Revolutionizing p-Vinylbenzoic Acid Production with Advanced Palladium Catalysis for Global Markets
Revolutionizing p-Vinylbenzoic Acid Production with Advanced Palladium Catalysis for Global Markets
The landscape of fine organic synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective manufacturing processes for critical intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN115888844A, which discloses a novel palladium catalyst and its application in the synthesis of p-vinylbenzoic acid. This compound serves as a vital building block in the fields of organic synthesis, material chemistry, and biomedical research, finding applications in everything from metal-organic frameworks to tumor suppression agents. The patent introduces a specialized catalyst, [bis(4-(trifluoromethyl)phenyl)phosphonite ferrocene]palladium dichloride, which enables a highly selective Hiyama cross-coupling reaction. Unlike traditional methods that suffer from harsh conditions or low yields, this innovation offers a robust pathway characterized by mild reaction parameters, simplified equipment requirements, and exceptional product quality. For global procurement and R&D teams, understanding this technological shift is crucial for securing a reliable p-vinylbenzoic acid supplier capable of meeting the rigorous demands of modern pharmaceutical and electronic material manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-vinylbenzoic acid has been plagued by significant technical and economic hurdles that hinder large-scale industrial adoption. One of the earliest reported methods involves a Wittig reaction using p-chloromethylbenzoic acid and triphenylphosphine, followed by reaction with formaldehyde under alkaline conditions. As illustrated in the reaction scheme below, this route is inherently inefficient, requiring large excesses of formaldehyde and sodium hydroxide, which generates substantial wastewater and complicates environmental compliance.
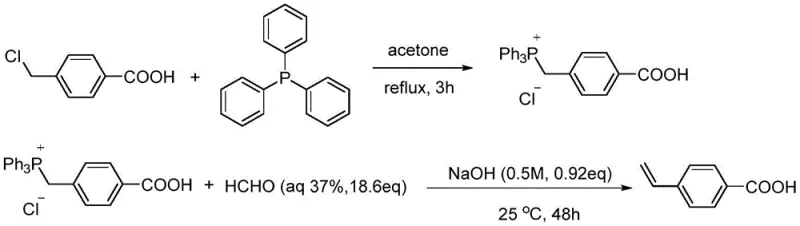
Furthermore, the yield of this Wittig pathway is notoriously poor, often capping at merely 59%, which is economically unsustainable for high-volume production. Alternative approaches, such as the copper-catalyzed carboxylation of vinylboronates with carbon dioxide, introduce different sets of challenges including the difficulty in sourcing specialized raw materials like 5,5-dimethyl-2-(4-vinylphenyl)-1,3,2-dioxaborinane and the necessity for high-pressure reaction vessels. Similarly, Heck-Mizoroki reactions using ethylene gas require extreme temperatures and pressures, posing serious safety risks and operational complexities that make them unsuitable for safe, continuous manufacturing environments. These legacy methods collectively represent a bottleneck in the supply chain, driving up costs and limiting the availability of high-purity materials.
The Novel Approach
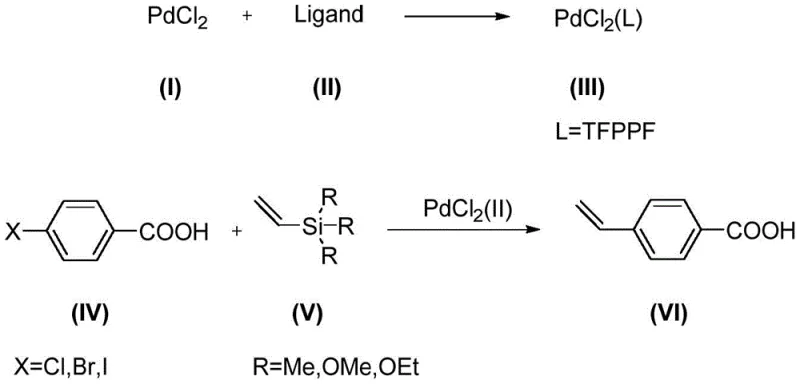
In stark contrast to these cumbersome legacy processes, the technology disclosed in CN115888844A presents a paradigm shift through the utilization of a tailored Hiyama cross-coupling strategy. This novel approach leverages a specifically designed palladium catalyst that facilitates the coupling of readily available p-halobenzoic acids with versatile vinylsilane compounds. The reaction proceeds under remarkably mild conditions, typically between 40-100°C, eliminating the need for the dangerous high-pressure setups required by previous methods. The core of this innovation lies in the catalyst's unique ligand structure, which enhances chemoselectivity and reaction rates without the need for additional activators that could contaminate the final product. This streamlined methodology not only simplifies the operational workflow but also dramatically improves the overall yield and purity profile, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ferrocene-Based Palladium Catalysis
To fully appreciate the technical superiority of this synthesis route, one must delve into the mechanistic intricacies of the ferrocene-based palladium catalytic cycle. The catalyst, [bis(4-(trifluoromethyl)phenyl)phosphonite ferrocene]palladium dichloride, is formed by coordinating the electron-rich ferrocene backbone with trifluoromethyl-substituted phosphonite ligands. This specific electronic configuration stabilizes the palladium center, allowing it to efficiently undergo the critical oxidative addition step with the aryl halide bond of the p-halobenzoic acid substrate. Following oxidative addition, the catalytic cycle proceeds through a transmetallation phase where the vinyl group from the silane reagent is transferred to the palladium center. Unlike many other cross-coupling reactions that require aggressive fluoride activators to break the strong silicon-carbon bond, this specific catalyst system is active enough to facilitate transmetallation under milder conditions, thereby preserving the integrity of sensitive functional groups.
The final stages of the mechanism involve cis-trans isomerization and reductive elimination, which release the desired p-vinylbenzoic acid product and regenerate the active palladium species for the next cycle. This efficient turnover is key to the high yields observed in the patent examples, often exceeding 80% even on multi-kilogram scales. Moreover, the robustness of this catalytic system contributes significantly to impurity control. By minimizing side reactions such as homocoupling or dehalogenation, the process ensures a cleaner reaction profile. The ability to recover and recycle the palladium catalyst from the reaction mixture further underscores the efficiency of this mechanism, as the catalyst remains stable throughout the process and can be separated via simple filtration and washing steps, reducing both metal residue in the product and the overall consumption of precious metals.
How to Synthesize p-Vinylbenzoic Acid Efficiently
The practical implementation of this synthesis route is designed for ease of operation, utilizing a convenient one-pot strategy that minimizes unit operations and solvent usage. The process begins with the in-situ generation of the active palladium catalyst by heating the ligand and palladium dichloride in THF, followed directly by the addition of the acid and silane substrates without intermediate isolation. This telescoped approach reduces handling time and exposure to air or moisture, which can degrade sensitive intermediates. For R&D teams looking to replicate or adapt this chemistry, the detailed standardized synthesis steps are provided in the guide below, ensuring reproducibility and adherence to the optimal parameters defined in the patent literature.
- Prepare the palladium catalyst by reacting ferrocene bis(4-(trifluoromethyl)phenyl)phosphonite with palladium dichloride in THF at 60-80°C for 6-10 hours.
- Without isolating the catalyst, add p-halobenzoic acid and vinylsilane compounds directly to the reaction mixture for the Hiyama cross-coupling.
- Heat the mixture to 40-100°C for 4-8 hours, then cool, filter to recover the catalyst, and purify the product via acidification and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel catalytic process represents a strategic opportunity to optimize costs and secure supply continuity. The shift away from high-pressure, hazardous reactions to ambient or mild pressure systems significantly reduces the capital expenditure required for specialized reactor infrastructure. Furthermore, the elimination of expensive and difficult-to-handle activators simplifies the raw material basket, relying instead on commodity chemicals like p-chlorobenzoic acid and trimethylvinylsilane which are widely available in the global market. This accessibility mitigates the risk of supply disruptions and provides a buffer against price volatility for niche reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the dramatic improvement in reaction yield and the recyclability of the catalyst system. By achieving yields consistently above 80% compared to the sub-60% yields of older Wittig routes, manufacturers can significantly reduce the cost per kilogram of the final active ingredient. Additionally, the ability to recover and reuse the palladium catalyst multiple times without significant loss of activity means that the consumption of this precious metal is drastically minimized. This reduction in catalyst loading, combined with the avoidance of costly purification steps needed to remove heavy metal residues from less selective reactions, translates into substantial cost savings in p-vinylbenzoic acid manufacturing.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of stable, non-gaseous reagents like vinylsilanes instead of ethylene gas removes the complexities associated with storing and transporting high-pressure gases. The reaction conditions are forgiving and do not require strictly anhydrous or oxygen-free environments, which simplifies warehouse and production line requirements. This robustness ensures that production schedules are less likely to be interrupted by minor environmental fluctuations or equipment failures, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring a steady flow of materials to downstream customers.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than conventional methods, aligning with modern green chemistry principles. The process generates significantly less wastewater compared to the alkaline-heavy Wittig reaction, and the solvents used, such as THF or DMF, can be effectively recovered and recycled through standard distillation protocols. The reduced generation of hazardous waste lowers disposal costs and simplifies regulatory compliance. Furthermore, the mild thermal conditions reduce energy consumption for heating and cooling, making the commercial scale-up of complex organic intermediates more sustainable and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to industry standards.
Q: What are the primary advantages of this new palladium catalyst over traditional methods?
A: The novel [bis(4-(trifluoromethyl)phenyl)phosphonite ferrocene]palladium dichloride catalyst eliminates the need for harsh activators like fluoride salts, operates under milder temperatures compared to Heck reactions, and achieves significantly higher yields (over 80%) with superior chemical selectivity.
Q: Is the catalyst recyclable for industrial scale-up?
A: Yes, the process allows for efficient catalyst recovery. After the reaction, the solid filter cake containing the palladium catalyst can be washed, dried, and reused, which drastically reduces heavy metal waste and lowers long-term production costs.
Q: How does this method improve product purity for pharmaceutical applications?
A: By avoiding high-pressure conditions and unstable intermediates found in older routes, this Hiyama coupling minimizes side reactions. The resulting crude product has high purity (often >99% by HPLC) which simplifies downstream purification and ensures compliance with stringent API intermediate specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Vinylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of cutting-edge synthetic methodologies is essential for maintaining competitiveness in the global fine chemicals market. Our team of expert chemists has thoroughly analyzed the potential of the ferrocene-based palladium catalytic system described in CN115888844A and is well-positioned to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of p-vinylbenzoic acid meets the highest standards required for pharmaceutical and electronic applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific volume and quality requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your product portfolio.
