Advanced Synthesis of N-Substituted Piperidine-4-Boronic Acid Intermediates for Pharmaceutical Manufacturing
Advanced Synthesis of N-Substituted Piperidine-4-Boronic Acid Intermediates for Pharmaceutical Manufacturing
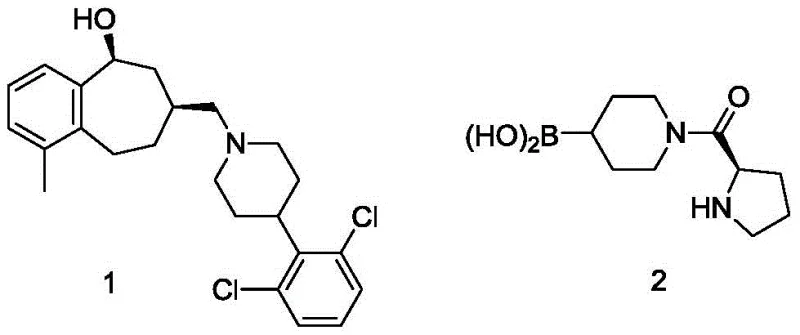
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more cost-effective, and scalable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111171063B, which outlines a novel process for synthesizing N-substituted piperidine-4-boronic acid derivatives. These compounds serve as critical building blocks for complex drug molecules, including opioid receptor antagonists and fatty acid amide hydrolase inhibitors. The traditional reliance on expensive precursors and hazardous catalytic hydrogenation has long been a bottleneck for supply chain efficiency. This new methodology addresses these challenges by introducing a streamlined two-step sequence that utilizes readily available alcohols and avoids precious metal catalysts entirely. By shifting the paradigm from palladium-catalyzed coupling to a lithiation-borylation strategy, manufacturers can achieve substantial improvements in both safety profiles and economic viability. This report analyzes the technical merits and commercial implications of this innovative approach for global procurement and R&D teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted piperidine-4-boronic acids has relied heavily on palladium-catalyzed coupling reactions. The standard literature methods typically utilize N-substituted-1,2,5,6-tetrahydropyridine-4-boric acid as the starting material. This precursor is not only prohibitively expensive, with market prices reaching exorbitant levels per kilogram, but its synthesis often involves complex multi-step sequences that degrade overall yield. Furthermore, the subsequent conversion to the saturated piperidine derivative necessitates a catalytic hydrogenation step using palladium hydroxide. This introduces significant safety risks associated with high-pressure hydrogen gas handling and the potential for pyrophoric catalyst exposure. From a supply chain perspective, dependence on palladium creates vulnerability to fluctuating precious metal markets and complicates impurity control due to the difficulty of removing trace heavy metals to ppm levels required for API production. The cumulative effect is a high-cost, high-risk manufacturing process that struggles to meet the demands of large-scale commercial production.
The Novel Approach
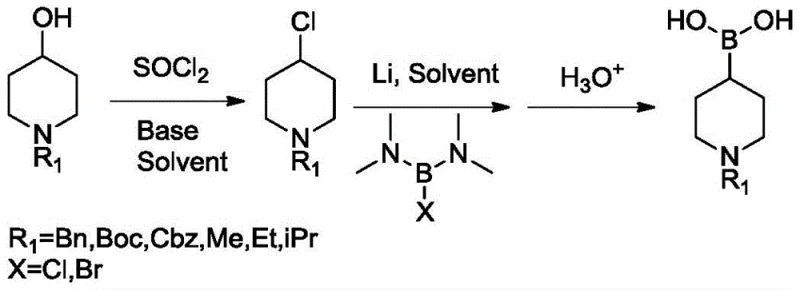
In stark contrast, the process disclosed in patent CN111171063B offers a transformative alternative that bypasses these inherent limitations. The new route begins with inexpensive N-substituted piperidine-4-alcohols, which are converted into the corresponding chlorides using thionyl chloride under mild conditions. This chlorination step is robust and scalable, avoiding the need for exotic reagents. The subsequent transformation involves a direct lithiation followed by reaction with bis(N,N-dimethylamino)haloborane to install the boronic acid functionality. This strategy completely eliminates the need for palladium catalysts and dangerous hydrogenation steps. The reaction conditions are温和 (mild), typically operating between -5°C and 20°C, which enhances operational safety and reduces energy consumption. By utilizing cheap, commodity-grade reagents like thionyl chloride and metallic lithium, the process achieves a drastic reduction in raw material costs while maintaining high product quality. This shift represents a fundamental optimization of the synthetic pathway, prioritizing safety and economics without compromising chemical integrity.
Mechanistic Insights into Lithiation-Borylation Strategy
The core of this innovative synthesis lies in the efficient generation of the organolithium intermediate and its subsequent trapping with a boron electrophile. In the second step of the process, metallic lithium reacts with the N-substituted piperidine-4-chloride in a solvent such as tetrahydrofuran (THF). This halogen-lithium exchange or direct insertion generates a highly reactive organolithium species at the 4-position of the piperidine ring. The presence of the N-protecting group (such as Boc, Cbz, or Benzyl) is crucial here, as it stabilizes the ring system and prevents unwanted side reactions at the nitrogen atom. Once formed, this nucleophilic carbon center immediately attacks the electrophilic boron atom in bis(N,N-dimethylamino)haloborane. The dimethylamino groups act as excellent leaving groups or stabilizing ligands during this transition, facilitating the formation of the carbon-boron bond. The reaction is carefully controlled at low temperatures (0-5°C) to minimize decomposition of the sensitive organolithium intermediate and to prevent over-reaction or polymerization. This precise control ensures that the boronic acid moiety is installed regioselectively, preserving the stereochemical integrity of the piperidine scaffold where applicable.
Impurity control is another critical aspect where this mechanism offers distinct advantages over palladium-catalyzed routes. In traditional methods, residual palladium can form stable complexes with the product or generate dehalogenated byproducts that are difficult to separate. In this lithium-mediated process, the primary byproducts are lithium salts and dimethylamine derivatives, which are water-soluble and easily removed during the acidic quench and aqueous workup. The quenching step involves adding dilute hydrochloric acid to adjust the pH to approximately 6, which protonates the boronate intermediate to the free boronic acid while dissolving inorganic salts. Subsequent extraction with organic solvents like ethyl acetate or toluene effectively separates the organic product from the aqueous waste stream. The final purification often involves a simple slurry or crystallization step using non-polar solvents like n-heptane, yielding a white solid with HPLC purity exceeding 98%. This clean impurity profile significantly reduces the burden on downstream purification processes, making it highly attractive for GMP manufacturing environments.
How to Synthesize N-Substituted Piperidine-4-Boronic Acid Efficiently
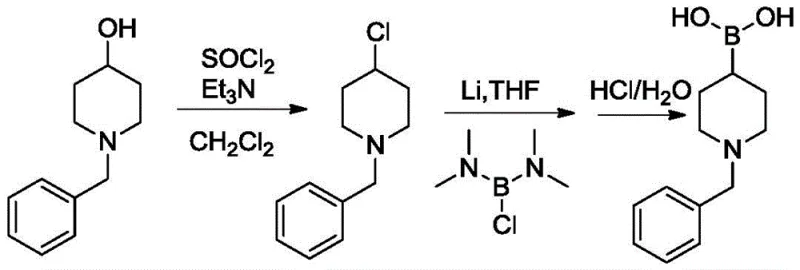
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to ensure optimal yield and safety. The process is divided into two distinct stages: the activation of the alcohol via chlorination and the subsequent metal-halogen exchange followed by borylation. Operators must ensure that the chlorination step is driven to completion to prevent unreacted alcohol from carrying over, which could interfere with the sensitive lithiation step. The use of anhydrous conditions in the second step is paramount, as moisture will rapidly quench the organolithium intermediate. Detailed standard operating procedures regarding the addition rates of thionyl chloride and the cooling capacity for the exothermic lithiation are essential for successful scale-up. For a comprehensive guide on the specific molar ratios, solvent choices, and workup protocols validated in the patent examples, please refer to the standardized synthesis steps outlined below.
- React N-substituted piperidine-4-alcohol with thionyl chloride and organic base in solvent at 20-40°C to obtain N-substituted piperidine-4-chlorine.
- React the resulting chloride with metallic lithium and bis(N,N-dimethylamino)haloborane in THF at -5 to 5°C.
- Quench with acidic aqueous solution, extract, and purify to obtain N-substituted piperidine-4-boronic acid with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic benefits that extend beyond simple unit price reductions. The shift away from precious metal catalysts and expensive specialized precursors fundamentally alters the cost structure of producing these key pharmaceutical intermediates. By relying on commodity chemicals like thionyl chloride and metallic lithium, the process insulates the supply chain from the volatility of the palladium market. Furthermore, the elimination of high-pressure hydrogenation equipment reduces capital expenditure requirements for manufacturing facilities and lowers the barrier for contract manufacturing organizations (CMOs) to produce these materials. The simplified workflow also translates to shorter cycle times and reduced utility consumption, contributing to a leaner and more responsive supply chain capable of meeting tight project deadlines.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of high-cost starting materials with affordable alternatives. The patent data indicates that the raw material cost for the alcohol precursor is a fraction of the cost of the tetrahydropyridine boronic acid used in conventional methods. Additionally, the removal of palladium hydroxide, a notoriously expensive catalyst, eliminates a major cost center. The overall yield of the two-step process is competitive, ensuring that material throughput remains high. When combined with the reduced need for extensive metal scavenging and purification steps, the total cost of goods sold (COGS) is significantly lowered. This allows pharmaceutical companies to allocate resources more efficiently across their development pipelines while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on single-source suppliers for complex, specialized reagents. This new method utilizes widely available chemicals that are produced by multiple global vendors, thereby diversifying the supply base and reducing the risk of shortages. The robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality or environmental factors. Moreover, the avoidance of hazardous hydrogenation simplifies logistics and storage requirements, as there is no need for specialized high-pressure gas infrastructure. This flexibility enables manufacturers to scale production up or down rapidly in response to clinical trial demands or commercial launch schedules without facing significant lead time penalties.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles. The absence of heavy metal catalysts reduces the generation of hazardous waste streams that require costly disposal and treatment. The solvents used, such as dichloromethane and THF, are standard industrial solvents with established recovery and recycling protocols. The mild reaction temperatures reduce energy consumption compared to high-temperature or high-pressure alternatives. As regulatory scrutiny on pharmaceutical manufacturing emissions intensifies, adopting a cleaner, metal-free synthesis route provides a proactive compliance advantage. This not only mitigates regulatory risk but also enhances the corporate sustainability profile of the final drug product, a factor increasingly valued by stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for R&D teams evaluating process transfer and procurement teams negotiating supply agreements. The focus is on clarifying the operational benefits and addressing potential concerns regarding scalability and purity standards.
Q: Why is the new synthesis method safer than conventional palladium-catalyzed hydrogenation?
A: The conventional method requires dangerous hydrogenation reactions using expensive palladium hydroxide catalysts. The novel process eliminates high-pressure hydrogenation entirely, utilizing mild lithiation conditions instead.
Q: What represents the primary cost advantage of this manufacturing route?
A: The primary advantage lies in raw material selection. Instead of using expensive N-substituted-1,2,5,6-tetrahydropyridine-4-boric acid precursors, this method utilizes significantly cheaper N-substituted piperidine-4-alcohols and thionyl chloride.
Q: What purity levels can be achieved with this process?
A: The process is designed to yield high-purity intermediates suitable for pharmaceutical applications, consistently achieving HPLC purity greater than 98% after standard workup and purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Piperidine-4-Boronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines. Our technical team has extensively analyzed the process described in CN111171063B and possesses the expertise to implement this advanced lithiation-borylation strategy at scale. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-substituted piperidine-4-boronic acid meets the highest industry standards. We are committed to delivering solutions that balance technical excellence with commercial practicality.
We invite you to collaborate with us to optimize your supply chain for these vital building blocks. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable, cost-effective supply of these complex intermediates, enabling you to focus on what matters most: bringing life-saving therapies to patients.
