Scalable Microbial Synthesis of Critical Corticosteroid Intermediates for Global Pharma Supply Chains
Scalable Microbial Synthesis of Critical Corticosteroid Intermediates for Global Pharma Supply Chains
The global pharmaceutical landscape is witnessing a pivotal shift in the production of corticosteroids, driven by the transition from traditional diosgenin-based starting materials to more sustainable phytosterol-derived pathways. Central to this evolution is the efficient synthesis of 11,17α-substituted pregna-1,4-diene-3,20-dione, a critical intermediate often referred to as Intermediate B. Patent CN102206696A discloses a groundbreaking preparation method that utilizes Arthrobacter Simplex (ATCC 6946) to catalyze the 1,2-dehydrogenation of the corresponding 4-ene precursors. This biotechnological approach addresses long-standing challenges in steroid manufacturing, offering a robust solution for producing high-purity intermediates essential for the next generation of anti-inflammatory and immunosuppressive therapies. By leveraging specific microbial strains, this technology enables industrial-scale production with unprecedented substrate loading capacities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of steroid hormones relied heavily on diosgenin extracted from wild yams, a process that often bypassed Intermediate B entirely due to economic constraints and synthetic route limitations. When chemical dehydrogenation methods were employed to introduce the 1,4-diene system, they frequently suffered from harsh reaction conditions, requiring toxic reagents, extreme temperatures, and complex protection-deprotection sequences. These traditional chemical routes often resulted in lower overall yields, significant environmental waste, and difficulties in controlling stereochemistry at the 11 and 17 positions. Furthermore, the inability to efficiently process high concentrations of hydrophobic steroid substrates in aqueous media limited the throughput of earlier biological attempts, making them less viable for large-scale commercial adoption compared to established chemical syntheses.
The Novel Approach
The methodology outlined in CN102206696A represents a paradigm shift by employing a specialized microbial fermentation process using Arthrobacter Simplex. Unlike previous biological methods that struggled with low substrate solubility and poor conversion rates, this novel approach allows for substrate feeding concentrations as high as 7% (70g/L). The process utilizes a carefully optimized culture medium containing nitrogen sources like peptone and yeast extract, alongside corn steep liquor and glucose, to sustain high cell density and enzyme activity. By introducing the substrate either as a dry heat-sterilized powder or dissolved in water-miscible organic solvents such as ethanol or DMF, the method ensures excellent bioavailability. This flexibility in substrate introduction, combined with precise pH control, facilitates a highly efficient 1,2-dehydrogenation reaction that transforms Intermediate A directly into the valuable Intermediate B with minimal byproduct formation.
Mechanistic Insights into Arthrobacter Simplex-Catalyzed Dehydrogenation
The core of this technological advancement lies in the specific enzymatic activity of Arthrobacter Simplex, which possesses a potent 1,2-dehydrogenase capable of introducing a double bond between the C1 and C2 positions of the steroid nucleus. This biocatalytic transformation is highly regioselective, avoiding unwanted side reactions at other sensitive functional groups such as the C3 ketone or the C20 side chain. The reaction mechanism relies on the maintenance of a specific physiological environment where the microbial cells remain metabolically active throughout the 48-hour fermentation period. Critical to this success is the strict control of the fermentation broth pH, which must be maintained within the narrow window of 6.9 to 7.2. Experimental data indicates that deviation from this optimal pH range leads to a marked decrease in dehydrogenase activity, thereby reducing the conversion efficiency. Additionally, the process temperature is tightly regulated at 30±1°C to maximize enzyme stability while preventing thermal denaturation.
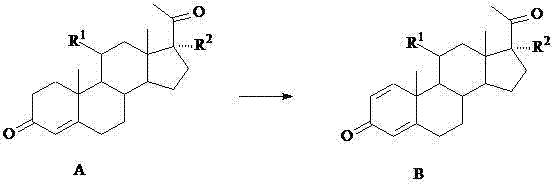
Following the biotransformation, the downstream processing is designed to maximize recovery and purity. The fermentation broth is filtered to collect the biomass containing the product, which is then extracted using organic solvents such as ethyl acetate or dichloromethane. A crucial step involves decolorization using activated carbon under reflux conditions, which effectively removes pigments and trace impurities generated during fermentation. The subsequent concentration and crystallization steps yield the final product with a purity exceeding 97% and a total yield (including mother liquor recovery) of over 85%. This high level of purity is essential for pharmaceutical applications, as it minimizes the burden on subsequent synthetic steps and ensures the final API meets stringent regulatory standards for impurity profiles. The versatility of this method is further demonstrated by its ability to accommodate various substituents at the R1 and R2 positions, including hydroxyl, acetoxy, and keto groups.
How to Synthesize 11,17α-substituted pregna-1,4-diene-3,20-dione Efficiently
The synthesis protocol described in the patent provides a comprehensive framework for replicating this high-efficiency transformation in a commercial setting. It details the precise composition of the culture medium, the sterilization parameters, and the critical timing for substrate addition relative to the growth phase of the bacteria. Operators must ensure that the seed culture reaches the appropriate optical density and enzyme activity levels, indicated by TTC staining, before initiating the bioconversion. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring consistent results across different batch sizes.
- Prepare the culture medium containing nitrogen sources, inorganic salts, glucose, and corn steep liquor, followed by sterilization at 120°C.
- Inoculate with Arthrobacter Simplex and cultivate at 30°C before adding the substrate (Intermediate A) at concentrations up to 7%.
- Maintain fermentation at pH 6.9-7.2 for 48 hours, then filter, decolorize with activated carbon, and crystallize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microbial fermentation technology offers substantial strategic advantages over traditional chemical synthesis routes. The ability to operate at high substrate concentrations directly translates to improved volumetric productivity, meaning that manufacturers can produce larger quantities of the intermediate using existing fermentation infrastructure without proportional increases in capital expenditure. This efficiency gain is critical for meeting the growing global demand for corticosteroids while maintaining competitive pricing structures. Furthermore, the biological nature of the process eliminates the need for expensive and hazardous heavy metal catalysts or stoichiometric oxidants, significantly reducing the cost of raw materials and waste disposal. The simplified downstream processing, characterized by straightforward filtration and crystallization, further contributes to overall cost reduction by shortening the production cycle time.
- Cost Reduction in Manufacturing: The elimination of complex chemical protection groups and harsh reagents drastically simplifies the production workflow, leading to significant operational savings. By utilizing renewable biological catalysts instead of precious metal complexes, the process reduces dependency on volatile commodity markets for rare earth elements. The high conversion rate (>90%) minimizes the loss of expensive steroid starting materials, ensuring that the majority of the input mass is converted into valuable product. Additionally, the recovery of mother liquor allows for the recycling of unreacted substrate, further enhancing the material efficiency and lowering the effective cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the Arthrobacter Simplex strain ensures consistent production outcomes, reducing the risk of batch failures that can disrupt supply schedules. The use of readily available agricultural byproducts like corn steep liquor and soybean cake powder as nutrient sources insulates the supply chain from fluctuations in specialized chemical feedstock availability. This reliance on stable, bulk commodities enhances the predictability of lead times and secures the continuity of supply for downstream API manufacturers. The scalability of the fermentation process from laboratory shake flasks to multi-ton industrial fermenters guarantees that supply can be rapidly ramped up to meet surges in market demand.
- Scalability and Environmental Compliance: The aqueous-based fermentation system inherently generates less hazardous waste compared to solvent-intensive chemical synthesis, facilitating easier compliance with increasingly stringent environmental regulations. The reduction in organic solvent usage during the reaction phase lowers the facility's VOC emissions and fire safety risks. The process is designed for seamless scale-up, as demonstrated by the successful transition from small-scale flask experiments to 20-liter fermenter trials without loss of efficiency. This scalability ensures that the technology remains viable and cost-effective as production volumes increase, supporting long-term sustainability goals for pharmaceutical manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microbial dehydrogenation technology. These insights are derived directly from the experimental data and specifications provided in the patent documentation, offering clarity on process capabilities and quality standards. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing synthesis pipelines.
Q: What is the maximum substrate concentration achievable in this fermentation process?
A: According to patent CN102206696A, the process supports substrate feeding concentrations up to 7% (70g/L), which is significantly higher than many conventional microbial methods.
Q: What represents the critical control parameter for enzyme activity in this reaction?
A: The pH value is critical; the dehydrogenase activity of Arthrobacter Simplex is optimal between pH 6.9 and 7.2. Deviations outside this range significantly reduce conversion rates.
Q: Can this method handle various 11,17α-substituted substrates?
A: Yes, the method is versatile and successfully converts various derivatives including 11α,17α-dihydroxy, 17α-hydroxy, and 17α-acetate substituted pregna-4-enes with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11,17α-substituted pregna-1,4-diene-3,20-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient manufacture of life-saving corticosteroids. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 11,17α-substituted pregna-1,4-diene-3,20-dione meets the highest industry standards. We are committed to leveraging advanced biotechnological processes like the one described in CN102206696A to deliver superior value to our global partners.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in steroid intermediate synthesis can accelerate your drug development timelines.
