Advanced Stereoselective Synthesis of Optically Active 2-Halo-3-Hydroxypropionate Esters for Diltiazem Production
The pharmaceutical industry continuously seeks robust pathways for synthesizing high-value chiral intermediates, particularly for cardiovascular medications like calcium channel blockers. Patent CN1168878A introduces a groundbreaking methodology for the preparation of optically active 2-halo-3-hydroxypropionate compounds, which serve as critical precursors for drugs such as diltiazem hydrochloride. This innovation addresses long-standing challenges in asymmetric synthesis by employing chiral Lewis acids to facilitate the reaction between aldehyde compounds and silylketene acetal derivatives. Unlike traditional methods that often struggle with low yields or poor stereocontrol, this process achieves high optical purity through a single-step catalytic reaction. For procurement leaders and R&D directors, this represents a significant opportunity to streamline the supply chain for high-purity pharmaceutical intermediates while mitigating the risks associated with complex multi-step syntheses.
The versatility of this approach allows for the use of various aldehyde substrates, including substituted phenyl and naphthyl groups, providing flexibility in designing diverse analog libraries. By leveraging specific chiral titanium or boron complexes, manufacturers can precisely dictate the stereochemical outcome, favoring either the (3S) or (3R) configuration as required for downstream processing. This level of control is essential for ensuring the efficacy and safety of the final active pharmaceutical ingredient (API). As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is key to delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of optically active 2-halo-3-hydroxypropionates relied heavily on methods that were fraught with industrial inefficiencies. Traditional approaches included the use of chiral lithium amide compounds, which require stringent anhydrous conditions and cryogenic temperatures that are energy-intensive and difficult to maintain in large reactors. Another common method involved asymmetric reduction using microorganisms, which, while selective, suffers from slow reaction rates, sensitivity to substrate toxicity, and the high cost of maintaining sterile fermentation environments. Furthermore, methods utilizing chiral boron compounds with tert-butyl haloacetates often resulted in low yields and generated significant amounts of hazardous waste, complicating disposal and increasing the overall environmental footprint of the manufacturing process.
Additionally, existing chemical routes often struggled with the steric and electronic effects of halogen atoms, leading to poor diastereoselectivity and the formation of unwanted isomers that required costly and time-consuming purification steps. The reliance on expensive chiral auxiliaries that could not be recovered further exacerbated the cost burden, making these processes economically unviable for cost reduction in pharmaceutical intermediates manufacturing. These limitations created a bottleneck in the supply chain, where the availability of high-quality intermediates was inconsistent, posing risks to the continuity of API production for major pharmaceutical companies.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by utilizing a chiral Lewis acid-catalyzed aldol-type reaction. This method reacts an aldehyde compound directly with a silylketene acetal derived from haloacetates in the presence of a chiral titanium or boron catalyst. This one-pot transformation is remarkably efficient, operating effectively at temperatures between -100°C and 20°C, with optimal results often observed between -78°C and -10°C. The use of common organic solvents such as toluene, dichloromethane, or tetrahydrofuran simplifies the process engineering requirements, allowing for easier commercial scale-up of complex pharmaceutical intermediates.
Crucially, this method accommodates both mono-halo and di-halo substrates, providing a versatile platform for synthesizing a wide range of derivatives. The stereoselectivity is driven by the specific geometry of the chiral ligand coordinated to the metal center, which creates a defined chiral environment that favors the formation of the desired enantiomer. This eliminates the need for resolution steps or the use of stoichiometric amounts of chiral reagents, drastically reducing material costs. The result is a streamlined process that delivers high optical purity with minimal waste, aligning perfectly with modern green chemistry principles and supply chain sustainability goals.
Mechanistic Insights into Chiral Lewis Acid-Catalyzed Stereoselection
The core of this technology lies in the precise interaction between the chiral Lewis acid catalyst and the reactants. The catalyst, typically a complex of titanium or boron with chiral ligands such as binaphthol derivatives or amino acid-based structures, activates the carbonyl group of the aldehyde while simultaneously organizing the silylketene acetal in a specific orientation. For instance, chiral titanium compounds of formula (IV) or (V) create a rigid coordination sphere that shields one face of the aldehyde, forcing the nucleophilic attack to occur from the less hindered side. This spatial arrangement is critical for achieving high enantiomeric excess, as even minor deviations can lead to the formation of the wrong isomer.
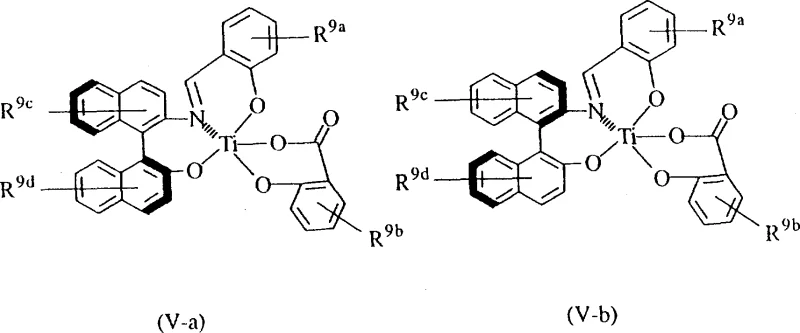
The presence of halogen atoms (chlorine or bromine) on the silylketene acetal introduces unique electronic and steric challenges due to their high electronegativity and bulk. However, the patented catalyst systems are specifically designed to accommodate these features. When a mono-halo compound is used, chiral titanium catalysts are preferred, whereas di-halo compounds respond better to chiral boron catalysts. This differentiation ensures that the transition state remains energetically favorable for the desired pathway. Impurity control is managed by optimizing the ratio of the catalyst to the substrate, typically ranging from 1 mol% to 200 mol%, with 10-120 mol% being preferred for maximum efficiency. This fine-tuning minimizes side reactions such as non-selective background aldol condensations.
Following the initial coupling, the resulting 2-halo-3-hydroxypropionate can be further transformed into trans-2,3-epoxypropionate derivatives through an intramolecular cyclization reaction under basic conditions. This subsequent step preserves the optical integrity established in the first step, converting the 3S-isomers into specific epoxy configurations required for diltiazem synthesis. The ability to predictably map the stereochemistry from the initial aldol adduct to the final epoxy intermediate provides R&D teams with a reliable roadmap for process development, ensuring that the reducing lead time for high-purity pharmaceutical intermediates is achieved without compromising on quality specifications.
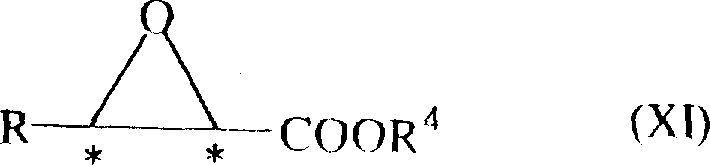
How to Synthesize Optically Active 2-Halo-3-Hydroxypropionate Efficiently
The synthesis protocol outlined in the patent provides a clear framework for executing this reaction in a laboratory or pilot plant setting. It begins with the preparation of the chiral catalyst, followed by the controlled addition of reactants at low temperatures to maintain stereocontrol. The workup involves standard aqueous quenching and extraction techniques, making it compatible with existing infrastructure. For detailed operational parameters, stoichiometry, and specific purification methods, please refer to the standardized guide below.
- Prepare the chiral Lewis acid catalyst, such as a chiral titanium or boron complex, by reacting ligands with metal sources like tetraisopropyl orthotitanate or borane.
- React the aldehyde compound with the silylketene acetal compound in the presence of the chiral Lewis acid at temperatures between -100°C and 20°C.
- Quench the reaction with aqueous solutions, extract the organic layer, and purify the resulting 2-halo-3-hydroxypropionate ester via column chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this catalytic methodology offers substantial strategic benefits for procurement and supply chain management, primarily by simplifying the sourcing of raw materials and reducing dependency on specialized biological processes. The shift from fermentation or stoichiometric chiral reagents to a catalytic chemical process means that manufacturers can leverage widely available commodity chemicals, thereby stabilizing costs and insulating the supply chain from volatility associated with niche reagents. This transition supports a more resilient manufacturing model capable of responding quickly to market demand fluctuations for cardiovascular drugs.
- Cost Reduction in Manufacturing: The elimination of expensive chiral auxiliaries and the avoidance of microbial fermentation significantly lowers the variable cost per kilogram of the intermediate. By using catalytic amounts of chiral Lewis acids instead of stoichiometric chiral reagents, the material cost is drastically reduced. Furthermore, the simplified workup procedure reduces solvent consumption and energy usage associated with distillation and drying, contributing to overall operational expenditure savings without the need for complex capital investment in bioreactors.
- Enhanced Supply Chain Reliability: The reliance on robust chemical catalysis rather than sensitive biological systems enhances the reliability of supply. Chemical processes are generally less prone to batch-to-batch variability caused by biological factors such as strain mutation or contamination. This consistency ensures a steady flow of intermediates, reducing the risk of production delays for downstream API manufacturers. The use of stable, shelf-stable catalysts and reagents also simplifies inventory management and logistics.
- Scalability and Environmental Compliance: This process is inherently scalable, moving seamlessly from gram-scale optimization to multi-ton commercial production. The reduction in hazardous waste generation, particularly the avoidance of heavy metal waste from stoichiometric reagents or biological sludge from fermentation, simplifies regulatory compliance and waste disposal. This aligns with increasingly stringent environmental regulations, reducing the administrative burden and potential liabilities associated with industrial chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this route for your specific production needs. Understanding these details is crucial for making informed decisions about process integration and vendor selection.
Q: What are the primary advantages of this chiral Lewis acid method over traditional microbial reduction?
A: This chemical catalytic method offers superior control over stereoselectivity without the complexities of fermentation, significantly reducing production time and eliminating the need for specialized biological containment facilities.
Q: Can this process be adapted for large-scale commercial manufacturing of diltiazem intermediates?
A: Yes, the process utilizes standard organic solvents like toluene and dichloromethane and avoids expensive, hard-to-source reagents, making it highly suitable for industrial scale-up from kilogram to metric ton quantities.
Q: How does the choice of halogen affect the optical purity of the product?
A: The patent indicates that while fluorine-containing compounds behave differently, chlorine and bromine-containing silylketene acetals can achieve high optical purity when paired with specific chiral titanium or boron catalysts tailored to the halogen's steric environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Halo-3-Hydroxypropionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-halo-3-hydroxypropionate meets the exacting standards required for pharmaceutical synthesis, providing you with a partner dedicated to excellence and consistency.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced catalytic capabilities can support your long-term strategic goals in the pharmaceutical sector.
