Revolutionizing Organic Photoelectric Material Production with Efficient Copper-Catalyzed Cyclization
The landscape of organic photoelectric material synthesis is undergoing a significant transformation driven by the urgent need for greener, more cost-effective manufacturing protocols. Patent CN115703807A introduces a groundbreaking methodology for the preparation of benzo-phosphacyclopentadiene compounds, which serve as critical building blocks for advanced applications such as organic light-emitting diodes (OLEDs) and solar cells. This innovation specifically addresses the longstanding challenges associated with constructing phosphorus-containing heterocyclic skeletons by leveraging a simple yet highly effective copper oxide catalytic system. By shifting away from complex multi-step sequences and hazardous reagents, this technology offers a streamlined pathway that enhances both atomic economy and operational safety. For R&D directors and process chemists, this represents a pivotal opportunity to optimize impurity profiles and reduce the environmental footprint of high-value electronic chemical production. The strategic implementation of this copper-catalyzed route promises to redefine the economic feasibility of scaling these sophisticated molecular architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzo-phosphacyclopentadiene derivatives has been plagued by severe operational constraints and safety hazards that hinder industrial scalability. Traditional routes often rely on the use of phosphorus tribromide, a highly toxic and corrosive reagent that necessitates rigorous containment measures and generates substantial hazardous waste streams. Furthermore, alternative methodologies frequently employ n-butyllithium for lithiation steps, which requires maintaining cryogenic conditions to prevent decomposition, thereby escalating energy consumption and equipment costs. Other approaches utilizing zirconium complexes involve expensive precursors and intricate workup procedures to remove transition metal residues, complicating the purification process and lowering overall throughput. These legacy methods not only pose significant risks to personnel and the environment but also introduce variability in product quality due to the sensitivity of the intermediates involved. Consequently, the industry has faced a persistent bottleneck in securing a reliable supply of high-purity phosphole-based materials for next-generation optoelectronic devices.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent utilizes a remarkably simple catalytic system centered around copper oxide to drive the cyclization reaction. This method employs readily available phenyl phosphine oxide derivatives and alkynyl-containing compounds as starting materials, reacting them directly in an organic solvent to form the target heterocycle. The elimination of cryogenic requirements and toxic halogenating agents simplifies the reactor setup and drastically reduces the complexity of the downstream processing. Experimental results demonstrate that when acetonitrile is utilized as the reaction medium, the system achieves superior yields compared to other solvents like DMF or toluene, highlighting the robustness of this catalytic cycle. This streamlined protocol not only accelerates the reaction kinetics but also ensures a cleaner crude product profile, minimizing the burden on purification resources. By adopting this technical scheme, manufacturers can achieve a simpler, more economic, and environmentally friendly production process that is inherently suitable for large-scale operations.
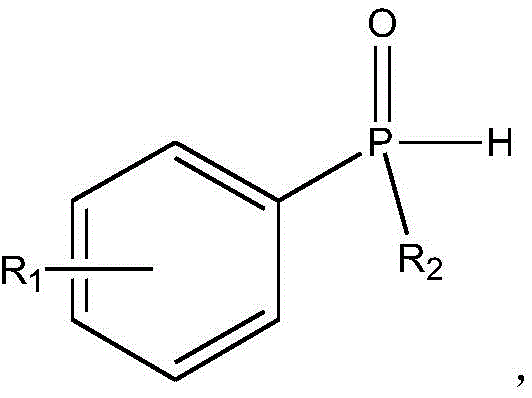
Mechanistic Insights into Copper-Oxide Catalyzed Cyclization
The mechanistic pathway of this transformation likely involves the activation of the phosphine oxide P-H bond or the alkyne moiety by the copper species, facilitating an intramolecular or intermolecular cyclization event. Copper oxide acts as a Lewis acid or a redox mediator that lowers the activation energy barrier for the formation of the carbon-phosphorus bonds within the heterocyclic ring. This catalytic cycle avoids the generation of highly reactive organolithium intermediates, thereby suppressing side reactions such as polymerization or over-reduction that often plague traditional syntheses. The tolerance of the catalyst towards various functional groups on the aryl rings suggests a versatile mechanism that can accommodate diverse substrate scopes without compromising efficiency. Understanding this catalytic behavior is crucial for process optimization, as it allows chemists to fine-tune reaction parameters like temperature and stoichiometry to maximize selectivity. The stability of the copper catalyst under the reaction conditions further implies a prolonged lifecycle, reducing the frequency of catalyst replenishment and contributing to overall process sustainability.
From an impurity control perspective, the use of copper oxide significantly mitigates the risk of introducing difficult-to-remove metal contaminants compared to noble metal catalysts. The reaction conditions, typically ranging from 80°C to 100°C, are mild enough to prevent thermal degradation of the sensitive phosphole core while being sufficiently energetic to drive the reaction to completion within a reasonable timeframe. This balance is essential for maintaining the integrity of the conjugated pi-system required for optoelectronic applications. Moreover, the absence of strong bases or nucleophiles minimizes the formation of salt byproducts, simplifying the aqueous workup and extraction phases. The resulting product exhibits high chemical and thermodynamic stability, a key attribute for materials intended for long-term device operation. By controlling the reaction environment through solvent selection and temperature modulation, manufacturers can ensure a consistent impurity profile that meets the stringent specifications of the electronic materials sector.
How to Synthesize Benzo-Phosphacyclopentadiene Efficiently
The synthesis of these valuable heterocycles can be executed with high precision by adhering to the optimized parameters outlined in the patent disclosure. The process begins with the precise charging of phenylphosphine oxide derivatives and alkynyl compounds alongside the copper oxide catalyst, ensuring the correct molar ratios are maintained to drive the equilibrium towards the product. Detailed standard operating procedures regarding solvent drying, atmosphere control, and heating ramps are critical to replicating the high yields observed in the experimental examples. While the general concept is straightforward, the nuances of mixing efficiency and heat transfer become paramount when transitioning from benchtop to pilot scale. For a comprehensive guide on the exact step-by-step execution, including specific workup and purification techniques, please refer to the standardized protocol below.
- Charge a reaction vessel with phenylphosphine oxide derivative, alkynyl-containing compound, and copper oxide catalyst in a molar ratio of approximately 2: 1:1.
- Add acetonitrile as the preferred solvent to the mixture and stir until uniform, then heat the system to a temperature range of 80-100°C.
- Maintain the reaction temperature for 3 to 6 hours, then cool, concentrate, and purify via column chromatography to isolate the target heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on cost stability and operational resilience. The substitution of expensive and hazardous reagents with commodity-grade copper oxide fundamentally alters the cost structure of the raw material bill, leading to substantial savings in direct material costs. Furthermore, the simplified reaction workflow reduces the demand for specialized cryogenic equipment and complex waste treatment infrastructure, translating into lower capital expenditure and overheads. The robustness of the process against variations in reaction conditions enhances supply continuity by minimizing batch failures and off-spec production. This reliability is critical for maintaining just-in-time inventory levels and meeting the demanding delivery schedules of downstream electronics manufacturers. Ultimately, this technology enables a more agile and responsive supply chain capable of adapting to market fluctuations without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The elimination of costly transition metal complexes and the avoidance of cryogenic operations result in a drastic simplification of the production process. By removing the need for expensive reagents like n-butyllithium and phosphorus tribromide, the direct input costs are significantly lowered, while the reduced hazard profile decreases insurance and safety compliance expenses. The use of acetonitrile, a common and recoverable solvent, further optimizes the solvent recovery loop, minimizing waste disposal fees. These cumulative efficiencies drive down the overall cost of goods sold, allowing for more competitive pricing strategies in the global market for organic electronic materials.
- Enhanced Supply Chain Reliability: Sourcing copper oxide and simple phosphine oxides is far less volatile than securing specialized organometallic reagents, which are often subject to geopolitical supply constraints. The simplified logistics of handling stable solid catalysts versus pyrophoric liquids reduce transportation risks and storage requirements. This stability ensures a consistent flow of materials into the production line, mitigating the risk of stoppages due to raw material shortages. Additionally, the shorter reaction times and streamlined workup procedures increase the throughput capacity of existing manufacturing assets, effectively expanding supply availability without the need for new facility construction.
- Scalability and Environmental Compliance: The benign nature of the catalyst and the absence of toxic halogenated byproducts align perfectly with increasingly stringent environmental regulations. Scaling this process does not require exponential increases in waste treatment capacity, as the effluent load is significantly lighter than that of traditional methods. The ability to operate at atmospheric pressure and moderate temperatures simplifies the engineering controls needed for scale-up, reducing the time and investment required to bring new products to commercial volume. This environmental compatibility not only future-proofs the manufacturing site against regulatory changes but also enhances the brand reputation of the supplier as a sustainable partner in the green chemistry initiative.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on process capabilities. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production portfolios. The answers reflect the practical realities of scaling copper-catalyzed heterocyclization for industrial applications.
Q: What are the primary advantages of using copper oxide over traditional catalysts for this synthesis?
A: Unlike traditional methods requiring toxic phosphorus tribromide or cryogenic n-butyllithium, copper oxide is inexpensive, environmentally benign, and operates under mild thermal conditions, significantly reducing safety hazards and waste disposal costs.
Q: Which solvent system provides the optimal yield for benzo-phosphacyclopentadiene formation?
A: Experimental data indicates that acetonitrile serves as the superior solvent compared to DMF, toluene, or ethanol, delivering the highest conversion rates and product purity for this specific copper-catalyzed cyclization.
Q: Is this process suitable for large-scale industrial production of OLED intermediates?
A: Yes, the elimination of sensitive reagents like n-BuLi and the use of robust, non-cryogenic conditions (80-100°C) make this protocol highly scalable and economically viable for commercial manufacturing of electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo-Phosphacyclopentadiene Supplier
As the demand for high-performance organic photoelectric materials continues to surge, partnering with a technically proficient manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand the critical nature of impurity profiles in electronic materials and have the expertise to refine this copper-catalyzed process to deliver products that exceed performance expectations. Our team is ready to collaborate on customizing this synthesis to fit your specific application requirements.
We invite you to engage with our technical procurement team to discuss how this innovative manufacturing route can benefit your specific project goals. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify the suitability of our materials for your downstream processes. Let us help you optimize your supply chain with reliable, high-quality benzo-phosphacyclopentadiene intermediates designed for the future of optoelectronics.
