Advanced Brønsted Acid-Catalyzed Synthesis for Commercial Scale Benzopyran Derivatives
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly benzopyran derivatives which serve as critical cores in numerous bioactive molecules. Patent CN111004198A introduces a groundbreaking total synthesis strategy that addresses long-standing inefficiencies in producing 4-sulfonyl functional group-substituted benzopyran derivatives. This innovative approach leverages a Brønsted acid-catalyzed [4+2] cyclization as the cornerstone reaction, fundamentally shifting the paradigm from traditional transition metal-dependent processes to a more sustainable and operationally simple protocol. By utilizing phenylethynyl ethyl sulfide and α-phenyl o-hydroxybenzyl alcohol as accessible starting materials, this method not only streamlines the synthetic sequence but also enhances the overall safety profile of the manufacturing process. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to optimize the supply chain for high-value pharmaceutical intermediates, offering a pathway that balances high chemical fidelity with commercial viability.
Furthermore, the strategic implementation of this synthesis route directly impacts the economic feasibility of producing mGluR7 agonists, RORγ activity modulators, and STAT3 protein activity inhibitors, all of which rely on the benzopyran backbone. The ability to construct these complex architectures under mild conditions without the burden of harsh reagents or extreme temperatures translates to tangible benefits in plant operations. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented technologies is essential for maintaining a competitive edge. The following analysis dissects the technical merits of this invention, providing a comprehensive view of how this specific chemical transformation can be leveraged to achieve cost reduction in fine chemical manufacturing while ensuring the highest standards of product quality and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzopyran frameworks has been plagued by significant synthetic hurdles that impede efficient large-scale production. Traditional methodologies often necessitate the use of complicated and specific reaction substrates that require multi-step pre-functionalization, thereby inflating the raw material costs and extending the overall lead time. Moreover, many established routes rely heavily on the addition of complex metal catalysts, which introduce severe challenges in downstream processing, particularly regarding the removal of trace metal residues to meet stringent pharmaceutical specifications. These metal-mediated reactions frequently demand harsh conditions, such as elevated temperatures or strong bases, which can compromise the stability of sensitive functional groups and lead to the formation of difficult-to-separate impurities. Consequently, these factors collectively contribute to increased production costs, higher waste generation, and potential safety hazards within the manufacturing facility, creating a bottleneck for the commercial scale-up of complex organic intermediates.
The Novel Approach
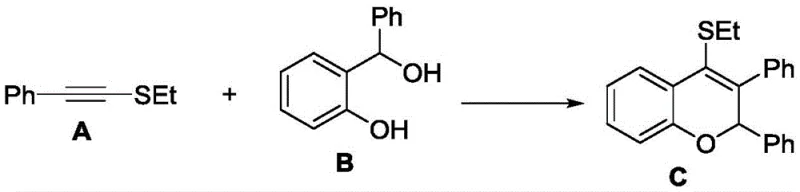
In stark contrast to these legacy methods, the novel approach detailed in the patent data utilizes a Brønsted acid-catalyzed strategy that elegantly bypasses the need for transition metals in the critical cyclization step. This method employs bis(trifluoromethanesulfonyl)imide (HNTf2) as a highly efficient catalyst to drive the [4+2] cyclization between phenylethynyl ethyl sulfide and α-phenyl o-hydroxybenzyl alcohol. The reaction proceeds smoothly at room temperature, demonstrating exceptional atom economy and operational simplicity. By avoiding the use of expensive and toxic metal catalysts, this route inherently reduces the complexity of the workup procedure, eliminating the need for costly metal scavenging resins or extensive washing protocols. The mildness of the reaction conditions also preserves the integrity of the substrate, minimizing degradation and side reactions, which ultimately results in a cleaner crude product profile and higher isolated yields. This shift towards organocatalysis represents a substantial advancement in process chemistry, aligning perfectly with modern green chemistry principles and industrial demands for safer, more sustainable manufacturing processes.
Mechanistic Insights into Brønsted Acid-Catalyzed Cyclization
The core innovation of this synthesis lies in the mechanistic pathway facilitated by the strong Brønsted acid catalyst. The reaction initiates with the activation of the alkyne moiety in phenylethynyl ethyl sulfide by the proton source, generating a highly reactive vinyl cation intermediate. This electrophilic species is then intercepted by the nucleophilic oxygen of the α-phenyl o-hydroxybenzyl alcohol, triggering an intramolecular cyclization event that constructs the 2H-chromene core with high regioselectivity. The use of HNTf2 is particularly advantageous due to its high acidity and non-coordinating nature, which ensures rapid turnover and prevents catalyst deactivation. This mechanistic elegance allows for the direct assembly of the benzopyran skeleton from simple precursors in a single operational step, drastically reducing the step count compared to traditional ring-closing metathesis or palladium-catalyzed coupling strategies. The efficiency of this transformation is evidenced by the short reaction time of merely 10 minutes, showcasing the potent catalytic activity and the favorable kinetics of the proposed pathway.
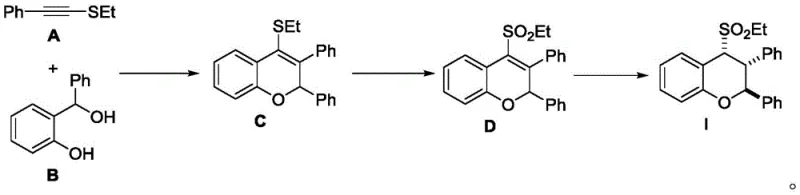
Following the initial cyclization, the synthetic sequence proceeds through a carefully orchestrated oxidation and reduction protocol to install the final 4-sulfonyl functionality and saturate the double bond. The oxidation step utilizes m-CPBA to convert the sulfide moiety into the corresponding sulfone, a transformation that is critical for the biological activity of the target molecules. Subsequently, catalytic hydrogenation using Pd/C under controlled pressure reduces the olefinic bond without affecting the sensitive sulfone group or the aromatic rings. This sequential functionalization demonstrates a high level of chemoselectivity, ensuring that the final product retains the desired structural features with minimal impurity formation. For R&D teams, understanding this mechanistic flow is crucial for troubleshooting and optimizing the process further, as it highlights the robustness of the intermediates and the compatibility of the reaction conditions with various substituents. The ability to control the stereochemistry and purity at each stage underscores the suitability of this method for producing high-purity benzopyran derivatives required for clinical applications.
How to Synthesize 4-Sulfonyl Benzopyran Derivatives Efficiently
Implementing this synthesis on a laboratory or pilot scale requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction vessel, typically a Schlenk tube or sealed reactor, which must be purged with nitrogen to maintain an inert atmosphere. The precise stoichiometric addition of phenylethynyl ethyl sulfide and α-phenyl o-hydroxybenzyl alcohol, along with the catalytic amount of HNTf2 in dichloroethane (DCE), is critical for maximizing the yield of the 2H-chromene intermediate. Monitoring the reaction progress via TLC is essential to prevent over-reaction or decomposition, although the rapid kinetics usually allow for a straightforward workup. The subsequent oxidation and reduction steps also demand careful control of temperature and pressure to maintain the integrity of the sulfonyl group while achieving complete conversion. Detailed standardized synthesis steps see the guide below.
- Perform Brønsted acid-catalyzed [4+2] cyclization of phenylethynyl ethyl sulfide and α-phenyl o-hydroxybenzyl alcohol using HNTf2 in DCE at room temperature.
- Oxidize the resulting 2H-chromene intermediate using m-CPBA in dichloromethane to form the 4-sulfonyl-2H-chromene derivative.
- Conduct catalytic hydrogenation of the sulfonyl intermediate using Pd/C under 3MPa H2 pressure in ethanol/ethyl acetate to yield the final benzopyran derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The elimination of transition metal catalysts in the primary bond-forming step translates to significant cost savings by removing the expense associated with purchasing precious metals and the subsequent disposal or recycling of metal-contaminated waste streams. Furthermore, the use of commercially available and inexpensive starting materials ensures a stable supply chain, reducing the risk of raw material shortages that often plague specialized chemical manufacturing. The mild reaction conditions, particularly the room temperature cyclization, lower the energy consumption of the process, contributing to a reduced carbon footprint and lower utility costs. These factors combined create a more resilient and cost-efficient manufacturing model that can withstand market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the avoidance of expensive transition metal catalysts and the associated purification infrastructure. By utilizing a Brønsted acid catalyst, the process eliminates the need for specialized metal scavenging resins and extensive washing protocols, which are both time-consuming and costly. Additionally, the high yield of the cyclization step minimizes raw material waste, ensuring that a greater proportion of the input materials are converted into valuable product. This efficiency extends to the solvent usage as well, as the reaction can be performed in standard chlorinated solvents that are easily recovered and recycled. Overall, the streamlined workflow reduces the operational expenditure per kilogram of product, making it a highly attractive option for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as phenylethynyl ethyl sulfide and α-phenyl o-hydroxybenzyl alcohol significantly mitigates supply chain risks. Unlike specialized organometallic reagents that may have long lead times or limited suppliers, these starting materials can be sourced from multiple vendors globally, ensuring continuity of supply. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, further stabilizing the production schedule. For supply chain planners, this translates to more predictable delivery timelines and the ability to scale production up or down based on demand without encountering significant logistical bottlenecks. This reliability is crucial for maintaining the inventory levels required to support downstream pharmaceutical manufacturing.
- Scalability and Environmental Compliance: The inherent safety and mildness of this synthetic route make it exceptionally well-suited for scale-up from kilogram to tonnage quantities. The absence of pyrophoric reagents or extreme temperatures reduces the engineering controls required for safe operation, simplifying the technology transfer to manufacturing sites. Moreover, the reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, minimizing the liability and cost associated with waste disposal. The high atom economy of the cyclization step further supports sustainability goals by maximizing resource utilization. These attributes ensure that the process remains viable and compliant as production volumes increase, supporting the long-term commercial success of the benzopyran derivative.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzopyran synthesis route. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for their specific applications. The responses cover aspects ranging from catalyst handling to product purity, ensuring a comprehensive understanding of the method's value proposition.
Q: What are the advantages of using Brønsted acid catalysis over transition metals for this synthesis?
A: The use of Brønsted acid (HNTf2) eliminates the need for expensive and toxic transition metal catalysts in the initial cyclization step. This significantly simplifies the post-reaction purification process by removing the requirement for heavy metal scavenging, thereby reducing production costs and environmental impact while maintaining high reaction efficiency at room temperature.
Q: How does this synthetic route improve supply chain stability for benzopyran intermediates?
A: This method utilizes readily available starting materials such as phenylethynyl ethyl sulfide and α-phenyl o-hydroxybenzyl alcohol. The mild reaction conditions, particularly the room temperature cyclization and standard hydrogenation steps, reduce the dependency on specialized high-pressure or cryogenic equipment, ensuring more consistent manufacturing timelines and reduced risk of production bottlenecks.
Q: What is the expected purity profile of the final benzopyran derivative using this method?
A: The stepwise approach involving distinct cyclization, oxidation, and reduction phases allows for rigorous intermediate monitoring and purification. The mild conditions minimize side reactions and decomposition, leading to a high-purity final product with a total yield reaching up to 66.8%, which is critical for meeting stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly evaluated the Brønsted acid-catalyzed route described in CN111004198A and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzopyran derivative meets the highest industry standards. By leveraging our expertise in process optimization, we can help you realize the full cost and efficiency benefits of this innovative synthesis.
We invite you to collaborate with us to optimize your supply chain for high-purity pharmaceutical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this technology into your production pipeline. Together, we can drive innovation and efficiency in the manufacturing of complex organic molecules.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
