Advanced Catalytic Asymmetric Transfer Hydrogenation for High-Purity Pharmaceutical Intermediates
Advanced Catalytic Asymmetric Transfer Hydrogenation for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands more efficient and scalable routes for the production of chiral building blocks, particularly for cardiovascular and neurological therapeutics. Patent CN1445224A introduces a groundbreaking methodology for the preparation of enantiomerically enriched arylaminopropanols, which serve as critical precursors for thrombotonin and norepinephrine uptake inhibitors. This technology represents a paradigm shift from traditional stoichiometric reductions to a highly efficient catalytic asymmetric transfer hydrogenation process. By leveraging specific Ruthenium-based catalyst systems, the invention achieves superior stereocontrol while drastically reducing the reliance on costly chiral auxiliaries. For global procurement teams and R&D directors, this patent outlines a pathway to secure a reliable pharmaceutical intermediate supplier capable of delivering high-value chiral amines with exceptional purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-aryl-3-amino-1-propanols has relied on methods that are economically and operationally inefficient for large-scale manufacturing. Traditional approaches, such as the Corey and Reichard method, utilize chiral borane reagents for the asymmetric reduction of ketones. While effective, these stoichiometric reagents are prohibitively expensive and generate significant amounts of boron-containing waste, complicating environmental compliance. Furthermore, alternative enzymatic resolution strategies suffer from an inherent theoretical yield limit of 50%, as they require the separation of racemic mixtures, discarding half of the produced material. These legacy processes result in inflated production costs and extended lead times, creating bottlenecks in the supply chain for cost reduction in API manufacturing.
The Novel Approach
The innovative process detailed in CN1445224A overcomes these historical barriers by employing a transition-metal catalyzed transfer hydrogenation strategy. Instead of using molecular hydrogen under high pressure or expensive stoichiometric reducers, this method utilizes a formic acid-triethylamine mixture as a convenient hydrogen donor. This approach allows for the direct conversion of achiral beta-keto esters into chiral beta-hydroxy esters with outstanding enantioselectivity. The elimination of the resolution step theoretically doubles the yield compared to enzymatic methods, while the catalytic nature of the chirality source significantly lowers the raw material burden. This novel route ensures a robust and scalable supply of complex pharmaceutical intermediates, addressing the critical need for commercial scale-up of complex polymer additives and fine chemicals.
Mechanistic Insights into Ru-Catalyzed Asymmetric Transfer Hydrogenation
The core of this technological breakthrough lies in the precise design of the chiral catalyst system, which dictates the stereochemical outcome of the reaction. The process utilizes a Ruthenium complex formed in situ from a ruthenium arene dimer, such as (cumene) ruthenium chloride dimer, and a chiral diamine ligand of general formula (XIII). These ligands, often derived from 1,2-diphenylethylenediamine scaffolds substituted with sulfonyl groups, create a highly defined chiral environment around the metal center. During the catalytic cycle, the Ruthenium hydride species transfers a hydride ion to the prochiral ketone substrate in a concerted manner, guided by the steric bulk of the ligand. This mechanism ensures that the reduction occurs exclusively from one face of the carbonyl group, establishing the desired stereogenic center with high fidelity.
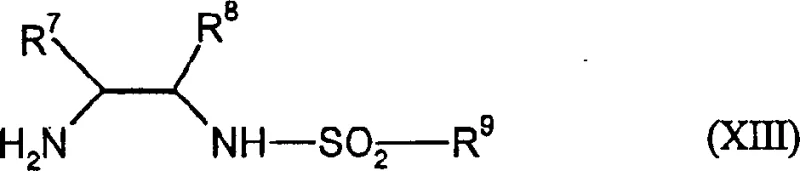
Beyond mere conversion, the mechanistic elegance of this system provides exceptional control over the impurity profile, which is paramount for regulatory compliance in drug substance manufacturing. The high turnover number of the catalyst, often operating at loadings as low as 0.02 to 0.1 mol%, minimizes the risk of heavy metal contamination in the final product. Furthermore, the mild reaction conditions, typically ranging from 20°C to 80°C, prevent thermal degradation of sensitive functional groups that might occur under harsher reducing conditions. The use of formic acid as a hydrogen source also generates carbon dioxide and water as byproducts, which are easily removed, thereby simplifying the workup procedure. This results in a high-purity pharmaceutical intermediate that requires minimal downstream purification, directly impacting the overall cost of goods sold.
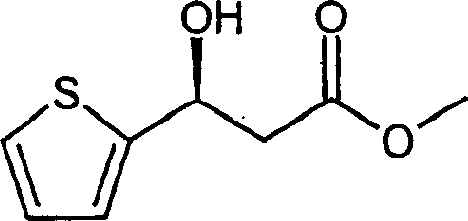
How to Synthesize Methyl (3S)-3-hydroxy-3-(2-thienyl)propionate Efficiently
The practical implementation of this patent involves a streamlined sequence that begins with the formation of the beta-keto ester substrate, followed by the pivotal asymmetric reduction step. The initial phase typically involves a Claisen condensation between an acetophenone derivative, such as 2-acetylthiophene, and a carbonate ester in the presence of a base like sodium methylate. Once the substrate is prepared, it undergoes the catalytic asymmetric transfer hydrogenation described previously. This step transforms the ketone into the corresponding chiral alcohol with excellent enantiomeric excess, often exceeding 98% ee as demonstrated in the patent examples. The resulting chiral hydroxy ester serves as a versatile platform molecule for subsequent functionalization into the final amine target.
- Prepare the beta-keto ester substrate via Claisen condensation of acetothiophene and methyl carbonate using sodium methylate.
- Perform asymmetric transfer hydrogenation using a Ruthenium-TsDPEN catalyst complex with a formic acid-triethylamine azeotrope to generate the chiral hydroxy ester.
- Convert the chiral ester to the amide using methylamine, followed by reduction with lithium aluminum hydride to yield the final arylaminopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1445224A offers transformative benefits regarding cost structure and operational reliability. The shift from stoichiometric chiral reagents to a catalytic system fundamentally alters the economics of production by decoupling the cost of chirality from the volume of production. Since the chiral information is imparted by a catalyst that is used in minute quantities, the marginal cost of producing each additional kilogram of product is significantly reduced. This efficiency gain allows suppliers to offer more competitive pricing structures without compromising on quality, directly supporting initiatives for cost reduction in API manufacturing across the organization.
- Cost Reduction in Manufacturing: The elimination of expensive chiral boranes and the avoidance of 50% yield loss from resolution steps creates a substantial economic advantage. By utilizing inexpensive hydrogen donors like formic acid and low-loading Ruthenium catalysts, the raw material costs are drastically minimized. Additionally, the simplified workup procedures reduce solvent consumption and energy usage during purification, further driving down the operational expenditure associated with producing these high-value intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as simple acetophenones and carbonates, mitigates the risk of supply disruptions often associated with specialized chiral pool reagents. The robustness of the catalytic system ensures consistent batch-to-batch reproducibility, which is critical for maintaining continuous manufacturing schedules. This stability allows for better inventory planning and reduces the lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug production lines remain uninterrupted.
- Scalability and Environmental Compliance: The process operates under mild conditions without the need for high-pressure hydrogen gas infrastructure, making it inherently safer and easier to scale from pilot plant to commercial tonnage. The generation of benign byproducts like carbon dioxide aligns with modern green chemistry principles, reducing the burden on waste treatment facilities. This environmental compatibility facilitates smoother regulatory approvals and supports corporate sustainability goals, making the technology attractive for long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing process workflows. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of the transfer hydrogenation method over traditional borane reduction?
A: The transfer hydrogenation method described in CN1445224A eliminates the need for expensive stoichiometric chiral borane reagents, utilizing a catalytic amount of Ruthenium complex instead, which significantly lowers material costs and simplifies waste handling.
Q: What level of enantiomeric excess (ee) can be achieved with this process?
A: The patent data demonstrates that the process can achieve extremely high stereochemical purity, with specific examples showing enantiomeric excess values of 98.2% ee or higher, ensuring minimal need for downstream chiral purification.
Q: Is high-pressure hydrogen gas required for this synthesis?
A: No, the process utilizes a hydrogen-transfer protocol using formic acid and triethylamine as the hydrogen source, which removes the safety hazards and specialized equipment requirements associated with high-pressure hydrogenation reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylaminopropanols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable supply chain for complex chiral intermediates essential for next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative methods described in CN1445224A can be successfully translated from the laboratory to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of arylaminopropanols meets the exacting standards required by global regulatory bodies, providing our partners with peace of mind and product consistency.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your development timeline with superior chemical solutions.
