proceeds effectively at mild temperatures ranging from 0 to 40°C. The key innovation lies in the precise control of a mixed solvent system, typically comprising an alcohol and an ester or ether. By adjusting the volume ratio of these solvents, the process selectively precipitates the target [(S)-(-)-alpha-methylamino phenylketone]2.(2R,3R)-tartaric acid derivative while keeping the unwanted diastereomer in solution. This simplifies the isolation procedure to a straightforward filtration step. Moreover, the utilization of tartaric acid derivatives allows for a more efficient molar usage of the resolving agent compared to camphor-based alternatives, directly impacting the cost structure of the manufacturing process.
Mechanistic Insights into Asymmetric Transformation and Resolution
The core scientific advancement of this patent lies in the phenomenon of asymmetric transformation occurring concurrently with the resolution process. In a standard resolution of a racemate, the maximum theoretical yield for a single enantiomer is capped at 50%. However, the data presented in CN1293039C demonstrates optical yields reaching as high as 158.7%. This is mechanistically explained by the dynamic equilibrium established in the reaction mixture. As the less soluble [(S)-(-)-alpha-methylamino phenylketone]2.(2R,3R)-tartaric acid derivative crystallizes out of the solution, it disrupts the equilibrium between the (R) and (S) isomers in the mother liquor. Under the specific solvent conditions employed, the (R)-isomer undergoes conversion to the (S)-isomer, which then immediately reacts with the resolving agent to form more precipitate. This continuous cycle effectively drives the reaction beyond the static 50% limit, converting what would traditionally be waste material into valuable product. The structural integrity of the substrate, alpha-methylaminopropiophenone, is maintained throughout this process due to the mild conditions employed.
Furthermore, the patent addresses the critical issue of impurity control and mother liquor recycling. In traditional methods, recovering the unreacted enantiomer from the mother liquor often requires harsh alkaline conditions and prolonged heating, leading to decomposition. The novel method described herein employs a mild racemization protocol where the mother liquor is heated to only 40-90°C for a short duration of 0.5 to 4 hours. Crucially, this can be achieved without the immediate addition of strong acids or bases that might degrade the sensitive ketone structure. The resolving agent remains largely stable under these conditions, allowing for its recovery and reuse. This mechanistic understanding ensures that the final product achieves a diastereomeric excess (%de) of approximately 97.2% as measured by capillary electrophoresis, negating the need for further purification steps and ensuring a consistent quality profile suitable for downstream pharmaceutical synthesis.
How to Synthesize [(S)-(-)-alpha-methylamino phenylketone] Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for industrial implementation, focusing on solvent engineering and stoichiometric precision. The process begins with the dissolution of the substrate, which can be racemic or enriched, in a primary solvent such as methanol. The resolving agent, typically a (2R,3R)-tartaric acid derivative, is introduced either as a solution or directly, depending on the specific embodiment. The critical step involves the addition of a secondary anti-solvent, such as ethyl acetate, which modulates the solubility product of the target salt. Detailed operational parameters, including specific solvent ratios and stirring times, are essential for reproducibility. For a comprehensive guide on the exact experimental procedures and safety considerations, please refer to the standardized synthesis steps provided below.
- Dissolve racemic or enriched alpha-methylaminopropiophenone in a lower alcohol solvent such as methanol under stirring conditions at ambient temperature.
- Slowly add a solution of the chiral resolving agent, specifically a (2R,3R)-tartaric acid derivative like dibenzoyl tartaric acid, dissolved in the same alcohol solvent.
- Introduce a second solvent component, typically an ester like ethyl acetate, to the mixture to induce selective precipitation of the target diastereomeric salt, then filter and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1293039C offers substantial strategic benefits beyond mere technical superiority. The primary advantage is the drastic reduction in manufacturing costs driven by improved material efficiency. By achieving optical yields significantly higher than the theoretical 50% limit, the process effectively doubles the output per unit of raw material input compared to conventional resolution methods. This enhancement in atom economy directly translates to a lower cost of goods sold (COGS). Additionally, the 2:1 stoichiometric ratio between the substrate and the tartaric acid resolving agent means that less of the expensive chiral auxiliary is required per mole of product compared to the 1:1 ratio needed for camphoric acid derivatives. This optimization reduces the dependency on costly chiral pool materials, providing a buffer against price volatility in the raw material market.
- Cost Reduction in Manufacturing: The elimination of complex heating and cooling cycles results in significant energy savings. Traditional methods require precise thermal management to dissolve and recrystallize salts, consuming substantial utility resources. The new method operates effectively at ambient or slightly elevated temperatures, reducing the load on HVAC and heating systems. Furthermore, the simplified workflow reduces labor hours and equipment occupancy time, allowing for higher throughput in existing facilities. The ability to recycle the resolving agent with minimal loss further compounds these savings, creating a leaner and more cost-effective production model that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the solvent system, which utilizes common industrial solvents like methanol and ethyl acetate, ensures high supply chain resilience. Unlike specialized solvents that may face availability constraints, these commodities are readily accessible from multiple suppliers globally. The process tolerance to variations in feeding modes, as noted in the patent examples, adds another layer of reliability, reducing the risk of batch failures due to minor operational deviations. This consistency is crucial for maintaining uninterrupted supply to downstream API manufacturers, minimizing the risk of stockouts and ensuring timely delivery of critical pharmaceutical intermediates to clients worldwide.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is inherently greener. The reduction in solvent usage and the ability to recycle the mother liquor minimize the generation of hazardous waste streams. The mild racemization conditions prevent the formation of degradation by-products that would otherwise complicate waste treatment. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The simplicity of the unit operations—primarily mixing and filtration—facilitates easy scale-up from pilot plant to commercial production scales without the need for specialized high-pressure or cryogenic equipment, ensuring that supply can be rapidly ramped up to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process transfer and procurement teams assessing vendor capabilities. The clarity provided here aims to mitigate risks associated with technology adoption and ensure alignment between technical specifications and commercial expectations.
Q: How does the new resolution method exceed the theoretical 50% yield limit?
A: The process utilizes an asymmetric transformation mechanism where the precipitation of the less soluble (S)-isomer salt shifts the equilibrium in the mother liquor, causing the (R)-isomer to convert into the (S)-form, thereby achieving optical yields exceeding 150%.
Q: What are the advantages of using tartaric acid derivatives over camphoric acid?
A: Tartaric acid derivatives allow for a 2:1 stoichiometric ratio with the substrate compared to the 1:1 ratio required for camphoric acid, effectively doubling the utilization efficiency of the expensive chiral resolving agent while simplifying the thermal profile of the reaction.
Q: Is the racemization of the mother liquor complex to perform?
A: No, the patented method allows for direct heating of the mother liquor at moderate temperatures (40-90°C) for a short duration to racemize the remaining (R)-isomer without the need for harsh acidic or basic additives that typically degrade the product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [(S)-(-)-alpha-methylamino phenylketone] Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the pharmaceutical value chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated resolution chemistry described in CN1293039C can be translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of [(S)-(-)-alpha-methylamino phenylketone] meets the exacting standards required for ephedrine synthesis. Our commitment to quality assurance ensures that our clients receive materials with consistent optical purity and minimal impurity profiles, safeguarding the integrity of their final drug products.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior resolution method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of our high-performance intermediates into your production schedule.
