Advanced Synthesis of 3-Hydroxy-2-Phenylpropionic Acid for High-Value Pharmaceutical Intermediates
Introduction to Patent CN110551027B
The pharmaceutical industry constantly demands higher purity standards for active pharmaceutical ingredient (API) intermediates to ensure patient safety and regulatory compliance. A significant technological breakthrough in this domain is detailed in Chinese Patent CN110551027B, which discloses an innovative synthetic method for 3-hydroxy-2-phenylpropionic acid, also known as tropic acid. This compound serves as a critical building block for the synthesis of tropicamide, a widely used mydriatic agent. The patent outlines a robust process that leverages dimethyl sulfoxide (DMSO) as a reaction medium and employs a sophisticated sequential recrystallization technique. By integrating real-time gas phase monitoring with a unique purification strategy involving toluene and water mixtures, this method successfully addresses the longstanding challenge of balancing high yield with ultra-high purity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patented route is essential for optimizing supply chains and reducing overall manufacturing costs in the production of anticholinergic drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxy-2-phenylpropionic acid has been plagued by inefficiencies that hinder large-scale commercial viability. The earliest documented approach, referenced in the National Formulary of Bulk Drugs (1980), involves the condensation of methyl phenylacetate with ethyl formate under sodium alkoxide conditions, followed by reduction with potassium borohydride. As illustrated in the reaction scheme below, this multi-step pathway is not only operationally complex but also suffers from a disappointingly low overall yield of approximately 56%. Such poor efficiency translates directly into higher raw material consumption and increased waste generation, making it economically unattractive for modern high-volume production environments where margin compression is a constant concern for procurement teams.
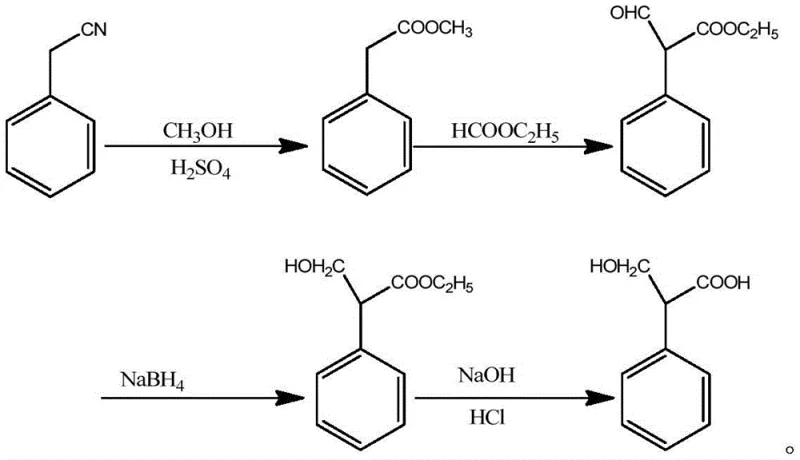
Subsequent improvements attempted to streamline the process by utilizing paraformaldehyde and sodium bicarbonate for direct condensation, as shown in the following diagram. While this second-generation method improved the total yield to roughly 74.4%, it introduced a different bottleneck: product purity. The resulting 3-hydroxy-2-phenylpropionic acid typically achieved a purity of only 98%, which falls short of the stringent specifications required for high-grade pharmaceutical applications. Impurities at this level can complicate downstream processing and necessitate additional, costly purification steps that erode the gains made in yield. Consequently, there remains a critical need for a manufacturing process that can simultaneously deliver both high throughput and the >99% purity demanded by top-tier API manufacturers.
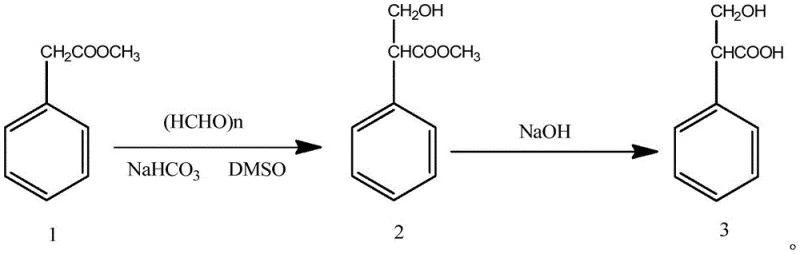
The Novel Approach
The methodology described in patent CN110551027B represents a paradigm shift by decoupling the trade-off between yield and purity. Instead of relying on harsh reducing agents or complex isolation procedures for intermediates, this novel approach utilizes a direct hydroxymethylation of methyl phenylacetate with paraformaldehyde in a DMSO solvent system catalyzed by potassium carbonate. The true innovation lies in the downstream processing; rather than attempting to purify the intermediate methyl tropate, the process proceeds directly to hydrolysis and then employs a triple-stage recrystallization protocol. By sequentially washing and recrystallizing the crude product with toluene, a toluene-water mixture, and finally pure water in specific mass ratios, the method effectively scrubs away trace impurities that single-solvent systems cannot remove. This ensures that the final product consistently exceeds 99% purity while maintaining a robust yield of over 76.5%, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into DMSO-Mediated Hydroxymethylation
The core chemical transformation in this synthesis is the base-catalyzed hydroxymethylation of the alpha-carbon of methyl phenylacetate. In this mechanism, potassium carbonate acts as a mild yet effective base to generate the enolate ion of the ester in the polar aprotic solvent DMSO. The high polarity of DMSO stabilizes the charged intermediates, facilitating the nucleophilic attack on the formaldehyde generated in situ from paraformaldehyde. This step is critical because the reaction equilibrium must be carefully managed to favor the formation of alpha-hydroxymethyl methyl phenylacetate (methyl tropate) without promoting side reactions such as polymerization of formaldehyde or over-alkylation. The patent specifies heating the mixture to 85-95°C, a temperature window optimized to provide sufficient activation energy for the condensation while minimizing thermal degradation of the sensitive ester functionality. Precise control over the molar ratios, specifically maintaining a slight excess of paraformaldehyde (1:1.1-1.2) and a significant excess of the base catalyst, drives the equilibrium toward the desired product, ensuring the GC content reaches the critical threshold of ≥70% before quenching.
Following the condensation, the purification mechanism relies on the differential solubility of the target acid versus its impurities in a gradient of solvent polarities. The sequential recrystallization is designed to exploit these solubility differences systematically. The initial wash with toluene removes non-polar organic impurities and residual starting materials that are soluble in aromatic hydrocarbons. The subsequent step using a toluene-water mixture creates a biphasic environment or a specific solubility pocket that precipitates the target acid while keeping polar by-products and salts in the aqueous phase or dissolved in the organic layer depending on their partition coefficients. Finally, the wash with pure water removes any remaining inorganic salts, such as sodium chloride formed during acidification, and highly polar organic contaminants. This graduated approach is far superior to single-solvent recrystallization, which often leaves behind specific classes of impurities, thereby explaining how the process achieves the exceptional purity levels required for a high-purity pharmaceutical intermediate.
How to Synthesize 3-Hydroxy-2-Phenylpropionic Acid Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters and purification sequence to replicate the high yields and purity reported in the patent. The process begins with the careful preparation of the reaction mixture, ensuring that the DMSO is dry and the potassium carbonate is finely powdered to maximize surface area for catalysis. Operators must monitor the reaction progress using gas chromatography to identify the exact endpoint, preventing unnecessary energy consumption and potential product degradation. Following the reaction, the removal of DMSO under reduced pressure is a critical unit operation that requires precise temperature and vacuum control to avoid thermal stress on the product. The hydrolysis step must be conducted with controlled addition of sodium hydroxide to manage exotherms, followed by careful acidification to precipitate the crude acid.
- Condense methyl phenylacetate with paraformaldehyde in DMSO using potassium carbonate catalyst at 85-95°C until GC content of methyl tropate reaches ≥70%.
- Neutralize the reaction mixture, filter, and remove DMSO via reduced pressure distillation at 108-110°C under 50-200 Pa vacuum.
- Hydrolyze the residue with methanol and sodium hydroxide solution at 75-85°C, then acidify with hydrochloric acid to pH 2-3 to obtain crude product.
- Purify the crude product through sequential recrystallization using toluene, followed by a toluene/water mixture, and finally water to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical performance. The elimination of the intermediate isolation step significantly simplifies the manufacturing workflow, reducing the number of unit operations required to produce the final API intermediate. This streamlining directly correlates to lower operational expenditures, as fewer reactors, filters, and drying cycles are needed per batch. Furthermore, the avoidance of expensive and hazardous reducing agents like potassium borohydride, which were necessary in older methods, removes a major cost driver and safety liability from the production line. By relying on commodity chemicals such as DMSO, paraformaldehyde, and toluene, the supply chain becomes more resilient and less susceptible to price volatility associated with specialized reagents, ensuring a stable and continuous supply of this critical medical intermediate.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the significant simplification of the downstream processing train. By bypassing the purification of the methyl tropate intermediate, the manufacturer saves on solvent usage, filtration media, and labor hours associated with handling semi-finished goods. Additionally, the high yield of over 76.5% means that less raw material is wasted per kilogram of final product, improving the overall atom economy of the process. The use of recyclable solvents like toluene and DMSO further enhances the cost structure, allowing for solvent recovery and reuse which drastically lowers the variable cost of goods sold. These factors combine to create a highly competitive cost position for producers adopting this technology.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on widely available bulk chemicals ensures that production schedules are not disrupted by the scarcity of niche reagents. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring exotic metallurgy or extreme pressures, facilitates easier technology transfer between manufacturing sites. This flexibility allows supply chain leaders to diversify their manufacturing footprint, mitigating the risk of regional disruptions. Moreover, the consistent achievement of >99% purity reduces the likelihood of batch failures or customer rejections, leading to more predictable inventory levels and improved service levels for downstream API customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the absence of highly exothermic or dangerous reduction steps. The reaction operates at moderate temperatures and atmospheric pressure, simplifying reactor design and safety protocols. Environmentally, the process generates less hazardous waste compared to borohydride-based routes, easing the burden on wastewater treatment facilities and reducing disposal costs. The ability to recover and recycle the bulk solvents used in the recrystallization steps aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without compromising on output volume or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of scaling this route and for quality assurance professionals ensuring that the final product meets all regulatory specifications. The answers provided are derived directly from the experimental data and technical disclosures within the patent documentation, offering a reliable foundation for decision-making.
Q: How does this new method improve purity compared to traditional routes?
A: Traditional methods often struggle to exceed 98% purity due to difficult-to-remove impurities. This patent utilizes a specific sequential recrystallization protocol (Toluene → Toluene/Water → Water) that effectively strips away residual solvents and by-products, consistently achieving purity levels above 99%.
Q: Why is GC monitoring critical in the first step of this synthesis?
A: Gas Chromatography (GC) monitoring allows for precise determination of the reaction endpoint when methyl tropate content reaches ≥70%. This prevents over-reaction or incomplete conversion, ensuring optimal yield before proceeding to the hydrolysis and purification stages, which is vital for cost-effective manufacturing.
Q: What are the key advantages for large-scale production of this intermediate?
A: The process eliminates the need to isolate the intermediate methyl tropate, significantly reducing unit operations. Furthermore, the use of common solvents like DMSO, toluene, and water, combined with high yields (>76.5%), makes the process highly scalable and economically viable for industrial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2-Phenylpropionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of reaction kinetics and purification thermodynamics described in this patent is maintained at an industrial scale. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify that every batch of 3-hydroxy-2-phenylpropionic acid exceeds the 99% purity threshold required for high-value pharmaceutical applications. Our infrastructure is designed to handle the specific solvent systems and reaction conditions of this novel route efficiently, guaranteeing supply continuity for our global partners.
We invite procurement directors and R&D leaders to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data from our recent batches and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a reliable source of high-quality intermediates that drive efficiency and innovation in your drug development pipeline.
