Scalable Synthesis of Chiral Halohydrin Amines from L-Phenylalanine for Antiviral Drug Manufacturing
Scalable Synthesis of Chiral Halohydrin Amines from L-Phenylalanine for Antiviral Drug Manufacturing
The global demand for potent antiretroviral therapies continues to drive innovation in the synthesis of complex chiral intermediates, particularly those required for protease inhibitors such as atazanavir and amprenavir. Patent CN114671782A introduces a groundbreaking preparation method for halohydrin amines, a critical structural motif characterized by adjacent hydroxyl and halogen functionalities on a chiral backbone. This technology leverages natural L-phenylalanine as a renewable and cost-effective starting material to construct the necessary stereocenters with exceptional precision. By bypassing hazardous reagents traditionally associated with this chemical class, the disclosed route offers a robust pathway for producing high-purity pharmaceutical intermediates. 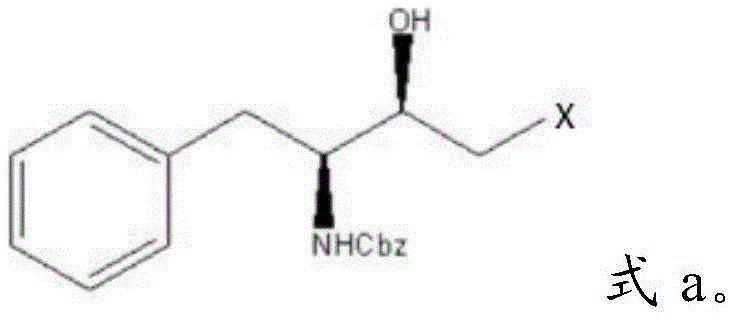 The significance of this development lies in its ability to streamline the supply chain for antiviral active pharmaceutical ingredients (APIs). The target molecule, depicted in the structural formula above, serves as a pivotal building block where the stereochemistry at both the amino-bearing carbon and the hydroxyl-bearing carbon is strictly defined. Achieving this dual chirality typically requires intricate resolution steps or asymmetric catalysis, yet this patent demonstrates a direct construction strategy that minimizes waste and maximizes atom economy. For R&D directors and procurement specialists alike, understanding this shift from dangerous diazomethane chemistry to stable dihalomethane substitution represents a major leap forward in process safety and commercial viability.
The significance of this development lies in its ability to streamline the supply chain for antiviral active pharmaceutical ingredients (APIs). The target molecule, depicted in the structural formula above, serves as a pivotal building block where the stereochemistry at both the amino-bearing carbon and the hydroxyl-bearing carbon is strictly defined. Achieving this dual chirality typically requires intricate resolution steps or asymmetric catalysis, yet this patent demonstrates a direct construction strategy that minimizes waste and maximizes atom economy. For R&D directors and procurement specialists alike, understanding this shift from dangerous diazomethane chemistry to stable dihalomethane substitution represents a major leap forward in process safety and commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral halohydrin amines has been plagued by significant safety hazards and operational complexities that hinder large-scale manufacturing. Traditional routes frequently employ diazomethane or its derivatives to extend the carbon chain or introduce functionality, but diazomethane is notoriously unstable, toxic, and explosive, posing unacceptable risks in an industrial setting. Furthermore, existing methodologies often involve multi-step sequences with poor overall yields, requiring extensive chromatographic purification to remove toxic heavy metal residues or difficult-to-separate diastereomers. These inefficiencies not only inflate the cost of goods sold (COGS) but also create bottlenecks in the supply chain, leading to inconsistent availability of key intermediates for HIV drug production. The reliance on such hazardous reagents necessitates specialized equipment and rigorous safety protocols, which further escalates capital expenditure and limits the number of qualified suppliers capable of meeting global demand.
The Novel Approach
In stark contrast, the methodology outlined in patent CN114671782A presents a streamlined, four-step sequence that prioritizes safety without compromising on stereochemical fidelity. 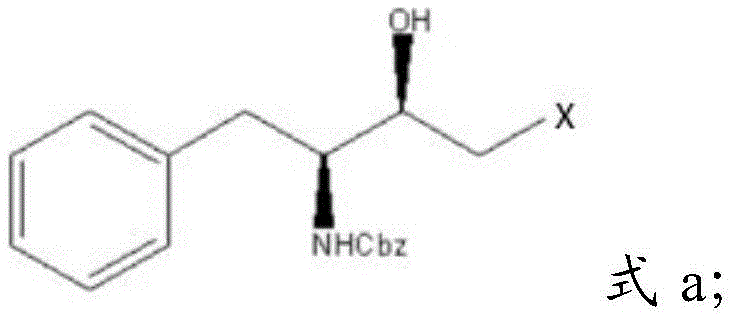 As illustrated in the reaction scheme, the process initiates with the protection of L-phenylalanine, followed by esterification and a highly controlled reduction to an aldehyde. The cornerstone of this innovation is the final substitution step, where a dihalomethane reagent reacts with the aldehyde intermediate in the presence of a Grignard-type reagent (isopropyl magnesium chloride). This approach effectively replaces the dangerous diazomethane insertion with a safer dihalomethane coupling, directly installing the halohydrin moiety. The result is a concise synthetic route that avoids toxic reagents, utilizes inexpensive commodity chemicals, and delivers the target compound with reported purities exceeding 99%. This paradigm shift enables reliable pharmaceutical intermediate suppliers to scale production confidently, ensuring a steady flow of materials for the downstream synthesis of life-saving antiretroviral medications.
As illustrated in the reaction scheme, the process initiates with the protection of L-phenylalanine, followed by esterification and a highly controlled reduction to an aldehyde. The cornerstone of this innovation is the final substitution step, where a dihalomethane reagent reacts with the aldehyde intermediate in the presence of a Grignard-type reagent (isopropyl magnesium chloride). This approach effectively replaces the dangerous diazomethane insertion with a safer dihalomethane coupling, directly installing the halohydrin moiety. The result is a concise synthetic route that avoids toxic reagents, utilizes inexpensive commodity chemicals, and delivers the target compound with reported purities exceeding 99%. This paradigm shift enables reliable pharmaceutical intermediate suppliers to scale production confidently, ensuring a steady flow of materials for the downstream synthesis of life-saving antiretroviral medications.
Mechanistic Insights into Amino Acid Derivatization and Grignard Substitution
The success of this synthetic strategy hinges on the meticulous preservation of chirality throughout the transformation of the amino acid scaffold. The process begins with the N-protection of L-phenylalanine using benzyl chloroformate (Cbz-Cl) under mild alkaline conditions, which locks the alpha-chiral center in place while rendering the amine inert to subsequent nucleophilic attacks. Following esterification to activate the carboxylic acid, the critical reduction step employs diisobutyl aluminum hydride (DIBAL-H) at cryogenic temperatures ranging from -75°C to -80°C. This low-temperature control is paramount; it ensures the selective reduction of the ester to the aldehyde without over-reduction to the alcohol or racemization of the sensitive alpha-center. The resulting Cbz-L-phenylalaninal retains the optical purity of the starting material, providing a rigid chiral template for the final carbon-carbon bond formation.
The final mechanistic event involves the nucleophilic addition of a halomethyl species generated in situ from dihalomethane and isopropyl magnesium chloride. This reaction proceeds through a coordinated transition state where the magnesium species facilitates the delivery of the halomethyl group to the aldehyde carbonyl. The stereochemical outcome is governed by the existing chiral center at the alpha-position, which directs the approach of the nucleophile to favor the formation of the desired (1S, 2R) or analogous configuration required for biological activity. By avoiding harsh acidic or basic workups that could epimerize the newly formed stereocenter, the process maintains high diastereoselectivity. This precise control over impurity profiles is essential for meeting the stringent regulatory standards imposed on pharmaceutical intermediates, ensuring that the final API contains minimal levels of unwanted isomers that could compromise efficacy or safety.
How to Synthesize Chiral Halohydrin Amine Efficiently
The patented procedure offers a clear, reproducible protocol for generating high-value halohydrin amines suitable for clinical and commercial applications. The synthesis is designed to be operationally simple, utilizing standard reactor setups and common organic solvents like toluene and tetrahydrofuran. Key to the success of the operation is the strict temperature control during the DIBAL-H reduction and the subsequent Grignard substitution, which dictates the quality of the intermediate aldehyde and the final product. Detailed standardized synthetic steps, including specific molar ratios and quenching procedures, are outlined below to guide process chemists in replicating these results.
- Perform amino protection on L-phenylalanine using benzyl chloroformate and an alkaline compound in water at 0-8°C to obtain Cbz-L-phenylalanine.
- Conduct esterification of Cbz-L-phenylalanine with thionyl chloride and methanol under reflux conditions to yield Cbz-L-phenylalanine methyl ester.
- Reduce the methyl ester to the corresponding aldehyde using diisobutyl aluminum hydride (DIBAL-H) in toluene at cryogenic temperatures (-75 to -80°C).
- Execute a substitution reaction between the aldehyde, methylene dihalide, and isopropyl magnesium chloride in tetrahydrofuran to finalize the halohydrin amine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift away from hazardous reagents like diazomethane fundamentally alters the risk profile of the manufacturing process, allowing for production in a wider range of facilities without the need for specialized explosion-proof infrastructure. This flexibility enhances supply chain resilience, reducing the likelihood of production stoppages due to safety audits or regulatory compliance issues. Furthermore, the use of L-phenylalanine, a bulk commodity amino acid produced via fermentation, ensures a stable and predictable raw material supply that is insulated from the volatility often seen with specialty fine chemical precursors.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents significantly lowers the direct material costs associated with production. By replacing diazomethane with inexpensive dihalomethanes and utilizing a high-yielding four-step sequence, the overall process mass intensity (PMI) is improved, leading to substantial cost savings in waste disposal and solvent recovery. The high purity achieved (>99%) reduces the need for resource-intensive recrystallization or chromatography steps, further driving down the cost per kilogram and enabling more competitive pricing for the final antiviral drugs.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit greatly from the use of universally available starting materials like L-phenylalanine and benzyl chloroformate, which are produced by multiple global vendors. This diversification of the supply base mitigates the risk of single-source dependency and ensures continuity of supply even during market fluctuations. Additionally, the robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, resulting in consistent batch-to-batch performance and reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, avoiding unit operations that are difficult to translate from the laboratory to the plant floor. The absence of toxic heavy metals and explosive intermediates simplifies environmental permitting and reduces the burden on wastewater treatment systems. This alignment with green chemistry principles not only lowers operational costs related to environmental compliance but also enhances the corporate sustainability profile of the manufacturing organization, a factor increasingly weighted in vendor selection processes by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halohydrin amine synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process capabilities. Understanding these details is crucial for technical teams assessing the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this synthesis route safer than conventional methods for halohydrin amines?
A: Conventional methods often rely on diazomethane, a highly toxic and explosive reagent unsuitable for large-scale production. This patented route eliminates diazomethane entirely, utilizing stable reagents like dihalomethanes and DIBAL-H, significantly enhancing operational safety and industrial feasibility.
Q: How does the process ensure high stereochemical purity?
A: The process utilizes natural L-phenylalanine as the starting material, which inherently possesses the required chirality at the alpha-position. The subsequent mild reaction conditions, particularly the controlled low-temperature reduction and substitution steps, preserve this chiral integrity while constructing the second chiral center with high selectivity.
Q: Is this method suitable for commercial scale-up?
A: Yes, the patent explicitly states the method is suitable for industrial production. It uses commercially available, low-cost raw materials like L-phenylalanine and avoids complex purification steps, with reported yields exceeding 80% and purities greater than 99% in laboratory examples, indicating strong potential for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halohydrin Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against HIV and other viral diseases. Our team of expert process chemists has thoroughly analyzed the technological advancements presented in patent CN114671782A and is fully prepared to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the stereochemical integrity and chemical purity of every batch, guaranteeing that the materials you receive meet the highest international standards.
We invite you to collaborate with us to optimize your supply chain for antiviral drug manufacturing. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced synthesis capabilities can drive value and reliability for your organization.
