Revolutionizing Organic Luminescent Material Production via Copper-Catalyzed Cyclization
Revolutionizing Organic Luminescent Material Production via Copper-Catalyzed Cyclization
The landscape of organic optoelectronic material synthesis is undergoing a significant transformation driven by the urgent need for safer, more cost-effective, and scalable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN115703810A, which discloses a novel preparation method for benzo phosphorus heterocycle pentadiene compounds, specifically benzo[b]phosphole oxides. These compounds serve as critical building blocks for next-generation organic light-emitting diodes (OLEDs), solar cells, and fluorescent probes due to their unique optical and electrochemical properties. The core innovation lies in the utilization of copper acetate as a simple, inexpensive, and highly efficient catalyst to drive the intramolecular cyclization of 2-alkynyl phosphine oxide derivatives. This technical breakthrough addresses long-standing inefficiencies in the field, offering a robust pathway for the reliable electronic chemical supplier market to meet the escalating demand for high-performance luminescent materials without compromising on safety or economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzophosphacyclopentadiene scaffolds has been plagued by severe operational hazards and complex procedural requirements that hinder large-scale adoption. Traditional routes often rely on the generation of dilithium transition state intermediates using n-butyllithium, a reagent that is not only extremely unstable and pyrophoric but also necessitates rigorous cryogenic conditions, typically requiring reaction temperatures well below zero degrees Celsius. Alternative methods involving diisobutylaluminum hydride (DIBAL-H) followed by bromination and subsequent lithiation further complicate the process with multiple steps and the handling of hazardous aluminum species. Furthermore, earlier catalytic approaches utilizing silver oxide or zinc nitrate often suffer from high catalyst loading costs and potential heavy metal contamination issues, which are detrimental to the purity standards required in electronic applications. These conventional pathways are characterized by low atom economy, harsh reaction environments, and significant safety liabilities, making them ill-suited for the cost reduction in OLED material manufacturing that modern industry demands.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN115703810A introduces a streamlined, copper-catalyzed cyclization strategy that fundamentally simplifies the synthetic architecture. By employing readily available copper acetate as the sole catalyst, the process eliminates the need for cryogenic cooling and highly reactive organometallic reagents, allowing the reaction to proceed efficiently at elevated temperatures ranging from 100°C to 155°C. The versatility of this approach is underscored by its compatibility with common industrial solvents such as toluene and dimethylformamide (DMF), which have been shown to deliver superior yields compared to other solvent systems. This shift from stoichiometric, hazardous reagents to a catalytic system represents a paradigm shift in process chemistry, enabling the commercial scale-up of complex polymer additives and electronic intermediates with dramatically improved safety profiles and operational simplicity. The ability to achieve high conversion rates in short reaction times (30 to 60 minutes) further underscores the efficiency of this novel catalytic cycle.
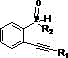
Mechanistic Insights into Copper-Catalyzed Intramolecular Cyclization
The mechanistic underpinning of this transformation involves the activation of the alkyne moiety within the 2-alkynyl phosphine oxide derivative by the copper catalyst, facilitating an intramolecular nucleophilic attack or insertion that closes the five-membered phosphorus-containing ring. Unlike base-mediated cyclizations that rely on deprotonation and can lead to various side reactions due to the high basicity of reagents like t-BuOK, the copper-catalyzed pathway likely proceeds through a coordination-insertion mechanism that is both chemoselective and mild. This selectivity is crucial for maintaining the integrity of sensitive functional groups on the aryl rings, such as halogens or alkoxy substituents, which are often necessary for tuning the electronic properties of the final luminescent material. The use of copper acetate, a mild Lewis acid, ensures that the reaction environment remains controlled, minimizing the formation of polymeric byproducts or decomposition products that frequently plague high-temperature organic syntheses. This precise control over the reaction trajectory is essential for producing high-purity OLED material intermediates that meet the stringent specifications of the display industry.
From an impurity control perspective, the absence of lithium or aluminum salts in the reaction mixture significantly simplifies the downstream workup and purification processes. In traditional lithiation methods, the quenching of excess organolithium reagents generates substantial amounts of inorganic salts that can be difficult to remove completely, potentially affecting the charge transport properties of the final device. The copper-catalyzed method described herein avoids these inorganic contaminants almost entirely, resulting in a cleaner crude product profile. This reduction in impurity burden not only enhances the overall yield of the isolated product but also reduces the consumption of silica gel and solvents during column chromatography, contributing to a more sustainable and environmentally friendly manufacturing process. The structural stability of the resulting benzophosphole oxide core, as depicted in the product structure, ensures that the material can withstand subsequent chemical modifications or device fabrication processes without degradation.
![Target benzo[b]phosphole oxide product structure showing the stable heterocyclic core](/insights/img/benzophosphole-oxide-synthesis-oled-supplier-20260307212136-01.png)
How to Synthesize Benzo[b]phosphole Oxide Efficiently
The implementation of this copper-catalyzed protocol offers a straightforward and reproducible route for laboratories and production facilities aiming to access these valuable optoelectronic intermediates. The process begins with the dissolution of the 2-alkynyl phosphine oxide substrate in a suitable organic solvent, preferably toluene or DMF, followed by the addition of a catalytic amount of copper acetate. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios, temperature gradients, and workup procedures optimized to maximize yield and purity while minimizing waste. By adhering to these parameters, manufacturers can consistently produce material that meets the rigorous quality standards required for high-end electronic applications.
- Combine 2-alkynyl phosphine oxide derivatives with copper acetate catalyst (0.1-10 mol%) in a reaction vessel containing toluene or DMF solvent.
- Heat the reaction mixture to a temperature range of 100-155°C and maintain stirring for 30 to 60 minutes to facilitate intramolecular cyclization.
- Upon completion, cool the system to room temperature, concentrate under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed synthesis route presents a compelling value proposition centered on cost efficiency, supply security, and operational scalability. The transition from expensive and hazardous reagents like n-butyllithium or silver oxide to commodity-grade copper acetate results in a drastic reduction in raw material costs, directly impacting the bottom line of manufacturing operations. Furthermore, the elimination of cryogenic infrastructure requirements lowers capital expenditure (CAPEX) for new production lines and reduces energy consumption associated with maintaining low-temperature reactors. This process intensification allows for faster batch turnover times, enhancing the overall throughput of the facility and ensuring a more responsive supply chain capable of meeting fluctuating market demands for organic luminescent materials.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts and stoichiometric organometallic reagents with inexpensive copper acetate drives significant cost savings in the bill of materials. Additionally, the simplified workup procedure reduces the consumption of purification media and solvents, lowering the variable costs per kilogram of produced intermediate. The avoidance of specialized low-temperature equipment further decreases maintenance and operational expenses, making the overall process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: Copper acetate and the required solvents (toluene, DMF) are globally available commodity chemicals with robust supply chains, mitigating the risk of raw material shortages that often plague specialty reagent markets. The stability of the reagents allows for easier storage and handling, reducing the logistical complexities and safety regulations associated with transporting pyrophoric materials like n-butyllithium. This reliability ensures consistent production schedules and reduces the lead time for high-purity electronic chemical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals like silver or excessive alkali waste streams align perfectly with modern environmental, health, and safety (EHS) standards. The process generates less hazardous waste, simplifying disposal and reducing the environmental footprint of the manufacturing site. Its proven efficiency at gram scales in the patent examples suggests a smooth path to ton-scale production, supporting the commercial expansion of advanced display and lighting technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is becoming the preferred choice for forward-thinking chemical manufacturers. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: What are the primary advantages of using copper acetate over traditional n-butyllithium reagents?
A: Traditional methods utilizing n-butyllithium require cryogenic conditions (deep cooling) and involve highly unstable, pyrophoric reagents that pose significant safety risks. In contrast, the copper acetate-catalyzed method operates at moderate temperatures (100-155°C) using stable, inexpensive, and easily handled catalysts, drastically improving operational safety and reducing equipment costs associated with low-temperature reactors.
Q: Which solvents provide the optimal yield for this cyclization reaction?
A: According to the experimental data in patent CN115703810A, polar aprotic solvents such as DMF and aromatic solvents like toluene demonstrate superior performance. Specifically, reactions conducted in toluene at 100°C and DMF at 150°C achieved yields exceeding 87%, significantly outperforming other solvents like acetonitrile or DMSO which resulted in lower conversion rates.
Q: How does this method impact the purity profile of the final luminescent material?
A: By eliminating the need for stoichiometric amounts of strong bases like potassium tert-butoxide or organolithium reagents, this catalytic approach minimizes the formation of inorganic salt byproducts and side-reaction impurities. The cleaner reaction profile simplifies downstream purification, leading to high-purity intermediates essential for high-performance OLED and optoelectronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo[b]phosphole Oxide Supplier
As the global demand for high-performance organic luminescent materials continues to surge, partnering with a technically proficient manufacturer is essential for securing a competitive edge in the marketplace. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in heterogeneous catalysis and process optimization to deliver superior quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of benzo[b]phosphole oxide meets the exacting standards required for OLED and optoelectronic applications.
We invite you to collaborate with us to explore how this innovative copper-catalyzed technology can enhance your product portfolio and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term strategic goals in the electronic materials sector.
