Revolutionizing OLED Displays: Scalable Synthesis of High-Efficiency Platinum Photoluminescent Complexes
Revolutionizing OLED Displays: Scalable Synthesis of High-Efficiency Platinum Photoluminescent Complexes
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that balance high performance with economic viability and environmental sustainability. Patent CN111909219A introduces a breakthrough in this domain by disclosing a novel platinum compound with exceptional photoluminescence properties, specifically designed to serve as a high-efficiency emitter in next-generation display technologies. This innovation addresses the critical industry pain point of relying on expensive and scarce rare-earth metals like iridium, offering a robust alternative based on platinum coordination chemistry. The disclosed compound, characterized by the molecular formula C29H30ClN3O2Pt, exhibits a planar quadrilateral structure that facilitates strong spin-orbit coupling, essential for harvesting triplet excitons in OLED devices. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to diversify supply chains and reduce the cost of goods sold (COGS) in electronic chemical manufacturing without compromising on luminous efficacy or device lifetime.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of phosphorescent OLED materials has been dominated by organometallic complexes based on heavy rare-earth metals, particularly iridium (Ir). While these Ir(I) complexes theoretically achieve internal quantum efficiencies approaching 100% due to their ability to harvest both singlet and triplet excitons via strong spin-orbit coupling, they present substantial commercial drawbacks. The primary limitation is the exorbitant cost and geopolitical supply risk associated with iridium, a metal that is significantly rarer than platinum. Furthermore, the synthesis of high-performance iridium emitters often involves complex, multi-step pathways requiring harsh reaction conditions and difficult-to-remove transition metal catalysts, which complicates downstream purification and increases waste generation. These factors collectively hinder the mass adoption of iridium-based materials in cost-sensitive consumer electronics markets, creating an urgent demand for alternative emitters that can deliver comparable performance with a more favorable economic profile.
The Novel Approach
The methodology outlined in patent CN111909219A circumvents these traditional bottlenecks by leveraging the unique photophysical properties of bivalent platinum complexes. Unlike the conventional reliance on scarce iridium, this approach utilizes platinum, which, while still a precious metal, offers better availability and a more stable pricing structure for large-scale industrial applications. The novel synthesis route employs a strategic sequence of nucleophilic substitution, reduction, condensation, and oxidative cyclization to construct a rigid pyridoimidazole C^N ligand system. This ligand architecture is specifically engineered to coordinate with the platinum center in a way that maximizes radiative decay rates while minimizing non-radiative losses. By shifting the focus to platinum-based architectures, manufacturers can achieve a drastic simplification of the supply chain logic, reducing dependency on single-source rare earth suppliers and enabling more flexible production scheduling for high-volume display panel manufacturing.
Mechanistic Insights into Pt(II) Coordination and Photophysics
At the heart of this technology lies the precise coordination geometry of the platinum center, which dictates the electronic transitions responsible for light emission. The central platinum atom in the disclosed complex adopts a typical planar quadrilateral configuration, coordinating with carbon and nitrogen atoms from the primary pyridoimidazole ligand and two oxygen atoms from the auxiliary 2,2,6,6-tetramethyl-3,5-heptanedione (dpm) ligand. Crystallographic analysis reveals distinct bond lengths that stabilize this geometry: the Pt-O bonds measure approximately 1.998(9) Å and 2.083(8) Å, while the Pt-N and Pt-C bonds are recorded at 1.979(10) Å and 1.993(12) Å respectively. This specific spatial arrangement enforces a rigid molecular structure that suppresses vibrational relaxation pathways, thereby enhancing the photoluminescence quantum efficiency. The C-Pt-N bond angle of 80.0(5)° further indicates a slightly distorted square planar environment, which is crucial for mixing singlet and triplet states to allow for efficient phosphorescence at room temperature.
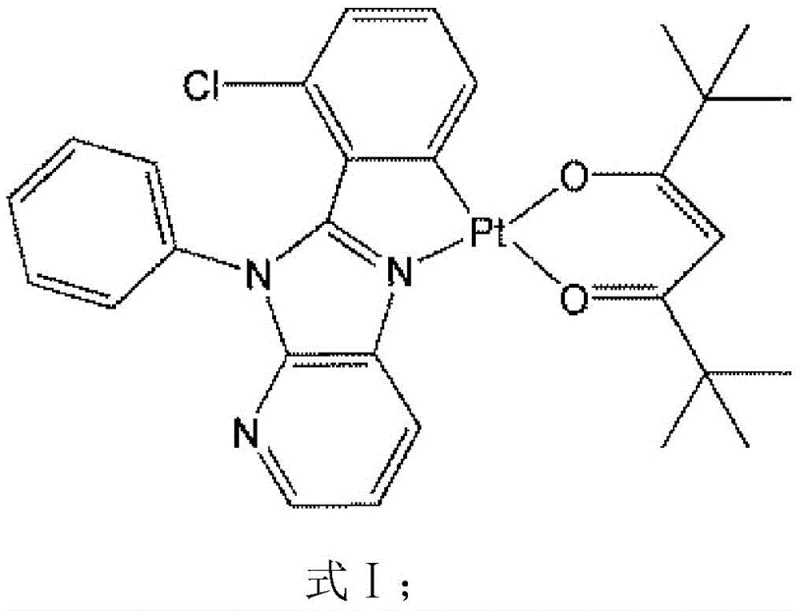
The photophysical mechanism involves a complex interplay of intraligand charge transfer (ILCT) and metal-to-ligand charge transfer (MLCT) transitions. Upon excitation at 329 nm, electrons are promoted from the ground state to higher energy orbitals, primarily localized on the pyridoimidazole moiety. The presence of the heavy platinum atom induces strong spin-orbit coupling, facilitating rapid intersystem crossing (ISC) from the singlet excited state to the triplet excited state. This process allows the material to harvest triplet excitons, which normally do not emit light in fluorescent materials, effectively doubling the theoretical internal quantum efficiency limit. The resulting emission occurs at 517 nm and 540 nm, corresponding to the green region of the visible spectrum, which is a critical color primary for full-color OLED displays. The observed lifetime of 9.53 μs and quantum efficiency of 22.08% in solid powder form confirm that the molecular design successfully balances radiative and non-radiative decay rates, making it a viable candidate for practical device integration.
How to Synthesize High-Purity Platinum OLED Emitters Efficiently
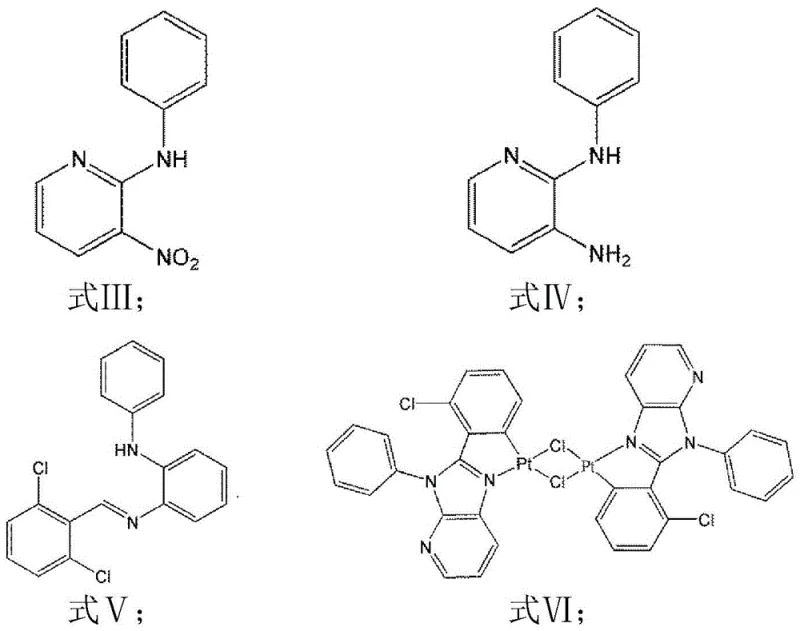
The preparation of this advanced luminescent material follows a logical, six-step synthetic pathway that prioritizes yield optimization and impurity control. The process begins with the construction of the organic ligand framework through classical organic transformations, followed by the introduction of the platinum metal center in the later stages to minimize metal contamination in early intermediates. Each step has been optimized for scalability, utilizing solvents like ethylene glycol and ethanol that are not only effective reaction media but also easily recoverable for reuse, aligning with green chemistry principles. The detailed operational parameters, including specific molar ratios, temperature gradients, and purification protocols such as column chromatography and recrystallization, are critical for achieving the reported 46-50% overall yield and high purity standards required for electronic grade materials. For a comprehensive breakdown of the standardized operating procedures (SOPs) required to replicate this synthesis in a GMP-compliant facility, please refer to the technical guide below.
- Synthesize Intermediate A by refluxing 2-chloro-3-nitropyridine and aniline in ethylene glycol at 60-65°C for 48-56 hours.
- Reduce Intermediate A using sodium dithionite in an ethanol/water mixture at 100-105°C to obtain Intermediate B.
- Condense Intermediate B with 2,6-dichlorobenzaldehyde in absolute ethanol under argon at 80°C to form Intermediate C.
- Oxidize Intermediate C with DDQ in dichloromethane at room temperature to generate the key Ligand L.
- Metallate Ligand L with potassium tetrachloroplatinate in ethylene glycol/water at 80°C to form Intermediate D.
- React Intermediate D with 2,2,6,6-tetramethylheptanedione and sodium carbonate in ethylene glycol at 130°C to yield the final platinum compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this platinum-based synthesis route offers profound advantages over legacy iridium technologies. The most significant benefit is the decoupling from the volatile rare-earth metal market, allowing procurement managers to forecast costs with greater accuracy and stability. By utilizing platinum, which has a more established and liquid global market compared to iridium, companies can mitigate supply chain risks associated with geopolitical tensions or mining disruptions. Furthermore, the synthetic pathway described in the patent is inherently designed for cost reduction in electronic chemical manufacturing through the use of commodity chemicals and recyclable solvents. The ability to recover and reuse solvents like ethylene glycol and dichloromethane significantly lowers the variable cost per kilogram of the final product, directly impacting the gross margin of the OLED panel production line.
- Cost Reduction in Manufacturing: The elimination of expensive iridium precursors and the use of readily available starting materials like 2-chloro-3-nitropyridine and aniline create a fundamentally lower cost base. Additionally, the process avoids the need for specialized, high-pressure equipment or extreme cryogenic conditions, relying instead on standard reflux setups that are ubiquitous in fine chemical plants. This simplicity translates to lower capital expenditure (CAPEX) for new production lines and reduced operational expenditure (OPEX) for existing facilities looking to diversify their product portfolio. The qualitative reduction in raw material complexity ensures that the cost of goods sold remains competitive even as production volumes scale up to meet the demands of the consumer electronics sector.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals for the ligand synthesis ensures a robust and redundant supply chain. Unlike specialized organometallic precursors that may have single-source suppliers, the building blocks for this platinum compound are produced by multiple global vendors, reducing the risk of stockouts. The moderate reaction temperatures (mostly between 60°C and 105°C) and ambient pressure conditions further enhance reliability by minimizing the risk of equipment failure or safety incidents that could disrupt production schedules. This operational stability is crucial for maintaining the continuous flow of materials required by just-in-time manufacturing models prevalent in the display industry.
- Scalability and Environmental Compliance: The synthesis protocol is highly amenable to scale-up, moving seamlessly from gram-scale laboratory experiments to multi-ton annual production capacities. The use of ethylene glycol and ethanol as primary solvents simplifies waste management, as these substances are biodegradable and easier to treat than halogenated alternatives often found in other organometallic syntheses. Moreover, the patent explicitly highlights the recyclability of the solvent system, which not only reduces environmental impact but also aligns with increasingly stringent global regulations on volatile organic compound (VOC) emissions. This environmental compliance future-proofs the supply chain against regulatory tightening, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this material into their existing workflows, we have compiled answers to common inquiries regarding its performance and handling. These insights are derived directly from the experimental data and characterization results presented in the patent documentation, ensuring accuracy and relevance for process engineers and quality assurance professionals. Understanding these technical nuances is essential for defining the correct specifications for incoming raw materials and establishing appropriate quality control checkpoints during the manufacturing process.
Q: What are the photoluminescence properties of this platinum compound?
A: The compound exhibits a maximum excitation wavelength of 329 nm and dual emission peaks at 517 nm and 540 nm. It demonstrates a solid-state photoluminescence quantum efficiency of 22.08% at 293K with a lifetime of 9.53 μs, making it suitable for green-emitting OLED applications.
Q: How does this platinum complex compare to traditional iridium-based emitters?
A: While iridium complexes offer high efficiency, they suffer from high costs and environmental concerns due to the rarity of iridium. This platinum-based alternative utilizes more abundant platinum metal, offers a simpler synthesis pathway with recyclable solvents, and provides competitive luminescent performance at a potentially lower cost basis.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes common organic solvents like ethylene glycol, ethanol, and dichloromethane, which can be recovered and reused. The reaction conditions are relatively mild (mostly below 100°C, except for the final step), and the purification involves standard techniques like column chromatography and recrystallization, facilitating scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Platinum OLED Material Supplier
As the global demand for high-resolution, energy-efficient displays continues to surge, the need for reliable sources of advanced emissive materials has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring innovations like the CN111909219A platinum complex to the market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of high-purity OLED material meets the exacting standards required for premium display applications. We understand that consistency is key in the semiconductor and display industries, and our robust quality management systems are designed to deliver batch-to-batch reproducibility that safeguards your production yields.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this novel platinum compound can optimize your material costs and enhance your product performance. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive the next generation of OLED technology forward with confidence and precision.
