Advanced Synthesis of Oxazoline Intermediates for High-Purity Nelfinavir Manufacturing
The pharmaceutical landscape for HIV treatment continues to evolve, demanding more efficient and cost-effective pathways for critical protease inhibitors like nelfinavir mesylate. Patent CN1390210A introduces a groundbreaking methodology for synthesizing key chemical intermediates, specifically focusing on the conversion of tetrahydrofuran derivatives into oxazoline compounds. This innovation addresses the longstanding need for streamlined processes in the production of antiretroviral therapies, offering a robust alternative to earlier, more cumbersome synthetic routes. By leveraging chiral amino-tetrahydrofuran starting materials, this technology enables the construction of complex molecular architectures with enhanced stereochemical control. For global supply chain leaders and R&D directors, understanding this patented approach is essential for securing reliable sources of high-purity pharmaceutical intermediates that meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nelfinavir and its free base relied heavily on precursors such as 1,3-dioxepan-5-ol or its derivatives, as disclosed in earlier patents like PCT/JP96/02756 (WO97/11937). These conventional pathways often involved multiple protection and deprotection steps, leading to increased material costs and extended processing times. The reliance on specific dioxepane scaffolds sometimes introduced challenges in stereochemical purity and overall yield, creating bottlenecks in large-scale manufacturing. Furthermore, the handling of certain intermediates in these older routes required rigorous condition controls to prevent racemization or degradation, adding complexity to the quality assurance protocols. These inefficiencies underscored the industry's demand for a more direct and economically viable synthetic strategy that could maintain high optical purity while reducing the total number of operational units.
The Novel Approach
The methodology described in CN1390210A represents a significant paradigm shift by utilizing tetrahydrofuran derivatives as the foundational scaffold for oxazoline formation. This novel approach allows for the direct conversion of amino-tetrahydrofuran compounds into oxazoline intermediates through a sequence involving acylation, sulfonylation, and electrophilic cyclization. By bypassing the need for dioxepane precursors, the process effectively reduces the step count and minimizes the accumulation of impurities associated with lengthy synthetic chains. The use of readily available chiral starting materials ensures that the stereochemical integrity is preserved throughout the transformation, which is critical for the biological activity of the final HIV inhibitor. This streamlined workflow not only enhances process efficiency but also aligns with modern green chemistry principles by potentially reducing solvent usage and waste generation.
Mechanistic Insights into Carbonyl-Philic Electrophile Promoted Cyclization
The core chemical transformation in this patent involves the intricate ring-opening of the tetrahydrofuran moiety followed by cyclization to form the oxazoline ring. This process is driven by the use of carbonyl-philic electrophiles, which act as powerful activators for the ether oxygen within the tetrahydrofuran ring. Suitable reagents include strong Lewis acids like titanium tetrachloride or protic acids such as trifluoromethanesulfonic acid, often used in combination with anhydrides. The mechanism proceeds through the generation of an acyl cation intermediate, which facilitates the nucleophilic attack by the amide nitrogen onto the activated carbon center. This results in the cleavage of the C-O bond in the tetrahydrofuran ring and the simultaneous formation of the C-N bond required for the oxazoline structure. The reaction conditions are carefully optimized to occur between -40°C and 70°C in aprotic solvents like ethyl acetate or toluene, ensuring high selectivity and minimizing side reactions.
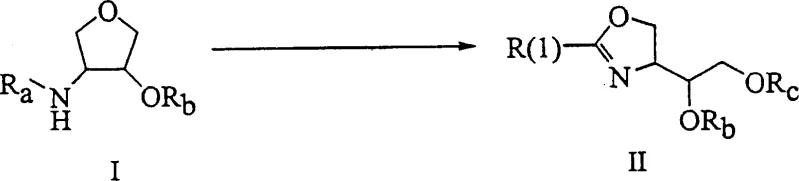
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this patent outlines specific strategies to manage potential byproducts. The selection of hydroxyl protecting groups, such as sulfonates (mesylates or tosylates), plays a crucial role in directing the regioselectivity of the ring-opening event. By converting the secondary hydroxyl group into a good leaving group prior to cyclization, the process ensures that the ring opens at the desired position to yield the correct regioisomer. Additionally, the use of excess molar equivalents of the electrophilic reagents helps drive the reaction to completion, reducing the levels of unreacted starting materials. The subsequent hydrolysis steps, typically performed using bases like potassium carbonate in methanol, are designed to remove acyl protecting groups without compromising the sensitive oxazoline ring, thereby delivering a clean product profile suitable for downstream coupling reactions.
How to Synthesize Oxazoline Intermediates Efficiently
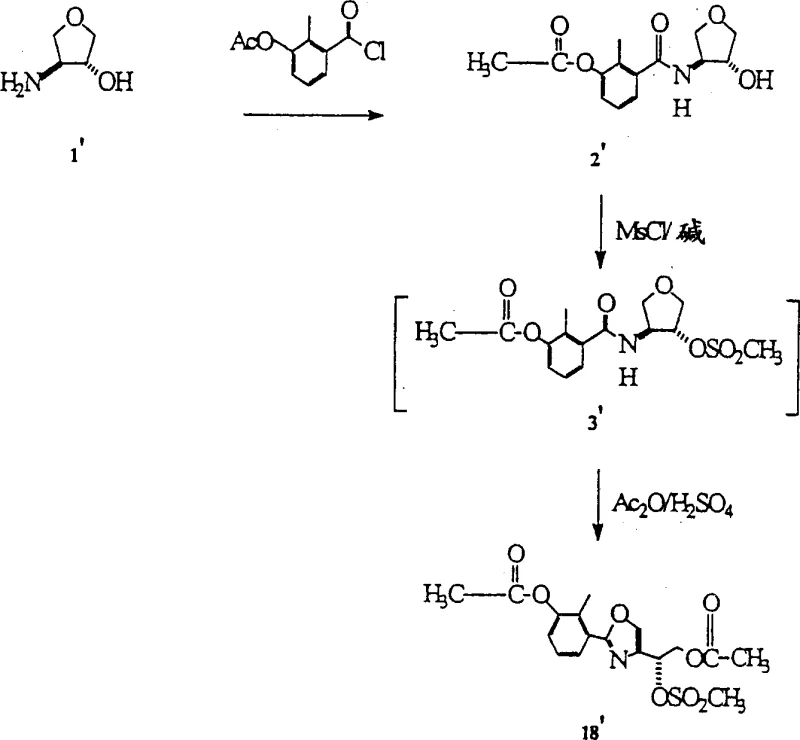
The practical execution of this synthesis involves a sequential protocol that begins with the acylation of the chiral amino-tetrahydrofuran starting material. This initial step establishes the amide linkage necessary for the subsequent cyclization, typically using acid chlorides like 3-acetoxy-2-methylbenzoyl chloride in the presence of a base. Following acylation, the secondary hydroxyl group is derivatized into a sulfonate ester, which serves as a pivotal intermediate for the ring-opening event. The final cyclization is achieved by treating the sulfonated amide with a mixture of acetic anhydride and a strong acid, promoting the formation of the oxazoline ring with high stereochemical fidelity. Detailed standardized synthesis steps for this specific pathway are provided in the guide below, offering a clear roadmap for technical teams looking to implement this technology.
- Acylation of chiral amino-tetrahydrofuran with an acid chloride (e.g., AMBC) to form the tetrahydrofuran-amide intermediate.
- Sulfonylation of the secondary hydroxyl group using methanesulfonyl chloride and a base like triethylamine to generate the sulfonate ester.
- Cyclization and ring-opening using a carbonyl-philic electrophile combination, such as acetic anhydride and sulfuric acid, to yield the oxazoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple chemical novelty. The streamlined nature of the process directly translates to reduced manufacturing lead times and lower operational overheads, as fewer unit operations are required to reach the key oxazoline intermediate. By utilizing common industrial solvents and reagents that are widely available in the global chemical market, the risk of supply disruption due to raw material scarcity is significantly mitigated. This reliability is crucial for maintaining continuous production schedules for life-saving antiretroviral medications, ensuring that patient needs are met without interruption. Furthermore, the robustness of the reaction conditions allows for greater flexibility in plant scheduling and resource allocation.
- Cost Reduction in Manufacturing: The elimination of complex precursor synthesis and the reduction in total step count lead to substantial cost savings in raw material consumption and labor. By avoiding the use of expensive and specialized dioxepane starting materials, manufacturers can leverage more economical tetrahydrofuran-based feedstocks. The simplified purification requirements associated with fewer intermediate isolations further contribute to a leaner cost structure, allowing for more competitive pricing in the final API market. This economic efficiency is vital for healthcare systems aiming to broaden access to HIV treatments.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetic anhydride, sulfuric acid, and standard organic solvents ensures a stable and resilient supply chain. Unlike proprietary reagents that may be sourced from single vendors, the inputs for this process are produced by multiple suppliers globally, reducing dependency risks. This diversification of the supply base enhances the overall security of the manufacturing pipeline, protecting against geopolitical or logistical disruptions. Consequently, partners can expect consistent delivery performance and improved inventory management capabilities.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to commercial scale. The use of standard equipment and the absence of extreme cryogenic or high-pressure requirements facilitate smooth technology transfer. Additionally, the potential for solvent recovery and the reduction in waste generation align with increasingly strict environmental regulations, supporting sustainable manufacturing practices. This compliance readiness minimizes regulatory hurdles and accelerates the time to market for new generic or branded formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazoline synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios. It clarifies the specific advantages over legacy methods and highlights the operational parameters required for success.
Q: What is the primary advantage of this tetrahydrofuran route over conventional methods?
A: This method eliminates the need for 1,3-dioxepan-5-ol precursors found in older patents like WO97/11937, resulting in fewer synthetic steps and potentially higher overall yields for nelfinavir production.
Q: What specific reagents drive the oxazoline ring formation?
A: The process utilizes carbonyl-philic electrophiles, preferably a combination of strong acids like sulfuric acid and anhydrides like acetic anhydride, to promote tetrahydrofuran ring opening and subsequent cyclization.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the reaction conditions utilize common industrial solvents such as ethyl acetate and toluene, and operate within a manageable temperature range of -40°C to 70°C, facilitating easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazoline Derivative Supplier
As the demand for high-quality HIV protease inhibitor intermediates grows, partnering with an experienced CDMO becomes a strategic imperative. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of oxazoline intermediate meets the highest international standards. We understand the critical nature of these supply chains and are committed to delivering products that support the uninterrupted manufacture of life-saving therapies.
We invite you to engage with our technical procurement team to discuss how this patented technology can be adapted to your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of technical excellence and supply chain security.
