Revolutionizing 9-(4-Bromophenyl)Carbazole Production via Rhodium-Catalyzed Dehydroxy Coupling for OLED Materials
Revolutionizing 9-(4-Bromophenyl)Carbazole Production via Rhodium-Catalyzed Dehydroxy Coupling for OLED Materials
The landscape of organic electronic material synthesis is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective, and highly selective manufacturing processes. A pivotal advancement in this domain is detailed in patent CN114853658A, which discloses a novel synthesis method for 9-(4-bromophenyl)carbazole, a critical intermediate for high-performance OLED materials. This technology shifts the paradigm from traditional halogen-heavy coupling reactions to a sophisticated dehydroxy C-N coupling strategy utilizing carbazole and p-bromophenol as primary feedstocks. By leveraging a specialized rhodium catalyst system paired with sterically demanding N-heterocyclic carbene (NHC) ligands, this method achieves exceptional atom economy and reaction selectivity. For R&D directors and procurement strategists in the fine chemical sector, this represents a substantial opportunity to optimize supply chains for next-generation display and lighting technologies while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 9-(4-bromophenyl)carbazole has relied heavily on Ullmann or Buchwald-Hartwig coupling reactions involving p-bromoiodobenzene or p-dibromobenzene. As illustrated in the reaction schemes below, these traditional pathways are fraught with significant economic and technical inefficiencies. The use of p-bromoiodobenzene, while effective, introduces prohibitive raw material costs due to the high price of iodine derivatives, and the reaction often requires extended reflux times of up to 24 hours. Furthermore, the reliance on copper or palladium catalysts in these systems frequently leads to unavoidable side reactions, such as halogen exchange, where sodium iodide generated in situ reacts with the product to form difficult-to-separate impurities like 9-(4-iodophenyl)carbazole.
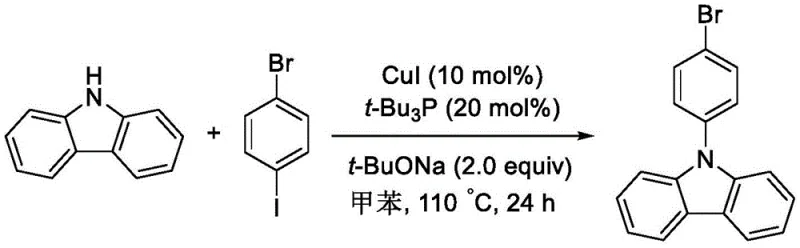
Moreover, alternative routes utilizing p-dibromobenzene suffer from severe selectivity issues. Under the harsh conditions often required, such as reflux temperatures reaching 246°C, the reaction lacks the precision to stop at the mono-substituted stage. This results in the formation of significant quantities of the disubstituted byproduct, 9,9'-(1,4-phenyl)di-9H-carbazole, as depicted in the following diagram. The structural similarity between the desired mono-substituted product and this disubstituted impurity makes purification extremely challenging, typically necessitating resource-intensive silica gel column chromatography. This not only escalates production costs but also generates substantial solvent waste, rendering these conventional methods unsuitable for sustainable, large-scale industrial application.
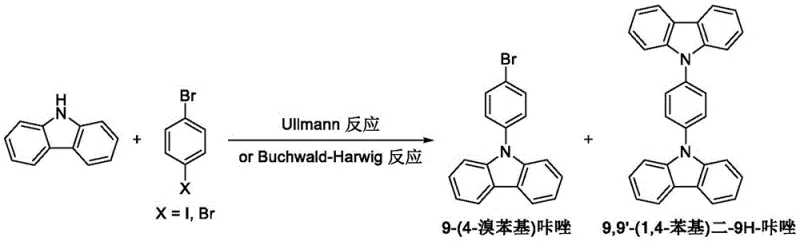
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114853658A introduces a groundbreaking dehydroxy C-N coupling reaction that fundamentally alters the substrate strategy. By replacing expensive and waste-generating dihalobenzenes with p-bromophenol, the process utilizes a much more economical and readily available starting material. The reaction proceeds under significantly milder conditions, typically between 60°C and 130°C, eliminating the energy-intensive requirements of high-temperature reflux. Most critically, the only byproduct of this transformation is water, which drastically simplifies the post-reaction workup and eliminates the generation of stoichiometric amounts of inorganic salt waste like sodium bromide or sodium iodide. This shift not only improves the atom utilization rate but also aligns perfectly with green chemistry principles, offering a streamlined pathway that bypasses the need for complex chromatographic purification.
Mechanistic Insights into Rhodium-Catalyzed Dehydroxy C-N Coupling
The success of this novel synthetic route hinges on the precise engineering of the catalytic system, specifically the synergy between the rhodium metal center and the specialized imidazole-based N-heterocyclic carbene (NHC) ligands. Unlike traditional phosphine ligands which may lack the necessary stability or steric profile, the NHC ligands employed here possess strong coordination chelation capabilities and significant steric bulk. When coordinated with the rhodium salt, these ligands facilitate the formation of a stable cyclic rhodium intermediate that accurately targets the N-H bond of the carbazole and the hydroxyl group of the p-bromophenol. This specific interaction lowers the activation energy for the C-N bond formation while simultaneously protecting the bromine atom on the phenol ring from participating in competitive dehalogenation reactions.
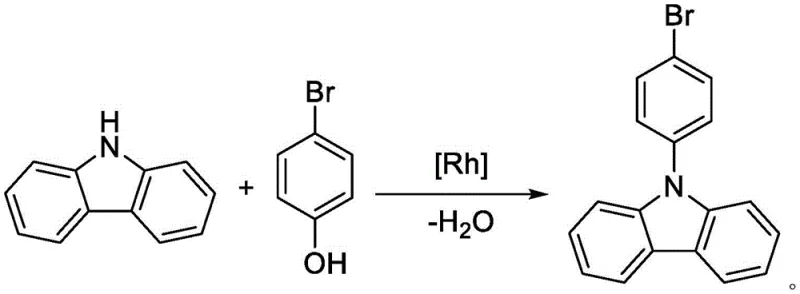
Furthermore, the steric hindrance provided by the NHC ligand plays a pivotal role in controlling reaction selectivity. In conventional systems, the lack of steric control often allows a second molecule of carbazole to attack the intermediate, leading to the aforementioned disubstituted byproducts. However, in this rhodium-catalyzed system, the bulky ligand effectively inhibits the free rotation of the reaction intermediate and physically blocks the approach of a second carbazole molecule after the first coupling event is complete. This ensures that the reaction stops exclusively at the mono-substituted 9-(4-bromophenyl)carbazole stage. The result is a reaction profile with exceptional specificity, where the functional group tolerance is high, and the formation of impurities is minimized, allowing for high-purity isolation through simple crystallization techniques rather than complex separation processes.
How to Synthesize 9-(4-Bromophenyl)Carbazole Efficiently
The operational simplicity of this rhodium-catalyzed protocol makes it highly attractive for process chemists aiming to scale up production. The general procedure involves charging a reactor with carbazole and p-bromophenol in a molar ratio optimized for high conversion, typically around 1:1 to 1:1.2. To this mixture, a catalytic amount of a rhodium precursor, such as [Cp*RhCl2]2, and a specific NHC ligand are added along with a suitable base like sodium carbonate or potassium phosphate. The reaction is conducted in common organic solvents such as n-heptane, toluene, or 2-methyltetrahydrofuran under an inert nitrogen atmosphere. Following the reaction period of 6 to 12 hours at moderate temperatures, the product can be isolated through standard filtration and washing steps, followed by solvent recovery and crystallization.
- Charge a reactor with carbazole, p-bromophenol, rhodium catalyst, NHC ligand, base, and solvent under inert gas.
- Heat the mixture to 80-110°C and stir for 6-12 hours to facilitate dehydroxy C-N coupling.
- Cool, filter, wash, extract, remove solvent, and crystallize the crude product to obtain high-purity material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers compelling strategic advantages that extend beyond mere technical feasibility. The transition from halogenated benzene derivatives to phenolic substrates represents a fundamental shift in cost structure and supply reliability. By eliminating the dependency on volatile and expensive iodine-containing reagents, manufacturers can stabilize their raw material costs and reduce exposure to supply chain disruptions associated with specialty halogenated compounds. Additionally, the drastic reduction in purification complexity translates directly into lower operational expenditures, as the elimination of column chromatography reduces both solvent consumption and labor hours required for processing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost raw materials with commodity chemicals and the simplification of downstream processing. The use of p-bromophenol instead of p-bromoiodobenzene removes a significant cost driver, as iodine derivatives are notoriously expensive and subject to market fluctuations. Furthermore, the ability to purify the final product via crystallization rather than silica gel chromatography significantly reduces the consumption of organic solvents and stationary phases. This reduction in material usage, combined with the higher atom economy of generating water as the sole byproduct, leads to substantial overall cost savings in the manufacturing of this key OLED intermediate.
- Enhanced Supply Chain Reliability: From a sourcing perspective, p-bromophenol is a widely available bulk chemical with a robust global supply chain, unlike specialized dihalobenzenes which may have limited suppliers. This availability ensures greater continuity of supply and reduces the risk of production delays caused by raw material shortages. The milder reaction conditions also imply less stress on production equipment, potentially extending the lifespan of reactors and reducing maintenance downtime. Consequently, manufacturers can maintain more consistent production schedules and meet delivery commitments with higher reliability, a critical factor for suppliers serving the fast-paced electronics industry.
- Scalability and Environmental Compliance: The environmental profile of this synthesis method is markedly superior to conventional routes, addressing growing regulatory pressures on chemical manufacturing. The avoidance of stoichiometric salt waste generation simplifies wastewater treatment and reduces the burden on effluent management systems. Moreover, the mild operating temperatures and the use of recyclable solvents enhance the safety profile of the process, making it easier to scale from pilot plant to commercial production without encountering the thermal hazards associated with high-temperature reflux. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the method's practical implications for industrial application.
Q: Why is the Rh-catalyzed phenol route superior to traditional Ullmann coupling?
A: The Rh-catalyzed route uses cheaper p-bromophenol instead of expensive p-bromoiodobenzene or p-dibromobenzene, generates water instead of salt waste, and avoids difficult-to-remove disubstituted byproducts.
Q: What is the role of the NHC ligand in this synthesis?
A: The imidazole-based NHC ligand provides strong coordination and large steric hindrance, which stabilizes the rhodium intermediate and prevents over-reaction, ensuring high selectivity for the mono-substituted product.
Q: Does this process require column chromatography for purification?
A: No, unlike traditional methods that often require silica gel column chromatography, this novel method allows for purification via simple filtration and crystallization, significantly reducing processing costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-(4-Bromophenyl)Carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Rh-mediated dehydroxy coupling described in CN114853658A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for high-performance electronic materials, guaranteeing that every batch of 9-(4-bromophenyl)carbazole delivers the consistency your R&D teams demand.
We invite you to collaborate with us to leverage these technological advancements for your supply chain optimization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener route can improve your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to jointly engineer a supply solution that balances performance, cost, and sustainability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
