Advanced Silver-Catalyzed Synthesis of Dibenzophospholane for High-Performance Optoelectronic Applications
The rapidly evolving landscape of organic electronics demands robust and scalable synthetic routes for high-performance phosphorus-containing heterocycles. Patent CN115677766A introduces a groundbreaking preparation method for dibenzophospholane compounds, which serve as critical building blocks for next-generation photoelectric functional materials. This innovation addresses the longstanding industry challenge of balancing high purity with economic feasibility by utilizing a silver nitrate catalytic system in an ethanol solvent medium. Unlike traditional approaches that rely on expensive transition metals and hazardous solvents, this methodology leverages the unique reactivity of silver to facilitate efficient cyclization under mild conditions. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing of OLED intermediates and related optoelectronic components. The technical breakthrough lies not only in the catalyst substitution but also in the comprehensive optimization of reaction parameters that ensure consistent quality and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dibenzophosphole derivatives has been plagued by significant technical and economic hurdles that hinder large-scale commercial adoption. Conventional methodologies predominantly rely on palladium-based catalytic systems, such as palladium acetate, which necessitate the use of toxic and expensive aprotic solvents like tetrahydrofuran (THF) or dimethylformamide (DMF). These processes often require elevated temperatures ranging from 65°C to 120°C and extended reaction times spanning from 3 to 24 hours to achieve acceptable conversion rates. Furthermore, the use of palladium introduces severe downstream processing challenges, including the absolute necessity for rigorous heavy metal removal steps to meet the stringent purity specifications required for electronic grade materials. The combination of high catalyst costs, hazardous solvent handling, and complex purification workflows results in a manufacturing process that is both economically inefficient and environmentally burdensome for industrial supply chains.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes silver nitrate as a highly efficient and economically accessible catalyst to drive the cyclization of biphenyl phosphine oxide compounds. This method operates effectively in alcohol solvents, with ethanol being the preferred medium due to its low toxicity and favorable solvation properties. The reaction conditions are remarkably mild, typically proceeding at temperatures between 30°C and 78°C, and achieving completion within a concise timeframe of 0.5 to 5 hours. By eliminating the need for precious palladium catalysts and hazardous organic solvents, this new route significantly simplifies the operational workflow and reduces the overall environmental footprint. The structural versatility of this method allows for a wide range of substituents on the biphenyl backbone, as illustrated by the general formula of the starting materials below, ensuring broad applicability across different molecular designs.
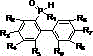
This strategic shift in reagent selection not only lowers the barrier to entry for production but also enhances the safety profile of the entire manufacturing facility, making it an ideal candidate for continuous process improvement initiatives.
Mechanistic Insights into Silver-Catalyzed Cyclization
The mechanistic pathway of this silver-catalyzed transformation involves the activation of the phosphorus-hydrogen bond and subsequent intramolecular cyclization to form the rigid dibenzophospholane core. Silver ions act as a Lewis acid to coordinate with the phosphine oxide moiety, thereby increasing the electrophilicity of the phosphorus center and facilitating the nucleophilic attack by the adjacent aromatic ring. This interaction promotes the cleavage of the C-H bond on the phenyl ring and the formation of the new C-P bond, resulting in the fused heterocyclic structure. The use of ethanol as a protic solvent likely plays a dual role by stabilizing the transition state through hydrogen bonding and assisting in the proton transfer steps necessary for the aromatization of the final product. Understanding this mechanism is crucial for process chemists aiming to fine-tune reaction parameters for specific derivatives, as the electronic nature of substituents on the aromatic rings can significantly influence the rate of cyclization and the overall yield of the desired photoelectric material.
From an impurity control perspective, the absence of palladium eliminates the risk of metal contamination which is a critical failure mode in organic light-emitting diode (OLED) fabrication. Residual palladium can act as a quenching site for excitons, drastically reducing the efficiency and lifespan of the final electronic device. The silver-catalyzed route inherently produces a cleaner crude reaction mixture, which simplifies the purification process to standard column chromatography or crystallization techniques. The final product structure, characterized by the fused phosphole ring system shown below, exhibits enhanced thermal stability and tunable electronic properties due to the interaction between the phosphorus d-orbitals and the pi-conjugated system. This structural integrity is paramount for ensuring the long-term reliability of the material in demanding optoelectronic applications.
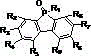
How to Synthesize Dibenzophospholane Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard laboratory and pilot plant settings without requiring specialized high-pressure equipment. The protocol involves a straightforward mixing of the biphenyl phosphine oxide substrate with silver nitrate in ethanol, followed by heating to the optimal temperature range identified in the patent examples. Reaction monitoring can be easily performed using standard analytical techniques such as TLC or HPLC to determine the endpoint, typically achieved within 1 to 3 hours for most substrates. Upon completion, the workup procedure is uncomplicated, involving concentration under reduced pressure and purification to isolate the high-purity target compound. For detailed operational parameters and specific stoichiometric ratios tailored to your specific derivative, please refer to the standardized synthesis guide provided below.
- Charge a reaction vessel with biphenyl phosphine oxide compound and silver nitrate catalyst in a 1: 1 molar ratio.
- Add ethanol as the organic solvent and stir the mixture uniformly to ensure complete dissolution and contact.
- Heat the reaction system to a temperature between 30°C and 78°C and maintain for 1 to 3 hours to achieve cyclization.
- Upon completion, cool the mixture to room temperature, concentrate under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this silver-catalyzed technology offers profound strategic advantages that extend far beyond simple chemical synthesis. The transition from palladium to silver catalysts represents a direct and substantial reduction in raw material expenditure, as silver nitrate is significantly more abundant and cost-effective than palladium salts. Additionally, the elimination of toxic solvents like THF and DMF reduces the costs associated with solvent procurement, storage, and hazardous waste disposal, leading to a leaner and more compliant operational model. The simplified downstream processing further contributes to cost efficiency by removing the need for expensive metal scavenging resins and reducing the cycle time for batch turnover. These factors collectively enhance the overall margin potential for manufacturers of electronic chemicals while mitigating supply chain risks associated with volatile precious metal markets.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with economical silver nitrate drives a significant decrease in the bill of materials for every production batch. Furthermore, the use of ethanol as a solvent eliminates the premium pricing associated with anhydrous aprotic solvents and reduces the energy consumption required for solvent drying and recovery. The streamlined purification process also lowers labor and consumable costs, as fewer unit operations are required to achieve the necessary purity levels. This holistic approach to cost optimization ensures that the final product remains competitive in the global market without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like silver nitrate and ethanol insulates the production process from the supply volatility often seen with specialized transition metal catalysts. Ethanol is universally available in bulk quantities, ensuring uninterrupted production schedules even during periods of regional logistical constraints. The robustness of the reaction conditions, which tolerate a wide range of temperatures and concentrations, further enhances process reliability and reduces the risk of batch failures due to minor parameter deviations. This stability is critical for maintaining consistent delivery timelines to downstream customers in the fast-paced electronics industry.
- Scalability and Environmental Compliance: The mild reaction conditions and non-toxic solvent system make this process inherently scalable from kilogram to multi-ton production volumes with minimal engineering modifications. The reduced environmental impact aligns perfectly with increasingly stringent global regulations regarding chemical manufacturing and VOC emissions. By adopting this greener synthesis route, companies can proactively meet sustainability goals and avoid potential regulatory fines or shutdowns. The ease of waste treatment for ethanol-based streams further simplifies the environmental compliance workflow, making it a future-proof solution for long-term industrial operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation to provide clarity for potential adopters. Understanding these nuances is essential for making informed decisions about integrating this method into existing production pipelines or developing new product lines based on dibenzophospholane derivatives.
Q: What are the primary advantages of using silver nitrate over palladium catalysts for this synthesis?
A: The use of silver nitrate offers significant economic and operational advantages compared to traditional palladium catalysts. Silver salts are substantially less expensive than palladium acetate or complexes, directly reducing raw material costs. Furthermore, silver catalysis avoids the stringent requirement for expensive heavy metal scavengers during purification, simplifying the downstream processing and ensuring higher purity profiles suitable for electronic applications.
Q: How does the choice of ethanol as a solvent impact the environmental profile of the process?
A: Replacing toxic aprotic solvents like THF or DMF with ethanol drastically improves the environmental sustainability of the manufacturing process. Ethanol is a low-toxicity, bio-based solvent that simplifies waste treatment protocols and reduces the regulatory burden associated with volatile organic compound (VOC) emissions. This switch aligns the production process with modern green chemistry principles and facilitates easier solvent recovery and recycling.
Q: What yields can be expected from this silver-catalyzed method compared to prior art?
A: Experimental data indicates that this novel method achieves yields ranging from 72% to 88%, depending on specific substituents and reaction temperatures. This performance is competitive with or superior to existing methods that often require harsher conditions or longer reaction times. Specifically, the optimization of temperature around 50°C to 78°C in ethanol consistently delivers high conversion rates with minimal byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzophospholane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this silver-catalyzed technology in advancing the field of organic electronics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of phosphorus chemistry, maintaining stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. We are committed to delivering high-quality dibenzophospholane intermediates that meet the exacting standards of the global optoelectronic industry.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener and more efficient process. Please contact our technical procurement team today to request specific COA data for our current inventory and to discuss route feasibility assessments tailored to your project timeline and volume requirements.
