Advanced Green Synthesis of 3,4,5,6-Tetrafluoro-N-methylphthalimide for Commercial Scale-up
The pharmaceutical industry constantly seeks more efficient pathways for producing critical antibiotic intermediates, particularly for the widely prescribed fluoroquinolone class of drugs such as Ofloxacin and Levofloxacin. A significant technological breakthrough in this domain is detailed in patent CN110407735B, which discloses a novel green synthesis process for 3,4,5,6-tetrafluoro-N-methylphthalimide. This compound serves as a pivotal building block in the manufacturing value chain of third-generation broad-spectrum antibacterial agents. The patented methodology addresses longstanding inefficiencies in halogen exchange reactions by introducing a specialized phase transfer catalytic system operating under mild micro-pressure conditions. By shifting away from traditional high-temperature atmospheric protocols, this innovation not only enhances the chemical yield and purity profiles but also fundamentally alters the economic feasibility of large-scale production. For global procurement teams and R&D directors, understanding the nuances of this process is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrafluorophthalimide derivatives has been plagued by severe thermodynamic and kinetic barriers that hinder commercial scalability. Traditional literature and prior art, such as the methods described in earlier patents like CN102627553A, typically rely on forcing the fluorination reaction at excessively high temperatures exceeding 150°C for prolonged durations often surpassing 10 hours. These harsh conditions inevitably lead to significant thermal degradation of the reactants, resulting in severe coking and the formation of complex impurity profiles that are difficult to separate. Furthermore, conventional atmospheric processes often struggle to achieve complete conversion, leaving behind substantial amounts of mono-, di-, and tri-fluorinated intermediates that compromise the overall purity of the final batch. The necessity for extensive post-reaction workup, including water addition, filtration, and drying steps, introduces multiple points of potential yield loss and increases the operational expenditure related to solvent recovery and waste treatment. Consequently, the inability to telescope this step directly into downstream decarboxylation reactions creates a disjointed manufacturing workflow that is ill-suited for modern continuous processing requirements.
The Novel Approach
In stark contrast to these legacy methods, the innovative process outlined in patent CN110407735B leverages a unique combination of a phosphorus-based phase transfer catalyst and controlled micro-pressure environments to drive the reaction forward with exceptional efficiency. By utilizing tetrakis(diethylamino)phosphonium bromide as the catalytic promoter, the system effectively enhances the nucleophilicity of the fluoride ion in an aprotic polar solvent, allowing the halogen exchange to proceed rapidly at significantly lower temperatures ranging between 110°C and 130°C. This reduction in thermal stress not only preserves the structural integrity of the phthalimide core but also drastically suppresses the formation of tar-like byproducts. The implementation of a slight positive pressure (0.01-0.5 MPa) further optimizes the reaction kinetics, ensuring that the conversion of 3,4,5,6-tetrachloro-N-methylphthalimide to the tetrafluoro derivative is both rapid and comprehensive. As illustrated in the reaction scheme below, this streamlined approach transforms a previously cumbersome multi-step isolation process into a highly efficient transformation suitable for direct telescoping.
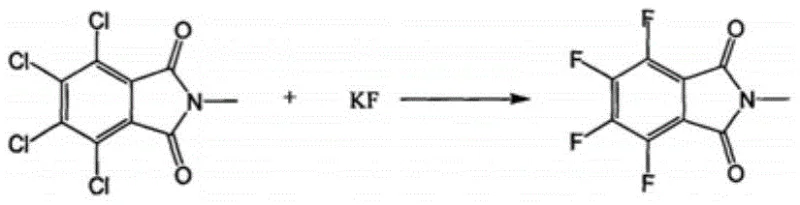
Mechanistic Insights into Phase Transfer Catalyzed Halogen Exchange
The core of this technological advancement lies in the sophisticated interplay between the phase transfer catalyst and the reaction medium, which fundamentally alters the solvation dynamics of the inorganic fluoride source. In standard nucleophilic aromatic substitution reactions, the lattice energy of potassium fluoride often limits its availability in organic solvents, necessitating extreme heat to overcome this barrier. However, the introduction of tetrakis(diethylamino)phosphonium bromide creates a lipophilic cationic species that effectively encapsulates the fluoride anion, dragging it into the organic phase where the chlorinated substrate resides. This "naked" fluoride ion exhibits heightened reactivity, allowing it to attack the electron-deficient aromatic ring of the tetrachloro-phthalimide with precision. The steric bulk and electronic properties of the diethylamino groups on the phosphorus atom are critical, as they prevent the catalyst from decomposing under the reaction conditions while maintaining optimal solubility in solvents like dimethyl sulfoxide (DMSO). This mechanistic efficiency ensures that the substitution of all four chlorine atoms occurs sequentially yet rapidly, minimizing the residence time of partially fluorinated intermediates that could otherwise undergo unwanted side reactions.
Furthermore, the strict control of water content within the solvent system plays a pivotal role in maintaining high product purity and preventing hydrolysis of the sensitive imide functionality. The patent specifies that the water content in the DMSO solvent must be rigorously controlled to less than 3%, and ideally reduced to below 0.1% after the addition of potassium fluoride. This dehydration step is crucial because water acts as a competing nucleophile and can lead to the hydrolysis of the anhydride or imide bonds, generating carboxylic acid impurities that are difficult to remove. By maintaining an anhydrous environment, the reaction selectively favors the formation of the carbon-fluorine bond over carbon-oxygen bond formation. Additionally, the ability to filter off the generated potassium chloride salt directly from the reaction mixture without quenching allows the resulting fluorination liquid to retain a purity of greater than 92%. This high level of crude purity is the key enabler for the subsequent decarboxylation step, eliminating the need for crystallization or chromatographic purification and thereby preserving the overall mass balance of the manufacturing campaign.
How to Synthesize 3,4,5,6-Tetrafluoro-N-methylphthalimide Efficiently
Implementing this green synthesis route requires precise adherence to the specified parameters regarding pressure, temperature, and reagent stoichiometry to replicate the high yields reported in the patent data. The process begins with the meticulous preparation of the reaction solvent, where DMSO is dehydrated and mixed with potassium fluoride under heating to ensure a homogenous and dry fluoride source. Once the catalyst and substrate are introduced under an inert nitrogen atmosphere, the system is pressurized slightly to maintain the optimized kinetic regime. The reaction is then allowed to proceed at a controlled temperature of approximately 120°C for a duration of 1.5 to 3 hours, which represents a drastic reduction in cycle time compared to traditional methods. Following the reaction, the mixture is cooled and subjected to pressure filtration to remove the inorganic salt byproduct, yielding a clear solution ready for immediate downstream processing. For a detailed breakdown of the specific operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Dehydrate the solvent DMSO to ensure water content is less than 3%, then add potassium fluoride and further dehydrate to less than 0.1%.
- Add 3,4,5,6-tetrachloro-N-methylphthalimide and the phase transfer catalyst tetrakis(diethylamino)phosphonium bromide, then replace air with nitrogen 2-3 times.
- Maintain system pressure at 0.01-0.2 MPa and react at 115-125°C for 1.5-3 hours, followed by pressure filtration to remove potassium chloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers profound advantages that extend well beyond simple chemical yield improvements, directly impacting the bottom line and supply chain resilience for API manufacturers. The most significant economic driver is the drastic simplification of the unit operations; by eliminating the isolation, washing, and drying steps typically required for intermediate purification, manufacturers can reduce capital expenditure on equipment and lower the operational costs associated with labor and energy consumption. The ability to telescope the fluorination step directly into the decarboxylation reaction means that the intermediate never leaves the liquid phase, which minimizes material handling losses and reduces the physical footprint required for production. This streamlining of the process flow translates into a more robust cost reduction in pharmaceutical intermediate manufacturing, allowing suppliers to offer more competitive pricing structures without compromising on margin. Moreover, the shortened reaction time from over 10 hours to merely 2-3 hours significantly increases the throughput capacity of existing reactor assets, effectively multiplying the production volume achievable within a fixed timeframe.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in energy intensity due to lower reaction temperatures contribute to a leaner cost structure. By avoiding the high-temperature coking issues associated with legacy methods, the frequency of reactor cleaning and maintenance is significantly reduced, leading to higher asset utilization rates. The qualitative shift towards a "telescoped" process removes the need for intermediate storage and quality control testing between steps, further compressing the working capital cycle and reducing inventory holding costs. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final fluoroquinolone APIs more affordable for healthcare systems globally.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials such as potassium fluoride and common aprotic solvents ensures that the supply chain is not vulnerable to the geopolitical risks often associated with exotic reagents. The robustness of the reaction conditions, specifically the tolerance for micro-pressure rather than high vacuum or extreme pressure, simplifies the engineering requirements for the manufacturing plant, reducing the risk of unplanned downtime due to equipment failure. Furthermore, the high consistency of the product quality, with purity levels consistently exceeding 92%, minimizes the risk of batch rejection and the need for reprocessing, thereby guaranteeing a steady and predictable flow of materials to downstream API synthesis facilities. This reliability is critical for maintaining the continuity of supply for essential antibiotics.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as the reduction of solvent usage and the minimization of waste generation, align perfectly with increasingly stringent environmental regulations. The absence of heavy metal contaminants simplifies the waste treatment protocol, reducing the environmental burden and the associated disposal costs. The process is inherently scalable, as demonstrated by the patent's emphasis on continuous industrial production, allowing manufacturers to ramp up from pilot scale to commercial tonnage with minimal process re-engineering. This scalability ensures that the technology can meet surging global demand for fluoroquinolone antibiotics without the long lead times typically required for new capacity installation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the rigorous testing performed to optimize catalyst loading, solvent ratios, and pressure conditions to ensure maximum efficiency. For further technical clarification or custom feasibility studies, our team is available to provide detailed support.
Q: What is the primary advantage of the micro-pressure condition in this synthesis?
A: Maintaining a micro-pressure of 0.01-0.5 MPa allows the reaction to proceed efficiently at lower temperatures (110-130°C) compared to conventional atmospheric methods requiring over 150°C, significantly reducing energy consumption and side reactions.
Q: How does this process improve supply chain continuity for fluoroquinolone production?
A: The process achieves a product purity of over 92% in the reaction liquid, allowing the fluorination solution to be directly used in the subsequent decarboxylation step without isolation, drying, or purification, thereby enabling continuous industrial production.
Q: Why is tetrakis(diethylamino)phosphonium bromide preferred as the catalyst?
A: This specific phase transfer catalyst effectively solubilizes potassium fluoride in the aprotic polar solvent, facilitating the nucleophilic substitution of chlorine atoms with fluorine while minimizing degradation side reactions common with other catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5,6-Tetrafluoro-N-methylphthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN110407735B requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with state-of-the-art reactors capable of handling the specific pressure and temperature requirements of this fluorination process, and our stringent purity specifications are enforced through rigorous QC labs that utilize advanced analytical instrumentation. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global regulatory landscape, providing you with a secure foundation for your API synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your next production campaign. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis route for your specific volume requirements. We encourage you to reach out today to obtain specific COA data from our recent batches and to discuss route feasibility assessments tailored to your project timelines. Let us help you optimize your supply chain and reduce your manufacturing costs with our premium grade 3,4,5,6-tetrafluoro-N-methylphthalimide.
