Advanced Synthesis of Decarboxylated Carnosine for High-Purity Cosmetic Manufacturing
Advanced Synthesis of Decarboxylated Carnosine for High-Purity Cosmetic Manufacturing
The landscape of cosmetic active ingredient manufacturing is undergoing a significant transformation, driven by the demand for safer, more effective, and economically viable production methods. A pivotal development in this sector is detailed in patent CN110981810A, which discloses a highly efficient synthesis method for decarboxylated carnosine. This molecule, known for its potent antioxidant and anti-glycation properties, is increasingly critical for formulators seeking to address skin aging and oxidative stress without the safety concerns associated with traditional whitening agents like hydroquinone. The patented route represents a substantial leap forward in short peptide synthesis, offering a streamlined pathway that bypasses the cumbersome protection strategies typically required for histamine derivatives. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a reliable supply of high-purity functional ingredients that can be scaled from laboratory benchtops to multi-ton commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex dipeptides like decarboxylated carnosine has been plagued by significant chemical and economic inefficiencies that hinder large-scale adoption. Traditional routes often necessitate the use of multiple protection groups to mask reactive functional sites on the amino acid and amine components, particularly on the imidazole ring of the histamine moiety. This requirement introduces additional synthetic steps, each demanding specific reagents, reaction conditions, and purification protocols, which cumulatively drive up the cost of goods sold and extend the manufacturing lead time. Furthermore, the use of strong catalysts or harsh deprotection conditions in older methodologies can lead to the formation of difficult-to-remove impurities and byproducts, compromising the final purity profile and necessitating expensive chromatographic purification. These factors create a bottleneck for supply chain managers who require consistent, high-volume output, as the complexity of the process increases the risk of batch-to-batch variability and potential production delays.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in the patent introduces a groundbreaking simplification by eliminating the need for side-chain protection on the histamine raw material. By utilizing histamine dihydrochloride directly in the coupling reaction, the process drastically reduces the number of operational steps, thereby minimizing the consumption of protecting group reagents and the associated waste generation. This direct coupling strategy not only simplifies the workflow but also enhances the overall atom economy of the synthesis, leading to a more environmentally friendly process that aligns with modern green chemistry principles. The result is a robust manufacturing protocol that yields a final product with exceptional purity, reported to be greater than or equal to 99%, while maintaining a total yield in the range of 65-75%. This efficiency makes the method particularly attractive for commercial scale-up of complex peptide intermediates, offering a competitive edge in terms of both cost structure and supply reliability.
Mechanistic Insights into Active Ester-Mediated Peptide Coupling
The core of this synthetic innovation lies in the strategic activation of the carboxyl group of Boc-β-Ala-OH to form a highly reactive active ester intermediate. The process begins by dissolving Boc-β-Ala-OH and N-hydroxysuccinimide (HOSU) in dichloromethane (DCM), followed by cooling the system to below 10°C using an ice bath to control the exothermic nature of the activation. The addition of EDC.HCl facilitates the formation of the O-acylisourea intermediate, which rapidly reacts with HOSU to generate the stable Boc-β-Ala-OSU active ester. This specific activation strategy is crucial because it creates an electrophilic center that is sufficiently reactive to couple with the amine of histamine but stable enough to minimize racemization and side reactions. The structural integrity of this activated intermediate is key to the success of the subsequent coupling step, ensuring that the peptide bond forms selectively at the desired position.
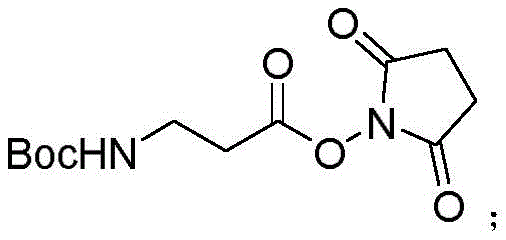
Following the formation of the active ester, the synthesis proceeds to the coupling phase where the nucleophilic attack occurs. The histamine dihydrochloride is neutralized in situ using sodium bicarbonate in an acetonitrile-water system, freeing the amine group to attack the carbonyl carbon of the Boc-β-Ala-OSU. This reaction is conducted under mild conditions, reacting for approximately 2 hours until thin-layer chromatography confirms the complete disappearance of the starting material. The resulting intermediate, Boc-β-Ala-histamine, retains the Boc protecting group on the beta-alanine nitrogen while successfully forming the peptide bond with the histamine side chain, all without requiring prior protection of the histamine imidazole ring. This mechanistic elegance allows for a cleaner reaction profile with fewer byproducts, simplifying the downstream isolation process significantly.
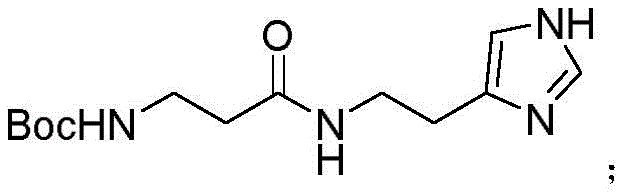
How to Synthesize Decarboxylated Carnosine Efficiently
Implementing this synthesis route in a production environment requires strict adherence to the specified reaction parameters to ensure optimal yield and purity. The process is designed to be operationally simple, relying on standard unit operations such as dissolution, cooling, dropwise addition, and filtration, which are easily scalable in standard reactor vessels. The initial activation step must be carefully monitored to maintain the temperature below 10°C, preventing the decomposition of the active ester, while the subsequent coupling benefits from the biphasic nature of the ACN-water system to solubilize both organic and inorganic components. The final deprotection step utilizes HCl in methyl acetate, a reagent system that efficiently cleaves the Boc group to reveal the free amine, resulting in the final hydrochloride salt of the target molecule. Detailed standardized synthesis steps for this process are provided in the guide below to assist technical teams in replicating these results.
- Activate Boc-β-Ala-OH using HOSU and EDC.HCl in DCM at low temperature to form the active ester intermediate Boc-β-Ala-OSU.
- Couple the active ester with histamine dihydrochloride in an ACN-water system using NaHCO3 as a base to form the protected peptide.
- Perform acid-mediated deprotection using HCl in methyl acetate to remove the Boc group and isolate the final decarboxylated carnosine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible strategic advantages that extend beyond mere chemical yield. The primary value driver is the significant reduction in manufacturing complexity, which directly correlates to lower operational costs and reduced risk of supply disruption. By eliminating the protection and deprotection steps for the histamine side chain, the process removes entire stages of material handling, reagent procurement, and waste disposal, streamlining the entire production timeline. This simplification allows for faster batch turnover and more efficient use of reactor capacity, enabling suppliers to respond more agilely to market demand fluctuations without the need for excessive inventory buffering.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group reagents and the associated solvents for their removal results in a drastic simplification of the bill of materials. Without the need for additional synthetic steps to mask and unmask the histamine functionality, the labor hours and utility consumption per kilogram of product are substantially decreased. This qualitative improvement in process efficiency allows for a more competitive pricing structure, making high-purity decarboxylated carnosine more accessible for mass-market cosmetic formulations while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like Boc-β-Ala-OH and histamine dihydrochloride, combined with common solvents such as DCM and ethyl acetate, mitigates the risk of raw material shortages. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic catalysts or extreme pressures, ensures that production can be maintained consistently across different manufacturing sites. This stability is critical for long-term supply agreements, as it reduces the likelihood of batch failures or delays caused by sensitive process parameters.
- Scalability and Environmental Compliance: The process generates fewer byproducts and utilizes a workup procedure based on simple filtration and washing, which significantly reduces the volume of organic waste requiring treatment. The high purity of the crude product minimizes the need for resource-intensive purification techniques like preparative HPLC, further lowering the environmental footprint of the manufacturing process. This alignment with green chemistry principles facilitates easier regulatory compliance and supports the sustainability goals of downstream customers who are increasingly prioritizing eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of decarboxylated carnosine synthesized via this novel route. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of this technology for potential partners. Understanding these details is crucial for evaluating the suitability of this ingredient for specific formulation requirements and supply chain strategies.
Q: Why is the histamine side chain protection unnecessary in this synthesis?
A: The novel approach utilizes histamine dihydrochloride directly, leveraging the specific reactivity conditions to avoid side reactions on the imidazole ring, thereby eliminating the cost and time associated with protection and deprotection steps.
Q: What is the expected purity of the final decarboxylated carnosine?
A: According to the patent data, the optimized process consistently achieves a final product purity of greater than or equal to 99%, meeting stringent requirements for cosmetic active ingredients.
Q: How does this method improve scalability compared to traditional routes?
A: By reducing the total number of reaction steps and utilizing common solvents like DCM and ethyl acetate with simple filtration workups, the process minimizes operational complexity and facilitates easier scale-up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decarboxylated Carnosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this streamlined synthesis are fully realized in large-scale manufacturing. We are committed to delivering decarboxylated carnosine that meets stringent purity specifications, supported by our rigorous QC labs which verify every batch against the highest industry standards for cosmetic active ingredients.
We invite you to collaborate with us to optimize your supply chain for this high-value ingredient. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient synthesis route can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering expertise can support your product development goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
