Advanced Oxidative Synthesis of Aryl Sulfonyl Fluoride for Commercial Scale-Up
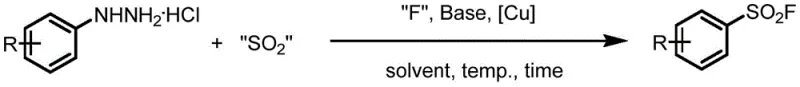
The chemical industry is witnessing a paradigm shift in the synthesis of sulfonyl fluoride compounds, driven by the increasing demand for robust building blocks in SuFEx click chemistry and drug discovery. Patent CN113603619A introduces a groundbreaking method for preparing aryl sulfonyl fluoride by utilizing aryl hydrazine hydrochloride as a primary raw material. Unlike conventional approaches that rely on sensitive reducing environments, this innovation employs a 'radical sulfur dioxide insertion fluorination' strategy under oxidative conditions. This technical breakthrough allows for the synthesis of high-purity aryl sulfonyl fluoride with exceptional selectivity and yield, fundamentally altering the manufacturing landscape for these critical pharmaceutical intermediates. The process leverages copper salt catalysis and alkali promotion to achieve efficient transformation, offering a reliable aryl sulfonyl fluoride supplier solution for global markets seeking stability and performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of aryl sulfonyl fluoride compounds has been fraught with significant operational challenges that hinder large-scale adoption. Most reported synthetic routes depend heavily on reduction conditions, making the reaction systems extremely susceptible to oxygen interference from the ambient air. This sensitivity necessitates rigorous oxygen removal treatments and the continuous use of inert gas atmospheres, such as argon or nitrogen, throughout the entire process. Such requirements not only complicate the operation but also escalate the capital expenditure for specialized reactor equipment and increase the overall production costs. Furthermore, traditional methods often involve harsh reaction conditions and complex multi-step sequences starting from less accessible precursors like aryl sulfonyl chlorides or thiols, which can introduce impurities and lower the overall atom economy of the synthesis.
The Novel Approach
In stark contrast, the methodology disclosed in CN113603619A revolutionizes the field by operating effectively under oxidative conditions where the influence of air is negligible. This novel approach utilizes aryl hydrazine hydrochloride, a readily available and stable starting material, reacting it with a sulfur dioxide source and a fluorinating reagent in the presence of a copper catalyst. The reaction proceeds smoothly at mild temperatures, typically around 40°C, and completes within a short timeframe of approximately 2 hours. By eliminating the need for strict inert gas protection, this method drastically simplifies the workflow and enhances safety. It provides a new idea for the synthesis of aryl sulfonyl fluoride that is universally applicable to various substrates, including those with electron-withdrawing or electron-donating substituents, ensuring consistent quality and cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Copper-Catalyzed Radical Sulfur Dioxide Insertion
The core of this technological advancement lies in its unique mechanistic pathway, which diverges from traditional reductive mechanisms. Under the promotion of a base and the catalysis of a copper salt, the aryl hydrazine hydrochloride undergoes oxidation to generate an aryl radical intermediate. This highly reactive species then combines with sulfur dioxide, provided by a source such as DABSO (1,4-diazabicyclo[2.2.2]octane-bis(sulfur dioxide) adduct), to form an aryl sulfonyl radical. Subsequently, a fluorinating reagent, preferably N-fluoro-bis-benzenesulfonamide (NFSI), facilitates the insertion of fluorine into the sulfonyl radical,最终 yielding the target aryl sulfonyl fluoride. This radical cascade is meticulously controlled by the copper catalyst, which ensures high selectivity and prevents the formation of unwanted by-products. The use of basic copper carbonate as the preferred catalyst further optimizes the reaction kinetics, allowing for efficient turnover and minimal metal residue in the final product.
Impurity control is another critical aspect where this mechanism excels, directly addressing the concerns of R&D directors focused on purity profiles. The oxidative nature of the reaction minimizes side reactions commonly associated with reducing agents, such as over-reduction or decomposition of sensitive functional groups. The compatibility of this system with a wide range of solvents, with acetonitrile being the optimal choice, allows for fine-tuning of the reaction environment to maximize solubility and reaction rates. Moreover, the mild conditions prevent the degradation of thermally labile substituents on the aryl ring, preserving the structural integrity of complex molecules. This level of control results in a cleaner crude product, reducing the burden on downstream purification processes and ensuring that the final high-purity aryl sulfonyl fluoride meets stringent quality specifications required for bioconjugation and medicinal chemistry applications.
How to Synthesize Aryl Sulfonyl Fluoride Efficiently
To implement this cutting-edge synthesis route effectively, precise adherence to the optimized reaction parameters is essential for achieving maximum yield and reproducibility. The process begins with the careful selection of reagents, specifically using aryl hydrazine hydrochloride as the substrate, DABSO as the sulfur dioxide source, and NFSI as the fluorine source, all mediated by a copper catalyst and pyridine base in acetonitrile. Detailed standardized synthesis steps see the guide below, which outlines the exact molar ratios and procedural nuances required to replicate the success demonstrated in the patent examples. Following these guidelines ensures that the transition from laboratory scale to pilot production is seamless, maintaining the high efficiency and selectivity that define this innovative method.
- Prepare the reaction mixture by combining aryl hydrazine hydrochloride, basic copper carbonate catalyst, DABSO as the sulfur dioxide source, and NFSI as the fluorinating agent in acetonitrile solvent.
- Add pyridine as the base promoter to facilitate the radical generation and ensure the reaction proceeds under mild oxidative conditions without the need for strict inert gas protection.
- Heat the reaction mixture to 40°C for approximately 2 hours, then filter solid impurities and purify the crude product via column chromatography to obtain high-purity aryl sulfonyl fluoride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxidative synthesis method translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced supply chain reliability. By removing the dependency on inert gas atmospheres and complex oxygen-scavenging protocols, facilities can utilize standard reactor setups, thereby lowering capital investment and maintenance costs. This operational flexibility allows for faster turnaround times and greater agility in responding to market demands, making it an ideal solution for reducing lead time for high-purity aryl sulfonyl fluorides in a competitive global market.
- Cost Reduction in Manufacturing: The elimination of expensive inert gas requirements and the use of commercially abundant raw materials like aryl hydrazine hydrochloride significantly lower the input costs associated with production. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes waste generation and the need for extensive purification steps. These factors collectively contribute to substantial cost savings without compromising on the quality of the final product, offering a more economically viable route for large-scale manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this method against atmospheric oxygen means that production is less susceptible to interruptions caused by gas supply issues or equipment failures related to inertion systems. The use of stable and widely available reagents ensures a consistent supply of raw materials, mitigating the risk of shortages. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments, thereby strengthening the overall resilience of the supply chain for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the absence of hazardous reducing agents make this process inherently safer and easier to scale up from laboratory to industrial levels. The reduced generation of toxic by-products and the potential for solvent recycling align with modern environmental regulations and sustainability goals. This eco-friendly profile not only facilitates regulatory approval but also enhances the corporate social responsibility standing of the manufacturing entity, appealing to environmentally conscious partners and clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this novel synthesis method, providing clarity for stakeholders evaluating its implementation. These insights are derived directly from the experimental data and technical disclosures within the patent, ensuring accuracy and relevance for decision-makers. Understanding these aspects is vital for assessing the feasibility of integrating this technology into existing production workflows and for leveraging its full potential in commercial applications.
Q: Why is this oxidative method superior to traditional reductive sulfonyl fluorination?
A: Traditional methods often require harsh reducing conditions and are highly sensitive to oxygen, necessitating complex inert gas setups. This novel oxidative strategy utilizes air-tolerant copper catalysis, significantly simplifying operation and reducing equipment costs while maintaining excellent yields.
Q: What are the key advantages of using aryl hydrazine hydrochloride as a starting material?
A: Aryl hydrazine hydrochlorides are commercially available, stable, and cost-effective raw materials. Their use avoids the need for pre-functionalized sulfonyl chlorides or unstable diazonium salts, streamlining the supply chain and enhancing the overall safety profile of the manufacturing process.
Q: Can this process be scaled up for industrial production of pharmaceutical intermediates?
A: Yes, the mild reaction conditions (40°C), short reaction time (2 hours), and insensitivity to air make this process highly scalable. The elimination of strict oxygen-free requirements reduces engineering bottlenecks, facilitating efficient commercial scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Sulfonyl Fluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this oxidative synthesis route and are fully equipped to bring it to life on a commercial scale. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to reality. Our state-of-the-art facilities are designed to handle complex chemistries with precision, adhering to stringent purity specifications and operating through rigorous QC labs to guarantee the highest quality standards. We are committed to delivering reliable aryl sulfonyl fluoride solutions that meet the exacting demands of the global pharmaceutical and fine chemical industries.
We invite you to collaborate with us to optimize your supply chain and unlock new efficiencies in your manufacturing processes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs, demonstrating how this advanced method can drive value for your organization. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge with our high-performance chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
