Scalable Production of High-Purity Cyclobutylamine Intermediates Using Novel Photochemical Methods
The synthesis of saturated nitrogen-containing heterocycles, particularly cyclobutylamine compounds, represents a significant frontier in modern organic chemistry due to their prevalence as active units in pharmaceuticals and agrochemicals. As detailed in the recent patent CN112794810B, a novel synthetic methodology has been developed that leverages continuous photochemical reactions to overcome the historical challenges associated with constructing the strained four-membered azetidine ring. Traditional approaches often struggle with the high ring energy barrier of approximately 25.2 kcal/mol, leading to unstable intermediates and undesirable yields. This breakthrough technology utilizes a specific combination of olefin and imine compounds under the catalytic action of tailored photosensitizers, facilitating a [2+2] cycloaddition that proceeds with remarkable efficiency. By shifting from batch processes to continuous photochemical flow systems, this innovation not only enhances the yield of target products but also establishes a robust foundation for the large-scale synthesis of complex amine derivatives required by the global fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of azetidine structures has relied heavily on methods such as intramolecular nucleophilic substitution, reduction cyclization of halogenated imines, or electrophilic activation of allylamines. While methods reported by researchers like Meyers and De Kimpe have provided foundational routes, they are frequently plagued by significant operational drawbacks that hinder commercial viability. For instance, nucleophilic substitution often requires harsh leaving groups like p-toluenesulfonate and stringent conditions that limit functional group tolerance. Furthermore, reduction cyclization using reagents like NaBH3CN in acetic acid introduces safety concerns regarding cyanide handling and waste disposal. Perhaps most critically, these conventional thermal pathways often lack precise stereochemical control, resulting in mixtures of isomers that are difficult and costly to separate, thereby reducing the overall process efficiency and increasing the cost of goods for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112794810B employs a continuous photochemical [2+2] cycloaddition between acyclic imines and olefins, offering a transformative solution to these legacy issues. By utilizing light energy to access excited triplet states via intersystem crossing, the reaction bypasses the high thermal activation barriers associated with ring closure. This photo-induced pathway allows the reaction to proceed under exceptionally mild conditions, typically between 20-30°C, which preserves sensitive functional groups that would otherwise degrade under thermal stress. The implementation of continuous flow technology ensures uniform irradiation and efficient heat dissipation, eliminating the hot spots common in batch photochemistry. Consequently, this approach delivers superior stereochemical outcomes with significantly reduced side reactions, providing a cleaner reaction profile that simplifies downstream purification and boosts the overall yield of the desired cyclobutylamine scaffold.
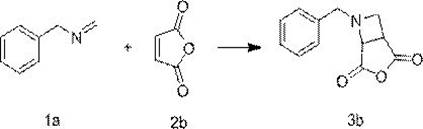
Mechanistic Insights into Photosensitized [2+2] Cycloaddition
The core of this synthetic advancement lies in the precise manipulation of electronic states through photosensitization. The mechanism initiates when the substrate or the added photosensitizer absorbs photons from a light source with an emission wavelength typically ranging from 280 nm to 480 nm, with 365 nm being optimal for organic sensitizers like benzophenone. Upon excitation, the molecule transitions from its ground singlet state to an excited singlet state, followed rapidly by intersystem crossing to a longer-lived triplet state. This triplet species is the key reactive intermediate that engages the olefin double bond in a concerted [2+2] cycloaddition. The choice of photosensitizer is critical; compounds such as xanthone, thioxanthone, and benzil act as efficient energy transfer agents, while metal complexes like ruthenium terpyridine chloride or copper trifluoromethanesulfonate offer alternative excitation pathways at 450 nm. This mechanistic flexibility allows chemists to tune the reaction conditions to match the absorption profiles of diverse substrates, ensuring high conversion rates across a broad spectrum of imine and olefin variations.
Furthermore, the continuous nature of the photochemical process plays a pivotal role in impurity control and product stability. In traditional batch photoreactors, uneven light penetration can lead to over-irradiation of some molecules while others remain unreacted, fostering degradation pathways and byproduct formation. The continuous flow system described in the patent circulates the reaction mixture through an FEP (perfluoroethylene propylene copolymer) tube, ensuring that every molecule receives a consistent photon flux. This uniformity minimizes the formation of secondary amines derived from imine decomposition and prevents the accumulation of thermal byproducts. The subsequent workup procedure, involving sequential washing with ammonium chloride and saturated sodium bicarbonate, effectively removes residual catalysts and acidic impurities. This rigorous control over the reaction environment results in a crude product of high purity, reducing the burden on final crystallization or chromatography steps and ensuring the structural integrity of the sensitive four-membered ring.
How to Synthesize Cyclobutylamine Efficiently
To implement this advanced synthesis route effectively, operators must adhere to precise parameters regarding substrate mixing, light exposure, and post-reaction processing. The process begins with the preparation of a homogeneous mixture containing the olefin compound, the imine substrate, and the selected photosensitizer dissolved in a compatible solvent such as dichloromethane or acetonitrile. This mixture is then introduced into a continuous flow photoreactor where it is subjected to controlled irradiation. The specific operational details, including flow rates, residence times, and exact stoichiometric ratios required to maximize yield and minimize waste, are critical for successful replication. For a comprehensive understanding of the standardized operating procedures and safety protocols necessary for executing this synthesis, please refer to the detailed technical guidelines provided below.
- Mix the olefin compound (Formula I) and imine compound (Formula II) with a photosensitizer such as benzophenone or xanthone in a suitable solvent like dichloromethane.
- Pump the reaction mixture through an FEP tube into a photoreaction zone and irradiate continuously with a light source emitting at 365-450 nm while maintaining temperature between 20-30°C.
- Wash the crude product sequentially with aqueous ammonium chloride and saturated sodium bicarbonate solutions, then dry over anhydrous sodium sulfate and concentrate to isolate the target cyclobutylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this continuous photochemical methodology offers substantial strategic benefits beyond mere technical novelty. The shift away from harsh thermal conditions and toxic reagents like cyanides significantly simplifies the regulatory compliance landscape, reducing the administrative burden associated with hazardous material handling and disposal. Moreover, the ability to operate at ambient temperatures (20-30°C) drastically lowers energy consumption compared to high-temperature reflux processes, contributing to a more sustainable and cost-effective manufacturing footprint. The robustness of the continuous flow system also意味着 enhanced supply chain reliability, as the modular nature of flow reactors allows for rapid capacity expansion without the need for massive infrastructure investments, ensuring consistent availability of critical intermediates even during periods of surging demand.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of organic photosensitizers like benzophenone or xanthone represents a direct material cost saving. Additionally, the high stereoselectivity of the photochemical [2+2] cycloaddition reduces the need for costly chiral resolution or extensive purification steps to remove isomeric impurities. By streamlining the synthesis into a continuous process, labor costs are minimized through automation, and solvent usage is optimized through efficient recycling loops inherent to flow chemistry systems, collectively driving down the total cost of ownership for the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as simple olefins and imines, combined with the use of common solvents like dichloromethane and toluene, mitigates the risk of raw material shortages. The continuous nature of the reaction allows for just-in-time manufacturing capabilities, reducing the need for large inventory buffers of unstable intermediates. This agility enables suppliers to respond more rapidly to fluctuations in market demand, ensuring that downstream pharmaceutical manufacturers receive their critical building blocks on schedule without the delays often associated with complex batch campaigns.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a bottleneck due to the Beer-Lambert law limiting light penetration in large vessels. However, the continuous flow approach described in the patent circumvents this by maintaining a thin film of reaction mixture within the FEP tubing, ensuring consistent light exposure regardless of production volume. This inherent scalability facilitates a seamless transition from gram-scale R&D to multi-ton commercial production. Furthermore, the process generates less hazardous waste compared to traditional methods, aligning with increasingly stringent global environmental regulations and supporting corporate sustainability goals without compromising on output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, aiming to clarify the operational feasibility and strategic value of adopting this method for industrial applications. Understanding these nuances is essential for technical teams evaluating the integration of continuous flow photochemistry into their existing manufacturing portfolios.
Q: What are the primary advantages of this photochemical method over traditional nucleophilic substitution?
A: Unlike traditional methods that often suffer from poor stereochemical control and harsh conditions, this continuous photochemical approach operates at mild temperatures (20-30°C) and utilizes specific wavelengths to achieve higher stereoselectivity and reduced side reactions.
Q: Which photosensitizers are most effective for this cycloaddition reaction?
A: The patent identifies several effective photosensitizers including benzophenone, xanthone, thioxanthone, and benzil. Benzophenone and xanthone generally provide superior yields (up to 69%) when irradiated at 365 nm, while copper complexes can be used at 450 nm.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for scalability using continuous flow technology. By circulating the reaction mixture through an FEP tube within a photoreaction zone, heat dissipation is managed effectively, allowing for prolonged reaction times and consistent product quality essential for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclobutylamine Supplier
As the demand for complex heterocyclic building blocks continues to rise in the development of next-generation therapeutics, partnering with a manufacturer capable of executing advanced synthetic routes is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art continuous flow photoreactors and stringent purity specifications that ensure every batch of cyclobutylamine intermediate meets the highest quality standards. With our rigorous QC labs and dedicated process development team, we are uniquely positioned to translate the innovative methods described in patent CN112794810B into reliable, commercial-grade supply solutions for our global partners.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains for these critical intermediates. By leveraging our technical expertise, you can secure a stable source of high-purity materials while benefiting from our commitment to process efficiency. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can accelerate your drug development timelines and reduce overall production costs.
