Revolutionizing Ambroxol Intermediate Production: A Deep Dive into High-Purity Transbroncho Alkali Refinement
Revolutionizing Ambroxol Intermediate Production: A Deep Dive into High-Purity Transbroncho Alkali Refinement
The pharmaceutical industry constantly seeks methodologies that balance high purity with operational efficiency, particularly in the synthesis of critical mucolytic agents like Ambroxol HCl. A significant breakthrough in this domain is detailed in patent CN103739502A, which discloses a novel process for separating and refining Transbroncho alkali, the pivotal free-base intermediate. Unlike traditional methods that often bypass the isolation of this intermediate to save time, this patented approach demonstrates that a dedicated purification step yields a product with purity exceeding 99%, drastically simplifying the subsequent salt formation. By leveraging a unique water-induced crystallization technique, manufacturers can eliminate persistent inorganic impurities that typically plague the final API, ensuring a superior quality profile for downstream processing.
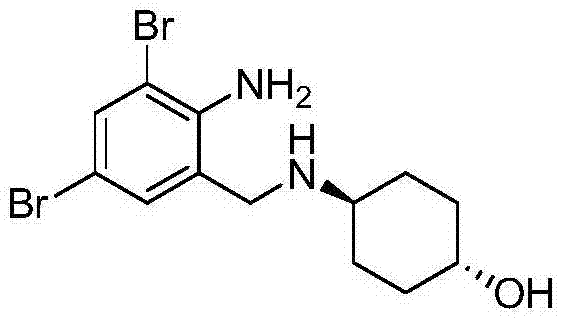
This technological advancement addresses a critical bottleneck in the supply chain of respiratory therapeutics. The ability to produce Transbroncho alkali with such high fidelity means that downstream processors can achieve final Ambroxol HCl specifications without the need for multiple, yield-eroding recrystallization cycles. For R&D directors and process chemists, this represents a shift from empirical trial-and-error to a mechanistic understanding of solubility-driven purification. The patent outlines a robust protocol where water acts not merely as a wash but as an anti-solvent to selectively precipitate the target molecule, leaving behind soluble byproducts such as sodium metaborate and unreacted starting materials. This level of control is essential for maintaining consistent batch-to-batch quality in a regulated GMP environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Ambroxol HCl has often favored a "one-pot" strategy where the reaction mixture is directly acidified to form the hydrochloride salt without isolating the free base intermediate. While this appears to save a unit operation, it introduces severe purification challenges that compromise the final product quality. As noted in prior art, including literature from the Chinese Journal of Modern Applied Pharmacy, direct salification results in a crude product that entraps significant amounts of inorganic salts, specifically metaboric acid and sodium chloride generated during the reduction step. These impurities are notoriously difficult to remove once incorporated into the API crystal lattice, often necessitating extensive and costly recrystallization processes that erode overall yield. Furthermore, alternative extraction-based methods described in older patents rely on organic solvents like methylene chloride, which pose environmental hazards and require complex solvent recovery systems due to boiling point similarities with reaction solvents like methanol.
The Novel Approach
The methodology presented in patent CN103739502A fundamentally rethinks this workflow by reintroducing the isolation of Transbroncho alkali as a distinct, optimized step. Instead of struggling to purify the salt, this approach purifies the free base, which possesses different solubility characteristics that are far more favorable for separation. The process utilizes a hydrophilic solvent system, preferably ethanol or methanol, and employs water as a crystallization inducer. By carefully controlling the addition of water and temperature, the Transbroncho alkali is forced out of the solution in a highly pure crystalline form, while the polar inorganic impurities remain dissolved in the aqueous mother liquor. This strategic decoupling of the reduction and salification steps ensures that the material entering the final acidification stage is already of exceptional purity, thereby guaranteeing a high-quality finished product with minimal additional processing.
Mechanistic Insights into Water-Induced Crystallization
The core innovation of this purification technology lies in the precise manipulation of solubility parameters within a hydrophilic medium. Transbroncho alkali exhibits poor solubility in water but is soluble in lower alcohols. The patented process exploits this differential by maintaining the reaction system in a hydrophilic state and then gradually introducing water. As the water content increases, the dielectric constant of the solvent mixture changes, reducing the solvation capacity for the organic amine. This triggers a controlled nucleation and growth phase where the Transbroncho alkali precipitates. Crucially, the byproducts of the sodium borohydride reduction, such as sodium metaborate, are highly water-soluble and remain in the supernatant. This mechanism effectively performs a "chemical filtration" where the physical act of crystallization separates the target molecule from ionic contaminants that would otherwise co-precipitate in a direct salification scenario.
Furthermore, the process incorporates a dual-stage crystallization and decolorization protocol to address organic impurities and color bodies. After the initial crude precipitation, the solid is redissolved in a warm hydrophilic solvent and treated with activated carbon. This step is vital for adsorbing trace organic impurities, such as unreacted 2-amino-3,5-dibromobenzaldehyde or Schiff base intermediates, which can affect the color and stability of the final API. Following filtration, a secondary crystallization is induced by adding water again under controlled cooling conditions. This recrystallization step further refines the crystal lattice, excluding any remaining occluded impurities and ensuring that the final Transbroncho alkali meets stringent purity specifications of greater than 99%. This rigorous attention to detail in the intermediate stage is what enables the production of pharmaceutical-grade Ambroxol HCl with superior consistency.
How to Synthesize Transbroncho Alkali Efficiently
The synthesis and purification of Transbroncho alkali via this patented route involves a sequence of precise operational steps designed to maximize yield and purity. The process begins with the condensation of 2-amino-3,5-dibromobenzaldehyde and trans-4-aminocyclohexanol in a hydrophilic solvent, followed by reduction with sodium borohydride. Once the reaction is complete, the innovative purification protocol takes over, utilizing water addition and temperature control to isolate the product. The following guide outlines the standardized procedure for implementing this high-efficiency workflow in a production setting.
- Primary Crystallization: Add water to the hydrophilic reaction solution containing Transbroncho alkali to induce precipitation, followed by filtration to obtain the crude product.
- Decolorization and Dissolution: Dissolve the crude product in a hydrophilic solvent (e.g., ethanol), treat with activated carbon to remove colored impurities, and filter to obtain a clear filtrate.
- Secondary Recrystallization: Add water to the filtrate to induce a second crystallization phase, filter the refined product, and dry to obtain high-purity Transbroncho alkali.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this purification technology translates into tangible operational efficiencies and risk mitigation. The elimination of complex liquid-liquid extraction steps using chlorinated solvents significantly reduces the environmental footprint and regulatory burden associated with solvent disposal and recovery. By relying primarily on water and ethanol, the process aligns with green chemistry principles, potentially lowering waste treatment costs and simplifying compliance with increasingly strict environmental regulations. Moreover, the simplified workflow reduces the number of unit operations required to reach the final API, which inherently shortens the production cycle time and enhances the overall throughput of the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substantial simplification of the purification train. Traditional methods often require multiple extraction cycles and extensive solvent swapping, which consume significant energy and raw materials. By contrast, this water-crystallization method minimizes solvent usage and eliminates the need for expensive solvent recovery distillation columns associated with separating close-boiling mixtures like methanol and methylene chloride. Additionally, the high purity of the intermediate reduces the loss of material during the final API recrystallization, leading to a higher overall mass balance and better utilization of expensive starting materials like dibromobenzaldehyde.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and scalability of this synthetic route. The reliance on commodity chemicals such as water and ethanol, rather than specialized extraction solvents, mitigates the risk of raw material shortages or price volatility. The process is also less sensitive to minor variations in reaction conditions compared to delicate extraction protocols, resulting in more consistent batch outcomes. This reliability ensures a steady flow of high-quality intermediates to downstream API manufacturers, reducing the likelihood of production delays caused by out-of-specification batches or lengthy rework procedures.
- Scalability and Environmental Compliance: From a scale-up perspective, crystallization is generally more predictable and easier to control in large reactors than multi-stage extractions. The absence of emulsion formation, a common issue in liquid-liquid extractions at scale, ensures smooth filtration and drying operations. Furthermore, the reduction in hazardous waste generation supports corporate sustainability goals and facilitates easier permitting for facility expansions. The ability to produce high-purity intermediates with a smaller environmental footprint positions suppliers as preferred partners for global pharmaceutical companies that prioritize sustainable sourcing in their vendor qualification audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced purification technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making.
Q: Why is isolating Transbroncho alkali superior to direct salification?
A: Direct salification often traps inorganic impurities like metaboric acid and sodium chloride within the crystal lattice, complicating final purification. Isolating the free base (alkali) first allows for the removal of these salts and unreacted aldehydes via water washing and crystallization, ensuring a significantly higher purity final API.
Q: What is the key mechanism behind this purification method?
A: The process exploits the poor solubility of Transbroncho alkali in water. By gradually adding water to a hydrophilic reaction system, the solubility limit is exceeded, causing the pure product to precipitate while soluble impurities like sodium borohydride byproducts remain in the aqueous phase.
Q: Can this process be scaled for industrial production?
A: Yes, the process is highly scalable as it eliminates complex extraction steps and hazardous solvent exchanges. It relies primarily on water and common hydrophilic solvents like ethanol, making it safer, more cost-effective, and easier to manage in large-scale reactors compared to traditional extraction methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Transbroncho Alkali Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final therapeutic agent is inextricably linked to the purity of its precursors. Our technical team has extensively analyzed the pathway described in CN103739502A and integrated similar high-efficiency crystallization principles into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Transbroncho alkali we supply meets the exacting standards required for modern pharmaceutical synthesis.
We invite potential partners to engage with us to explore how our advanced manufacturing capabilities can optimize your supply chain. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and discuss route feasibility assessments that demonstrate how our refined processes can deliver both economic and quality advantages for your Ambroxol HCl production needs.
