Advanced Manufacturing of Eletriptan Intermediates: A Novel Metal-Free Synthetic Route
Advanced Manufacturing of Eletriptan Intermediates: A Novel Metal-Free Synthetic Route
The pharmaceutical landscape for migraine treatment relies heavily on the efficient production of high-quality active pharmaceutical ingredients (APIs), with Eletriptan standing out as a potent 5-HT1B/1D receptor agonist. Patent CN102786514B introduces a groundbreaking preparation method that addresses critical bottlenecks in traditional synthesis, offering a pathway that is both chemically robust and commercially viable. This technical insight report analyzes the novel four-step sequence detailed in the patent, which transforms a bromo-substituted indole precursor into the final therapeutic agent through a series of highly controlled organic transformations. By eliminating the dependency on transition metal catalysts and harsh reducing agents, this route not only enhances the environmental profile of the manufacturing process but also streamlines the purification workflow, ensuring a reliable eletriptan intermediate supplier can meet stringent global quality standards.
The significance of this patent lies in its ability to deliver high yields while maintaining exceptional product purity, a dual requirement that often challenges process chemists. The described methodology leverages well-understood reaction mechanisms such as N-protection, organolithium-mediated carbon-carbon bond formation, and nucleophilic substitution, all of which are amenable to rigorous process control. For R&D directors and procurement managers alike, understanding the nuances of this synthetic strategy is paramount for evaluating potential technology transfers or sourcing partnerships. The following analysis dissects the chemical logic, operational parameters, and commercial implications of this innovative approach to Eletriptan manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex indole-based therapeutics like Eletriptan has been plagued by reliance on expensive transition metal catalysts, such as palladium or platinum, which are required for key coupling reactions. These conventional routes often necessitate rigorous downstream processing to remove trace metal residues to levels compliant with ICH Q3D guidelines, adding significant cost and time to the production cycle. Furthermore, traditional methods may employ strong reducing agents that pose safety hazards and generate substantial quantities of hazardous waste, complicating environmental compliance and increasing the burden on waste treatment facilities. The cumulative effect of these factors is a manufacturing process that is not only costly but also fragile, with potential supply chain disruptions arising from the scarcity of specialized catalysts or the complexity of waste disposal logistics.
The Novel Approach
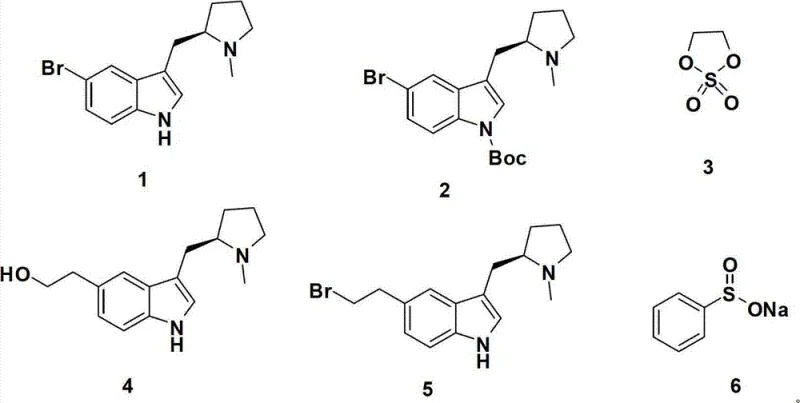
In stark contrast, the method disclosed in CN102786514B utilizes a metal-free strategy that fundamentally alters the economic and operational dynamics of Eletriptan production. As illustrated in the reaction scheme below, the process initiates with the protection of the indole nitrogen, followed by a lithiation step that enables the precise installation of the side chain using ethylene sulfate. This approach bypasses the need for cross-coupling catalysts entirely, replacing them with readily available and cost-effective reagents like n-butyllithium and sodium benzenesulfinate. The elimination of heavy metals simplifies the workup procedure, as there is no need for specialized scavenging resins or extensive filtration steps to ensure metal clearance.
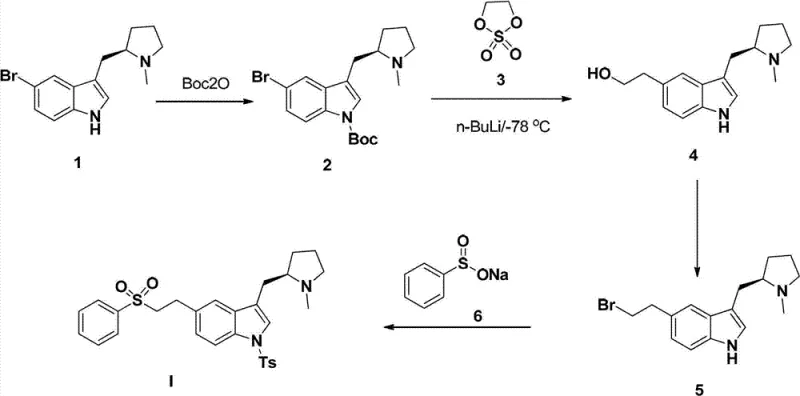
Furthermore, the novel route demonstrates remarkable versatility in solvent selection, utilizing common industrial solvents such as dichloromethane, tetrahydrofuran, and ethanol. This flexibility allows for easier solvent recovery and recycling, contributing to a greener manufacturing footprint. The stepwise progression from the protected indole to the final sulfone product is characterized by high conversion rates and manageable reaction conditions, typically ranging from cryogenic temperatures for lithiation to mild reflux for substitution. This balance of reactivity and control ensures that the process is not only scientifically elegant but also practically robust for large-scale operations, offering a compelling alternative to legacy synthesis methods.
Mechanistic Insights into the Metal-Free Substitution and Cyclization Strategy
The core of this synthetic innovation lies in the strategic manipulation of the indole scaffold to introduce the necessary functional groups with high regioselectivity. The initial protection of the indole nitrogen with di-tert-butyl dicarbonate (Boc2O) serves a dual purpose: it prevents unwanted side reactions at the nitrogen atom during the subsequent lithiation step and enhances the solubility of the intermediate in organic media. The use of a catalytic amount of DMAP (4-dimethylaminopyridine) accelerates this acylation, ensuring rapid and complete conversion to the N-Boc protected intermediate (Compound 2). This protection group is stable enough to withstand the harsh conditions of the next step yet can be removed or transformed as needed, although in this specific route, it facilitates the directed metallation at the C5 position.
The subsequent lithiation using n-BuLi at low temperatures (e.g., -78°C) generates a highly reactive aryl lithium species at the 5-position of the indole ring, ortho to the bromine substituent or directed by the nitrogen protecting group depending on the specific substrate electronics. This nucleophilic center then attacks ethylene sulfate (Compound 3), a cyclic sulfonate ester, resulting in the ring-opening and formation of a hydroxyethyl side chain. The mechanistic elegance continues in the final step, where the hydroxyl group is converted to a leaving group (bromide) and subsequently displaced by the sulfur nucleophile from sodium benzenesulfinate. This SN2-type substitution installs the phenylsulfonyl moiety, completing the construction of the Eletriptan pharmacophore without the need for oxidation states changes that typically require harsh oxidants.
Impurity control is inherently built into this mechanism. By avoiding radical pathways or transition metal cycles, the formation of homocoupling byproducts or metal-complexed impurities is minimized. The use of stoichiometric reagents allows for precise control over reaction kinetics, reducing the likelihood of over-reaction or decomposition. Additionally, the intermediate compounds (4 and 5) possess distinct physical properties that facilitate purification via extraction and crystallization, effectively scrubbing away unreacted starting materials and minor side products before they can propagate through the synthesis. This inherent cleanliness of the reaction pathway is a critical factor in achieving the high purity specifications required for pharmaceutical intermediates.
How to Synthesize Eletriptan Efficiently
The practical execution of this synthesis requires careful attention to temperature control and reagent addition rates, particularly during the exothermic lithiation and ring-opening steps. The patent provides detailed embodiments that serve as a blueprint for scaling this chemistry from gram to kilogram quantities. Operators must ensure anhydrous conditions for the organolithium step to prevent quenching of the reactive intermediate, while the subsequent hydrolysis and substitution steps benefit from vigorous stirring to maintain phase contact. The standardized protocol outlined in the patent ensures reproducibility, making it an ideal candidate for technology transfer to manufacturing sites. Detailed standardized synthesis steps are provided in the guide below.
- Protect the indole nitrogen of the starting bromo-indole (Compound 1) using di-tert-butyl dicarbonate (Boc2O) in DCM with TEA and DMAP to form Compound 2.
- Perform lithiation on Compound 2 using n-BuLi at low temperature (-78°C), followed by reaction with ethylene sulfate (Compound 3) and subsequent acid hydrolysis to yield the hydroxyethyl derivative (Compound 4).
- Convert the hydroxyl group of Compound 4 to a bromide using hydrobromic acid (HBr) under reflux conditions to generate the alkyl bromide intermediate (Compound 5).
- React Compound 5 with sodium benzenesulfinate (Compound 6) in ethanol under reflux to install the phenylsulfonyl group, yielding the final Eletriptan (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the shift to this novel synthetic route represents a significant opportunity to optimize the cost structure and reliability of the Eletriptan supply chain. The primary driver of value is the drastic simplification of the raw material portfolio. By removing noble metals and specialized ligands from the bill of materials, the direct material costs are substantially reduced. Moreover, the reliance on commodity chemicals like n-BuLi, ethylene sulfate, and sodium benzenesulfinate ensures a stable and competitive supply market, mitigating the risk of price volatility associated with scarce catalytic metals. This stability is crucial for long-term contract planning and budget forecasting in the pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost center from the production process. In traditional routes, the cost of the catalyst plus the cost of its removal (scavengers, filtration) can be prohibitive. This new method replaces those costs with inexpensive, bulk-available reagents. Furthermore, the simplified workup procedures reduce the consumption of auxiliary materials and energy, leading to lower overall operating expenses. The high yields reported in the patent embodiments further amplify these savings by maximizing the output per unit of input, directly improving the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic catalysts creates a vulnerability in the supply chain. This metal-free route diversifies the supply risk by utilizing reagents that are produced by multiple global manufacturers. The robustness of the chemistry also means that production schedules are less likely to be disrupted by batch failures due to catalyst deactivation or sensitivity. The ability to source key intermediates like the bromo-indole and ethylene sulfate from established vendors ensures a continuous flow of materials, supporting just-in-time manufacturing strategies and reducing inventory holding costs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, using solvents and conditions that are standard in multi-purpose pharmaceutical plants. The absence of heavy metals simplifies the environmental permitting process and reduces the cost of wastewater treatment, as effluent streams do not require specialized metal precipitation steps. This aligns with increasingly strict global environmental regulations, future-proofing the manufacturing asset against regulatory tightening. The straightforward isolation of intermediates allows for easy scale-up from pilot plant to commercial production without the need for specialized equipment, facilitating rapid response to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures within CN102786514B, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of adopting this technology for their specific supply chain needs.
Q: Does this synthesis route require expensive noble metal catalysts?
A: No, the method described in patent CN102786514B specifically avoids the use of noble metal catalysts and strong reductants, relying instead on standard organic reagents like n-BuLi and sodium benzenesulfinate, which significantly lowers raw material costs.
Q: What is the overall purity profile of the Eletriptan produced via this method?
A: The patent indicates that the final product possesses high purity. The stepwise purification processes, including washing with saturated ammonium chloride and brine, along with recrystallization or column chromatography where necessary, ensure a clean impurity profile suitable for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the methodology is designed for industrial suitability. It utilizes common solvents like DCM, THF, and ethanol, and avoids hazardous or difficult-to-handle reagents, making the commercial scale-up of complex pharmaceutical intermediates feasible and safe.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eletriptan Supplier
The technical advancements detailed in patent CN102786514B underscore the potential for more efficient and sustainable production of Eletriptan. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of Eletriptan intermediate we produce. We understand that consistency is key in the pharmaceutical industry, and our quality management systems are designed to deliver products that meet or exceed international regulatory standards.
We invite you to collaborate with us to leverage this superior synthetic route for your Eletriptan requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this metal-free approach can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in securing a reliable, high-quality, and cost-effective supply of this critical migraine medication intermediate.
