Advanced Enantioselective Hydrocyanation for Commercial Scale-up of Complex Pharmaceutical Intermediates
The chemical industry is constantly evolving towards more efficient and sustainable synthetic pathways, particularly in the realm of chiral synthesis where optical purity is paramount for biological activity. Patent CN1314880A introduces a groundbreaking methodology for the preparation of optically active alpha-aminonitriles through enantioselective hydrocyanation of ketimines. This technology represents a significant leap forward from traditional racemic synthesis followed by resolution, offering a direct route to chiral building blocks that are essential precursors for alpha-amino acids, esters, and amides. The core innovation lies in the utilization of chiral or chirality-inducing metal complexes that facilitate the addition of cyanide to the imine double bond with high stereocontrol. This approach not only enhances the enantiomeric excess of the final product but also streamlines the overall manufacturing process by eliminating the need for costly and wasteful separation of unwanted enantiomers. For global pharmaceutical and agrochemical manufacturers, this patent provides a robust framework for producing high-value intermediates with superior quality attributes.
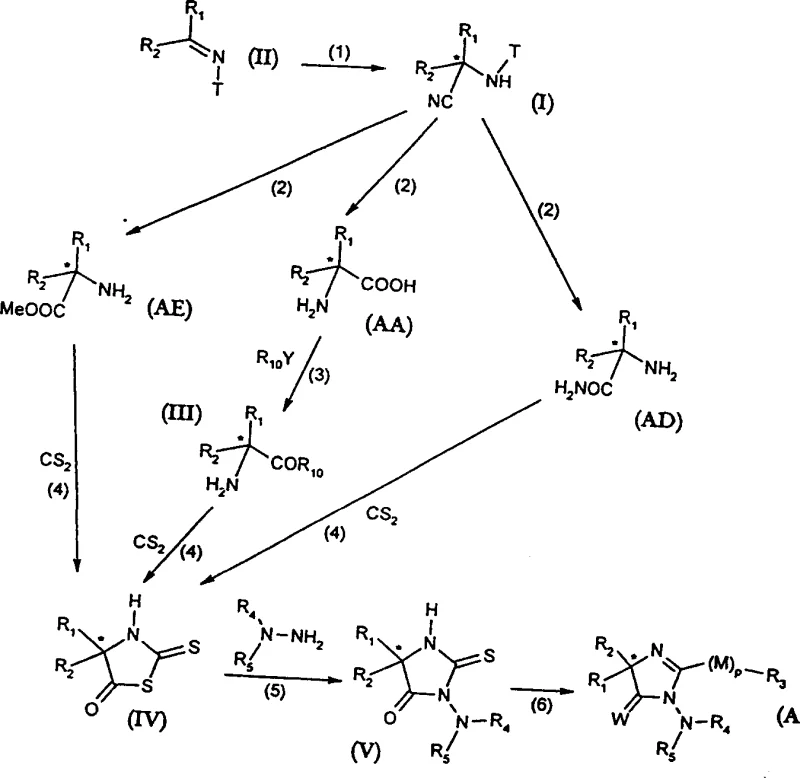
The versatility of this synthetic route is further evidenced by its applicability in the production of complex bioactive molecules, including fungicides such as 2-imidazolin-5-ones. By establishing a reliable supply chain for these critical chiral intermediates, manufacturers can secure the raw materials necessary for next-generation therapeutic and agricultural products. The method described allows for the precise tuning of stereochemistry, ensuring that the resulting alpha-aminonitriles possess the specific R or S configuration required for downstream biological efficacy. This level of control is indispensable in modern drug development, where regulatory agencies demand rigorous characterization of chiral impurities. Consequently, adopting this technology positions companies at the forefront of fine chemical innovation, enabling them to meet the stringent purity specifications of international markets while optimizing their production costs through improved catalytic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active alpha-amino acids and their precursors has relied heavily on the resolution of racemic mixtures, a process fraught with inherent inefficiencies and economic drawbacks. Traditional resolution techniques, whether through liquid chromatography, enzymatic catalysis, or diastereomeric salt formation, fundamentally suffer from a maximum theoretical yield of 50% for the desired enantiomer, meaning half of the synthesized material is essentially wasted or requires energy-intensive recycling. Furthermore, these separation processes often involve expensive chiral resolving agents, complex purification steps, and significant solvent consumption, all of which contribute to a high cost of goods sold and a larger environmental footprint. In an industrial setting, the difficulty of scaling these resolution methods often leads to bottlenecks in supply, inconsistent batch-to-batch optical purity, and prolonged lead times that can disrupt downstream manufacturing schedules. The reliance on stoichiometric amounts of chiral auxiliaries in older synthetic routes further exacerbates these issues, as the removal and recovery of these auxiliaries add additional unit operations that increase both capital expenditure and operational complexity.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN1314880A utilizes a catalytic enantioselective hydrocyanation strategy that directly generates the desired chiral center with high fidelity, effectively bypassing the limitations of resolution. By employing chiral metal complexes, such as those derived from Titanium or Aluminum with specialized ligands, the reaction selectively favors the formation of one enantiomer over the other, potentially achieving yields and optical purities that far exceed the 50% ceiling of resolution methods. This catalytic cycle allows for the turnover of the chiral information, meaning a small amount of expensive chiral ligand can generate a large quantity of product, drastically reducing the material cost per kilogram. The process is designed to be operationally simple, often proceeding under mild conditions in common organic solvents like toluene or dichloromethane, which facilitates easier handling and safer scale-up. Moreover, the resulting alpha-aminonitriles are versatile intermediates that can be readily hydrolyzed to amino acids or converted into amides and esters, providing a flexible platform for synthesizing a wide array of chiral organic compounds without the need for multiple protection and deprotection steps.
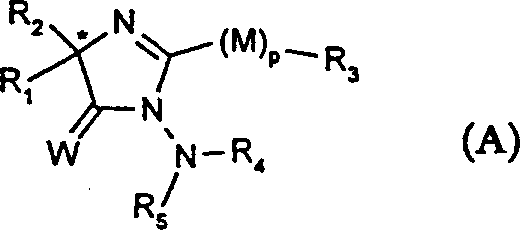
Mechanistic Insights into Chiral Metal-Catalyzed Hydrocyanation
The success of this enantioselective transformation hinges on the precise design of the chiral catalyst, which creates a sterically defined environment around the metal center to direct the approach of the cyanide nucleophile. The patent describes both monometallic and bimetallic catalyst systems, where the metal ions (such as Ti(IV), Al(III), or Zr(IV)) are coordinated by chiral ligands like BINOL (1,1'-bi-2-naphthol) or TADDOL (alpha,alpha,alpha',alpha'-tetraaryl-1,3-dioxolane-4,5-dimethanol). These ligands impart chirality to the metal complex either through their own chiral axes or centers, or by inducing a chiral conformation due to steric bulk. During the reaction, the ketimine substrate coordinates to the Lewis acidic metal center, activating the carbon-nitrogen double bond towards nucleophilic attack. The chiral pocket formed by the ligand framework ensures that the cyanide ion attacks the imine carbon from only one specific face, thereby establishing the new stereocenter with high enantiomeric excess. This mechanism avoids the formation of racemic byproducts and minimizes the generation of impurities that are difficult to separate, leading to a cleaner reaction profile and simplified downstream processing.
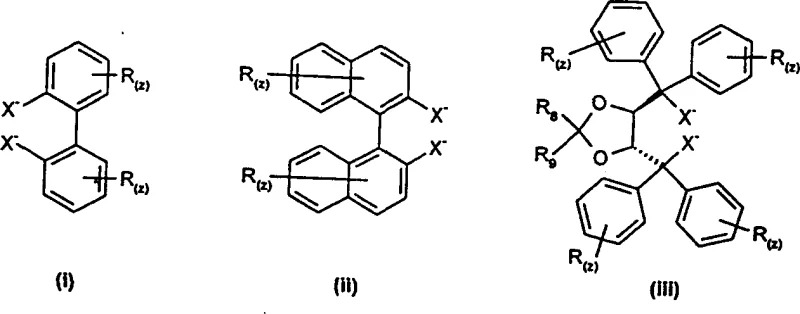
Furthermore, the choice of ligand substituents plays a critical role in fine-tuning the catalyst's performance, allowing chemists to optimize both activity and selectivity for specific substrates. For instance, the introduction of bulky groups like tert-butyl or phenyl rings on the ligand backbone can enhance the steric differentiation between the two prochiral faces of the ketimine, leading to higher enantiomeric purity. The patent highlights that the catalyst can be prepared either ex-situ or in-situ, offering flexibility in process design; in-situ generation eliminates the need for isolating sensitive catalyst intermediates, reducing exposure to air and moisture and simplifying the workflow. Understanding these mechanistic nuances is vital for R&D teams aiming to adapt this chemistry to new substrates, as it provides a rational basis for ligand selection and reaction condition optimization. By controlling factors such as temperature, solvent polarity, and the nature of the hydrocyanation reagent (e.g., TMSCN vs. HCN), manufacturers can achieve reproducible results that meet the rigorous quality standards required for pharmaceutical grade intermediates.
How to Synthesize Optically Active Alpha-Aminonitriles Efficiently
The synthesis of these valuable chiral intermediates follows a streamlined protocol that integrates catalyst preparation, reaction, and workup into a cohesive manufacturing sequence. The process begins with the generation of the active chiral metal species, typically by mixing a metal salt with the chosen chiral ligand in an anhydrous solvent under inert atmosphere. Once the catalyst is formed, the ketimine substrate and the hydrocyanation reagent are introduced, often at controlled low temperatures to maximize stereocontrol. The reaction progress is monitored using standard analytical techniques until completion, after which the mixture is quenched and the product is isolated through conventional extraction and purification methods. For a detailed breakdown of the specific reagents, molar ratios, and temperature profiles required to replicate this high-efficiency synthesis, please refer to the standardized guide below.
- Preparation of the chiral metal complex catalyst (monometallic or bimetallic) using ligands such as BINOL or TADDOL.
- Reaction of the ketimine substrate with a hydrocyanation reagent (e.g., TMSCN or HCN) in the presence of the catalyst at low temperatures.
- Workup and purification of the resulting optically active alpha-aminonitrile, followed by optional hydrolysis to amino acids.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enantioselective hydrocyanation technology offers transformative benefits that extend well beyond the laboratory bench. By shifting from a resolution-based model to a direct asymmetric synthesis, companies can fundamentally alter their cost structures and supply reliability. The elimination of the resolution step means that the theoretical yield is no longer capped at 50%, effectively doubling the output from the same amount of starting materials and significantly lowering the cost per unit of the active ingredient. This efficiency gain translates directly into improved margins and a more competitive pricing strategy in the global market. Additionally, the reduced number of processing steps simplifies the manufacturing workflow, decreasing the demand for equipment, labor, and utilities, which further contributes to substantial cost savings in large-scale production environments.
- Cost Reduction in Manufacturing: The catalytic nature of this process means that expensive chiral ligands are used in sub-stoichiometric amounts, unlike traditional methods that require stoichiometric quantities of resolving agents. This drastic reduction in the consumption of high-value chiral materials leads to a significant decrease in raw material costs. Furthermore, the avoidance of resolution waste means that less raw material is needed overall to produce the same amount of final product, optimizing the atom economy of the entire synthesis. The simplified purification requirements also reduce the consumption of solvents and adsorbents, lowering waste disposal costs and environmental compliance expenses. Overall, the process economics are vastly superior, enabling manufacturers to offer high-purity chiral intermediates at a more attractive price point without compromising on quality.
- Enhanced Supply Chain Reliability: Relying on resolution methods often introduces volatility into the supply chain due to the variability in separation efficiencies and the potential loss of material during recycling. In contrast, this direct catalytic method provides a more predictable and consistent output, ensuring that delivery schedules can be met with greater certainty. The robustness of the catalyst systems described allows for stable long-term storage and usage, reducing the risk of production delays caused by catalyst degradation or supply shortages of resolving agents. By securing a manufacturing route that is less dependent on complex separation technologies, companies can build a more resilient supply chain capable of withstanding market fluctuations and unexpected disruptions. This reliability is crucial for maintaining continuous production lines for downstream pharmaceutical and agrochemical products.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common industrial solvents and reaction conditions that are easily transferred from pilot plant to full commercial production. The reduction in waste generation, particularly the elimination of the unwanted enantiomer waste stream, aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. Lower solvent usage and simpler workup procedures result in a smaller environmental footprint, facilitating easier permitting and compliance with sustainability goals. This makes the technology not only economically viable but also environmentally responsible, appealing to stakeholders who prioritize corporate social responsibility and sustainable manufacturing practices in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enantioselective hydrocyanation technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production portfolios. For further customization or specific technical data sheets, our team is available to provide detailed consultations tailored to your project requirements.
Q: What are the advantages of this enantioselective hydrocyanation method over traditional resolution?
A: Unlike traditional resolution which theoretically limits yield to 50% and requires expensive separation steps, this catalytic method directly synthesizes the desired enantiomer with high optical purity, significantly improving atom economy and reducing waste.
Q: Which catalysts are preferred for this synthesis?
A: The patent highlights chiral or chirality-inducing metal complexes, particularly those based on Titanium (IV) or Aluminum (III) coordinated with ligands like BINOL, TADDOL, or substituted biphenols.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes robust catalytic systems and standard organic solvents like toluene or dichloromethane, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Alpha-Aminonitriles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of advanced pharmaceuticals and agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of optically active alpha-aminonitriles we produce. We are committed to delivering products that meet the highest international standards, providing you with the confidence and reliability needed to accelerate your drug development pipelines.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific applications. Our technical procurement team is ready to assist you with a Customized Cost-Saving Analysis to demonstrate how switching to this catalytic route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these vital chiral building blocks for your future projects.
