Scalable Copper-Catalyzed Synthesis of Benzo-Phosphole Compounds for Advanced Optoelectronic Applications
The landscape of organic optoelectronic material synthesis is undergoing a significant transformation, driven by the urgent need for safer, more cost-effective, and scalable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN115703809A, which discloses a novel preparation method for benzo-phosphole pentadiene compounds. These heterocyclic structures are critical building blocks for high-performance organic light-emitting diodes (OLEDs), solar cells, and fluorescent probes due to their unique optical and electrochemical properties. The patent introduces a streamlined synthetic route that utilizes 2-alkynyl phosphine oxide derivatives as substrates and copper acetate as a catalyst, marking a departure from traditional, hazard-prone methodologies. By leveraging earth-abundant transition metal catalysis, this innovation addresses key bottlenecks in the supply chain of high-purity electronic chemicals, offering a robust pathway for reliable photoelectric material suppliers to meet the escalating demands of the display and energy sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzo-phosphole frameworks has been plagued by severe operational challenges and safety hazards inherent to organometallic chemistry. Traditional routes often rely on the generation of dilithium transition state compounds using n-butyllithium (n-BuLi), a pyrophoric reagent that necessitates rigorous cryogenic conditions, typically below -78°C, to maintain stability. This requirement for extreme低温 not only imposes a heavy energy burden on the manufacturing facility but also introduces significant safety risks regarding thermal runaway and reagent handling. Furthermore, alternative methods utilizing silver oxide or zinc nitrate catalysts often suffer from complex reaction systems, prolonged reaction times exceeding 8 hours, and the use of expensive noble metals that complicate downstream purification and waste management. These legacy processes frequently result in inconsistent yields and poor universality across different substrate scopes, creating substantial barriers for procurement managers seeking cost reduction in electronic chemical manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology outlined in CN115703809A presents a paradigm shift towards efficiency and economic viability. By employing copper acetate as a sole catalyst, the new process eliminates the need for cryogenic cooling and unstable organolithium intermediates, allowing reactions to proceed under much milder thermal conditions ranging from 100°C to 155°C. The simplicity of the catalyst system, combined with the use of common organic solvents like toluene and DMF, drastically reduces the complexity of the reaction setup and the associated capital expenditure for specialized equipment. This approach not only enhances the overall atom economy but also simplifies the workup procedure, as the removal of copper residues is generally more straightforward than dealing with lithium salts or silver byproducts. Consequently, this novel route offers a compelling value proposition for supply chain heads focused on reducing lead time for high-purity photoelectric materials while maintaining stringent quality standards.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this technological breakthrough lies in the efficient activation of the 2-alkynyl phosphine oxide derivative by the copper catalyst. Mechanistically, the copper(II) species likely coordinates with the alkyne moiety and the phosphine oxide oxygen, facilitating an intramolecular cyclization that constructs the benzo-phosphole ring system. This coordination lowers the activation energy barrier for the ring-closing step, enabling the reaction to reach completion within a remarkably short timeframe of 30 to 60 minutes. The catalytic cycle is robust enough to tolerate a variety of substituents on the heterocyclic and aryl rings, including thienyl, benzothienyl, and various halogenated groups, demonstrating excellent functional group compatibility. This mechanistic efficiency is crucial for R&D directors who require versatile synthetic platforms capable of generating diverse libraries of analogues for structure-activity relationship studies without the need for extensive process re-optimization for each new derivative.
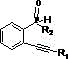
Furthermore, the choice of solvent plays a pivotal role in the success of this transformation, acting as more than just a medium for the reactants. Experimental data within the patent highlights that polar aprotic solvents like DMF and non-polar aromatic solvents like toluene yield superior results compared to alternatives such as DMA or DMSO. In toluene, yields reaching 85.5% have been observed, suggesting that the solvent environment optimally stabilizes the transition state or facilitates the solubility of the copper catalyst species. Understanding these subtle solvent effects allows process chemists to fine-tune the reaction parameters to minimize impurity formation, such as incomplete cyclization products or polymerization byproducts. This level of control over the impurity profile is essential for producing commercial scale-up of complex photoelectric materials that must meet the rigorous purity specifications demanded by the semiconductor and display industries.
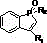
How to Synthesize Benzo-Phosphole Pentadiene Efficiently
Implementing this copper-catalyzed protocol requires careful attention to reagent stoichiometry and thermal management to maximize throughput. The process begins with the precise charging of the 2-alkynyl phosphine oxide substrate and the copper acetate catalyst, typically used in a molar ratio ranging from 0.1% to 10%, into a suitable reaction vessel. Following the addition of the preferred solvent system, the mixture is heated to the optimal temperature window, where the rapid kinetics ensure high conversion rates within an hour. For a comprehensive understanding of the specific operational parameters, safety precautions, and purification techniques required for GMP-compliant production, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with 2-alkynyl phosphine oxide derivative substrate and copper acetate catalyst (0.1-10 mol%).
- Add organic solvent, preferably toluene or DMF, and stir the mixture to ensure homogeneity.
- Heat the reaction mixture to 100-155°C for 30-60 minutes, then cool, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed synthesis route offers transformative benefits for the supply chain of organic electronic materials. The shift away from expensive and hazardous reagents like n-BuLi and silver salts directly translates to a reduction in raw material costs and a simplification of the procurement process. By utilizing copper acetate, a commodity chemical with a stable global supply, manufacturers can mitigate the risks associated with price volatility and supply disruptions often seen with specialty organometallic reagents. This stability is critical for procurement managers tasked with securing long-term contracts for high-volume production runs, ensuring that the cost of goods sold remains competitive in a rapidly evolving market.
- Cost Reduction in Manufacturing: The elimination of cryogenic infrastructure and the use of inexpensive copper catalysts significantly lower the operational expenditure (OPEX) associated with the synthesis. Without the need for energy-intensive cooling systems or the disposal of hazardous lithium waste, the overall production cost is drastically simplified. Additionally, the high yields achieved in optimized solvents mean less raw material is wasted per unit of product, further enhancing the economic efficiency of the process and driving substantial cost savings in electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and solvents ensures a robust and resilient supply chain. Unlike methods dependent on sensitive reagents that require special shipping and storage conditions, the components for this reaction are stable and easy to source globally. This accessibility reduces the lead time for high-purity photoelectric materials, allowing suppliers to respond more agilely to fluctuating market demands and preventing production bottlenecks that could delay downstream device fabrication.
- Scalability and Environmental Compliance: The short reaction times and mild conditions make this process highly scalable from laboratory benchtop to multi-ton industrial reactors. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, minimizing the burden on waste treatment facilities. This environmental compliance not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a key factor for partners committed to green chemistry principles in the production of advanced display materials.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and optimization of this synthesis method, we have compiled a set of answers based on the specific data points and embodiments found in the patent literature. These insights are designed to clarify the operational boundaries and potential advantages for teams evaluating this technology for integration into their existing manufacturing workflows. Understanding these nuances is vital for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of using copper acetate over n-BuLi for benzo-phosphole synthesis?
A: Unlike n-BuLi methods which require cryogenic conditions and generate hazardous waste, the copper acetate catalyzed process operates at moderate temperatures (100-155°C) using stable, inexpensive reagents, significantly improving safety and operational simplicity.
Q: Which solvents provide the highest yield for this cyclization reaction?
A: Experimental data indicates that toluene and DMF are the superior solvent choices, delivering yields upwards of 83-85%, whereas solvents like DMA or DMSO result in significantly lower conversion rates.
Q: Is this method suitable for large-scale production of OLED materials?
A: Yes, the short reaction time (30-60 minutes), use of earth-abundant copper catalysts, and absence of sensitive organolithium reagents make this protocol highly amenable to commercial scale-up for electronic chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo-Phosphole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to stay ahead in the competitive landscape of fine chemicals. Our team of expert process chemists has extensively evaluated the copper-catalyzed route described in CN115703809A and possesses the technical capability to translate this laboratory-scale innovation into a robust industrial process. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of benzo-phosphole compound meets the exacting standards required for next-generation optoelectronic applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this copper-catalyzed protocol. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the development and supply of high-performance electronic materials.
